An Introduction to Biological Anthropology
Katie Nelson, Ph.D., Inver Hills Community College
Lara Braff, Ph.D., Grossmont College
Beth Shook, Ph.D., California State University, Chico
Kelsie Aguilera, M.A., University of Hawai‘i: Leeward Community College
This introduction is a section of a revision from “Chapter 1: Introduction to Biological Anthropology” by Katie Nelson, Lara Braff, Beth Shook, and Kelsie Aguilera. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Introductory Statement
This preliminary section offers an overview of the discipline of Anthropology. Students who have previously completed an introductory course (such as ANTH 202 or its equivalent) will likely find the material familiar. Although reviewing this section is recommended, those confident in their understanding of Anthropology and its four subdisciplines may choose to proceed directly to Chapter 1. It is important to note that this textbook reflects an American approach to organizing the discipline; in other academic traditions, anthropological knowledge may be divided into different disciplinary frameworks.
Learning Objectives
- Describe anthropology and the four subdisciplines.
- Explain the main anthropological approaches.
- Define biological anthropology, describe its key questions, and identify major subfields
Diving in caves along the Caribbean coast of Mexico, archaeologist Octavio del Rio and his team spotted something unusual in the sand 26 feet below the ocean surface. As they swam closer, they suspected it could be a bone—and likely a very ancient one, as this cave system is inaccessible today without modern diving equipment. However, in the distant past, these caves were dry land formations high above the ocean. The divers ended up recovering not just one but many bones from the site. Eventually they were able to reconstruct an 80% complete human skeleton that they named “Eve of Naharon.” Dated to 13,600 years ago, she is (as of today) the oldest known North American skeleton (TANN 2018).
Who was Eve? What was her life like? How did she end up in the cave? What can we learn about her from the bones she left behind? Anthropologists have determined that Eve was 4.6 feet tall, had a broken back, and died in her early 20s. Although it is rare to find an ancient, nearly complete skeleton in the ocean depths, Eve is not the only such find. In underwater caves along Mexico’s Yucatan Peninsula, archaeologists have found eight well-preserved skeletons dated between 9,000 to 13,000 years old. With each new discovery—whether it is a skeleton in North America, fossil footprints in Tanzania, or a mandible with teeth in China—we come another step closer to understanding the evolution of our species.
Biological anthropologists study when and how human beings evolved; their intriguing findings are the focus of this book. Biological anthropology is one of four subdisciplines within anthropology; the others are cultural anthropology, archaeology, and linguistic anthropology. All anthropological subdisciplines seek to better understand what it means to be human.
What is Anthropology?
Why are people so diverse (Figure 1.1)? Some people live in the frigid Arctic tundra, others in the arid deserts of sub-Saharan Africa, and still others in the dense forests of Papua New Guinea. Human beings speak more than 6,000 distinct languages. Some people are barely five feet tall while others stoop to fit through a standard door frame. What makes people, around the world, look, speak, and behave differently from one another? And what do all humans share in common?

Anthropology is a discipline that explores human differences and similarities by investigating our biological and cultural complexity, past and present. Derived from Greek, the word –anthropos means “human” and –logy refers to the “study of.” Therefore, anthropology is, by definition, the study of humans. Anthropologists are not the only scholars to focus on the human condition; biologists, sociologists, psychologists, and others also examine human nature and societies. However, anthropology is a uniquely dynamic, multifaceted discipline that emerged from a deep-seated curiosity about who we are as a species.
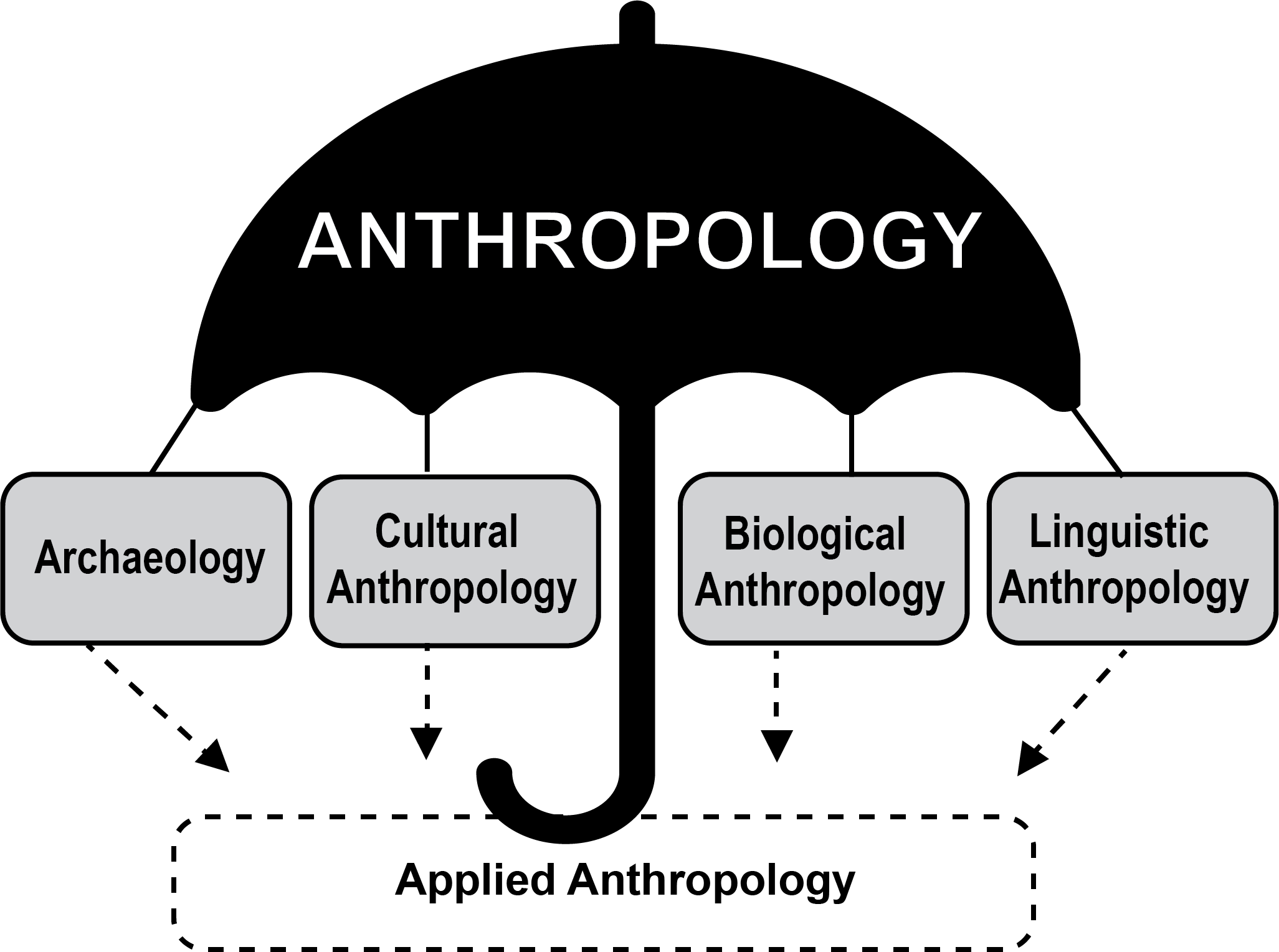
The Subdisciplines
In the United States, the discipline of anthropology includes four subdisciplines: cultural anthropology, biological anthropology, archaeology, and linguistic anthropology. In addition, applied anthropology is sometimes called the fifth subdiscipline (Figure 1.2). Each of the subdisciplines provides a distinct perspective on the human experience. Some (like biological anthropology) use the scientific method to develop theories about human origins, evolution, material remains, or behaviors. Others (like cultural anthropology) use humanistic and interpretive approaches to understand human beliefs, languages, behaviors, cultures, and societies. Findings from all subdisciplines, together, contribute to a multifaceted appreciation of human biocultural experiences, past and present.
Cultural Anthropology
Cultural anthropologists focus on similarities and differences among living persons and societies. They suspend their sense of what is expected in their own culture in order to understand other perspectives without judging them (cultural relativism). They learn these perspectives through participant-observation fieldwork. Beyond describing another way of life, cultural anthropologists ask broader questions about humankind: Are human emotions universal or culturally distinct? Is maternal behavior learned or innate? How and why do groups migrate to new places? For cultural anthropologists, no aspect of human life is outside their purview: They study art, religion, medicine, migration, natural disasters, even video gaming. While many cultural anthropologists are intrigued by human diversity, they recognize that people around the world share much in common.
One famous U.S. cultural anthropologist, Margaret Mead (1901–1978, Figure 1.3), conducted cross-cultural studies of gender and socialization. In the early twentieth century, people in the U.S. wondered if the emotional turbulence exhibited by American adolescents was caused by the biology of puberty, and thus natural and universal. To find out, Mead went to the Samoan Islands, where she lived for several months getting to know Samoan teenagers. She learned that Samoan adolescence was relatively tranquil and happy. Based on her fieldwork, Mead wrote Coming of Age in Samoa, a best-selling book that was both sensational and scandalous (Mead 1928). In it, she critiqued U.S. parenting as restrictive in contrast to Samoan parenting, which allowed teenagers to freely explore their community and even their sexuality. Ultimately, she argued that nurture (i.e., socialization) more than nature (i.e., biology) shaped adolescent development. Despite her expressed relativism, she has been critiqued recently for exploiting the people she studied.

Cultural anthropologists do not always travel far to learn about human experiences. In the 1980s, American anthropologist Philippe Bourgois (1956–) asked how pockets of extreme poverty persist in the United States, a country widely perceived as wealthy with an overall high quality of life compared to other countries. To answer this question, he lived with Puerto Rican drug dealers in East Harlem, contextualizing their experiences both historically and presently, in terms of ongoing social marginalization and institutional racism. Rather than blame drug dealers for their choices, Bourgois argued that both individual choices and social inequality can trap people in the overlapping worlds of drugs and poverty (Bourgois 2003).
Linguistic Anthropology
The study of people is incomplete without attending to language, a defining trait of human beings. While other animals have communication systems, only humans have complex symbolic languages—and more than 6,000 of them! Human language makes it possible to teach and learn, plan and think abstractly, coordinate our efforts, and contemplate our own demise. Linguistic anthropologists ask questions like: How did language first emerge? How has it evolved and diversified over time? How has language helped our species? How can linguistic style convey social identity? How does language influence our worldview? Some linguistic anthropologists track the emergence and diversification of languages, while others focus on language use in social context. Still others explore how language is crucial to socialization: children learn their culture and identities through language and nonverbal forms of communication (Ochs and Schieffelin 2017; Figure 1.4).

One line of linguistic anthropological research focuses on the relationships among language, thought, and culture. For example, Benjamin Whorf (1897–1941) observed that whereas the English language has grammatical tenses to indicate past, present, and future, the Hopi language does not; instead, it indicates whether or not something has “manifested.” Whorf argued that this grammatical difference causes English and Hopi speakers to think about time in distinct ways: English speakers think about time in a linear way, while Hopi think about time in terms of a cycle of things or events that have manifested or are manifesting (Whorf 1956). Based on his research, Whorf developed a strong version of the Sapir-Whorf hypothesis (also known as linguistic relativity), which states that language shapes thought. Some critics, like German American linguist Ekkehart Malotki (1938–), recognized that English and Hopi tenses differ but argued against Whorf by claiming that the Hopi language does, in fact, have linguistic terms for time and that a linear sense of time may be universal (Malotki 1983). Nevertheless, anthropological linguists tend to see human languages as a unique form of communication, linked to our ability to think and process our world.
Archaeology
Archaeologists focus on material remains—tools, pottery, rock art, shelters, seeds, bones, and other objects—to better understand people and societies. Archaeologists ask specific questions like: How did people in a particular area live? How did they utilize their environment? What happened to their society? They also ask general questions about humankind: When did our ancestors begin to walk on two legs? How and why did they leave Africa? Why did humans first develop agriculture? How did the first cities develop?
One critical method that archaeologists use to answer these questions is excavation, which involves carefully digging and removing sediment to uncover material remains while recording their context. Take the example of Kathleen Kenyon (1906–1978), a British archaeologist and one of few female archaeologists in the 1940s. While excavating at Jericho, which dates back to 10,000 BCE (Figure 1.5), she discovered city structures and cemeteries built during the Early Bronze Age (3,200 YBP in Europe). Based on her findings, she argued that Jericho is the oldest city continuously occupied by different groups of people for thousands of years (Kenyon 1979).

While most archaeologists study the past, some excavate at contemporary sites to gain new perspectives on present-day societies. For example, participants in the Garbage Project, which began in the 1970s in Tucson, Arizona, excavate modern landfills as if they were a conventional dig site. They have found that what people say they throw out differs from what is actually in the trash. The landfill holds large amounts of paper products (that people claim to recycle) as well as construction debris (Rathje and Murphy 1992). This finding indicates the need to create more environmentally conscious waste-disposal practices.
Biological Anthropology
Biological anthropology—the focus of this book—is the study of human evolution and biological variation. Some biological anthropologists study our closest living relatives—monkeys and apes—to learn how nonhuman and human primates are alike and how they differ both biologically and behaviorally (Figure 1.6). Other biological anthropologists focus on extinct human species and subspecies, asking questions like: What did they look like? What did they eat? When did they start to speak? How did they adapt to new environments? Still other biological anthropologists focus on modern human diversity, asking questions about the evolution of traits, like lactose tolerance or skin color, that differ between populations. Throughout this book, we will learn about biological anthropological research that explores our nonhuman primate cousins, the origins of hominins (i.e. humans and fossil human relatives), how they adapted over time, and how we – modern humans – continue to change.

Applied Anthropology
Sometimes considered the fifth subdiscipline, applied anthropology involves the practical application of anthropological theories, methods, and findings to solve real-world problems. Applied anthropologists span the subdisciplines. An applied archaeologist might work in cultural resource management to assess a potentially significant archaeological site unearthed during a construction project. An applied cultural anthropologist could work for a technology company that seeks to understand how people interact with their products in order to design them better. Applied anthropologists are employed outside of academic settings, in public and private sectors, including business firms, advertising companies, city government, law enforcement, hospitals, nongovernmental organizations, and even the military.

Trained as both a physician and anthropologist, Paul Farmer (1959–2022, Figure 1.7) demonstrated the potential of applied anthropology to improve lives. As a college student in North Carolina, Farmer became interested in the Haitian migrants working on nearby farms. This led him to visit Haiti, the most resource-poor country in the Western Hemisphere, where he was struck by the deprived state of its health care facilities. Years later, he would return to Haiti, as a physician, to treat diseases that had been largely eradicated in the United States, such as tuberculosis and cholera. Drawing on his anthropological training, he also did fieldwork and wrote books that contextualize the suffering of Haitians in relation to historical, social, and political conditions (Farmer 2006). Finally, as an applied anthropologist, he took action by co-founding Partners in Health, a nonprofit organization that establishes health clinics in resource-poor countries and trains local staff to administer care.
Anthropological Approaches
Each of the four main anthropological subdisciplines contributes to our understanding of humankind by exploring cultures, languages, material remains, and biological adaptations. To study these phenomena, anthropologists draw upon distinct research approaches, including holism, comparison, dynamism, and fieldwork.
Holism
Anthropologists are interested in the whole of humanity. We look at the interactions among several aspects of our complex bodies or societies, rather than focusing on a singular aspect (Figure 1.8). For example, a biological anthropologist studying the social behaviors of a monkey species in South America may not only observe how they interact with one another, but also how physical adaptations, foraging patterns, ecological conditions, and habitat changes also affect their behaviors. By focusing on only one factor, the anthropologist would attain an incomplete understanding of the species’ social life. A cultural anthropologist studying marriage in a small village in India would not only consider local gender norms but also family networks, laws regarding marriage, religious rules, and economic factors. All of these aspects can influence marital practices in a given context. In both examples, the anthropologist is using a holistic approach by considering the multiple interrelated and intersecting factors that comprise a given phenomena.
Comparison
Anthropologists use comparative approaches to compare and contrast data from different populations, from groups within a population, or from the same group over time. For example: How do humans today differ from prior Homo sapiens populations? How does Egyptian society today compare to ancient Egyptian society? How do male and female behaviors differ within a given human society or a particular primate group? Comparative analyses help us understand commonalities and differences within or across species, groups, or time.
Dynamism
Comparative analysis is facilitated by the fact that humans are extremely dynamic. Our ability to change, both biologically and culturally, has enabled us to persist over millions of years and to thrive in different environments. Anthropologists ask about all kinds of changes: short-term and long-term, temporary and permanent, cultural and biological. For example, a cultural anthropologist might look at how people in a relatively isolated society are affected by globalization. A linguistic anthropologist might explore how a hybrid form of language, like Spanglish, emerged. An archaeologist might study how climate change influenced the emergence of agriculture. A biological anthropologist might consider how diseases affecting our ancestors led to physical changes that persist today. All of these examples highlight the dynamic nature of human bodies and societies.

Fieldwork
Throughout this book, you will read examples of anthropological research from around the world. Anthropologists do not only work in laboratories, libraries, or offices. To collect data, they travel to where their data lives, whether it is a city, village, cave, tropical forest, or desert. At their field sites, anthropologists collect data that, depending on subdiscipline, may include interviews with local peoples (Figure 1.9), examples of language in use, skeletal features, or cultural remains like stone tools. While anthropologists ask an array of questions and use diverse methods to answer their research questions, they share this commitment to conducting research in the field.
What is Biological Anthropology?
Biological anthropology uses a scientific and evolutionary approach to answer many of the same questions that all anthropologists are concerned with: What does it mean to be human? Where do we come from? Who are we today? Biological anthropologists are concerned with exploring how humans vary biologically, how humans adapt to their changing environments, and how humans have evolved over time and continue to evolve today. Some biological anthropologists also study what humans and nonhuman primates have in common and how we differ.
You may have heard biological anthropology referred to by another name—physical anthropology. Physical anthropology is a discipline that dates to as far back as the eighteenth century, when it focused mostly on physical variation among humans. Some early physical anthropologists were also physicians or anatomists interested in comparing and contrasting the human form. These researchers dedicated themselves to measuring bodies and skulls (anthropometry and craniometry) in great detail (Figure 1.10). Many also acted under the misguided racist belief that human biological races existed and that it was possible to differentiate between, or even rank, such races by measuring differences in human anatomy. Anthropologists today agree that there are no biological human races and that all humans alive today are members of the same species, Homo sapiens, and subspecies, Homo sapiens sapiens. We recognize that the differences we can see between peoples’ bodies are due to a wide variety of factors, including environment, diet, activities, and genetic makeup.
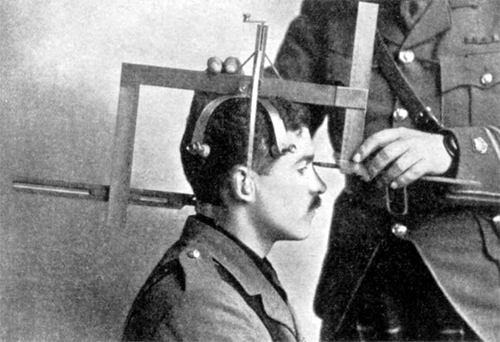
The subdiscipline has changed a great deal since these early years. Biological anthropologists no longer identify human differences in order to assign people to groups, like races. The focus is instead on understanding how and why human and primate variation developed through evolutionary processes. The name for the subdiscipline has transitioned in recent years (from physical anthropology to biological anthropology) to reflect these changes. Many believe the term biological anthropology better reflects the subdiscipline’s focus today, which includes genetic and molecular research.
The Scope of Biological Anthropology
Just as anthropology as a discipline is wide ranging and holistic, so too is the subdiscipline of biological anthropology. There are at least six subfields within biological anthropology (Figure 1.11): primatology, paleoanthropology, molecular anthropology, bioarchaeology, forensic anthropology, and human biology. Each subfield focuses on a different dimension of what it means to be human from a biological perspective. Through their varied research in these subfields, biological anthropologists try to answer the following key questions:
- What is our place in nature? How are we related to other organisms? What makes us unique?
- What are our origins? What influenced our evolution?
- How and when did we move/migrate across the globe?
- How are humans around the world today different from and similar to each other? What influences these patterns of variation? What are the patterns of our recent evolution and how do we continue to evolve?
The terms subfield and subdiscipline are very similar and are often used interchangeably. In this book we use subdiscipline to refer to the four major areas of focus that make up the discipline of anthropology: biological anthropology, cultural anthropology, archaeological anthropology, and linguistic anthropology. When we use the term subfield we are referring to the different specializations within biological anthropology.

Primatology
Primatologists study the anatomy, behavior, ecology, and genetics of living and extinct nonhuman primates, including apes, monkeys, tarsiers, lemurs, and lorises. Primatology research gives us insights into how evolution has shaped our species, since nonhuman primates are our closest living biological relatives. Through such studies, we have learned that all primates share a suite of traits. Primates, for instance, have nails instead of claws, possess hands that can grasp and manipulate objects (Figure 1.12), invest great amounts of time and energy in raising a small number of offspring, and employ complex social behaviors. Behavioral studies, such as those by Jane Goodall of wild chimpanzees and others, reveal that great apes are like humans in that they have families and form strong maternal-infant relationships. Gorillas mourn the deaths of their group members, and they exhibit behaviors similar to humans such as playing and tickling. Importantly, the work of Goodall, Karen B. Strier (see Appendix B), and others focus on primate conservation: They have brought attention to the fact that 60% of primates are currently threatened with extinction (Estrada et al. 2017).

Paleoanthropology
Paleoanthropologists study human ancestors from the distant past to learn how, why, and where they evolved. Because these ancestors lived before there were written records, paleoanthropologists have to rely on various types of physical evidence to come to their conclusions. This evidence includes fossilized remains (particularly fossilized bones; Figure 1.13), DNA, artifacts such as stone tools, and the contexts in which these items are found. In recent years, paleoanthropologists have made some monumental discoveries about hominin evolution.

These findings helped us learn that human evolution did not occur in a simple, straight line but, rather, branched out in many directions. Most branches were evolutionary “dead ends.” Humans are now the only living hominins left on planet Earth. Paleoanthropologists frequently work together with other scientists such as archaeologists, geologists, and paleontologists to interpret and understand the evidence they find. Paleoanthropology is a dynamic subfield of biological anthropology that contributes to our understanding of human origins and evolution.
Molecular Anthropology
Molecular anthropologists use molecular techniques (primarily genetics) to compare ancient and modern populations as well as to study living populations of humans or nonhuman primates. By examining DNA sequences, molecular anthropologists can estimate how closely related two populations are, as well as identify population events, like a population decline, that explain the observed genetic patterns. This information helps scientists trace patterns of migration and identify how people have adapted to different environments over time.
Several molecular anthropologists have recently attracted international recognition for their groundbreaking work. For instance, in 2022, Svante Pääbo won the Nobel Prize in physiology (medicine) for his work extracting the DNA from 40,000-year-old Neanderthal bones and producing the first complete genome of Homo neanderthalensis. This was a challenging task because ancient DNA does not preserve well and older extraction techniques tended to become contaminated by the researcher’s and other environmental DNA. Pääbo and his team designed specialized clean rooms for handling ancient DNA and made advances in DNA sequencing. Their research helped scientists identify genetic differences between modern humans and Neanderthals and analyze how those differences influence how diseases, such as COVID-19, affect our bodies. Molecular anthropology is a quickly changing field as new techniques and discoveries shape our understanding of ourselves, our ancestors, and our nonhuman primate relatives.
Bioarchaeology
Bioarchaeologists study human skeletal remains along with the surrounding soils and other materials. They use the research methods of skeletal biology, mortuary studies, osteology, and archaeology to answer questions about the lifeways of past populations. Through studying the bones and burials of past peoples, bioarchaeologists search for answers to how people lived and died, including their health, nutrition, diseases, and/or injuries. Most bioarchaeologists study not just individuals but entire populations to reveal biological and cultural patterns.
People have always been intrigued by the remains of the dead, however historically, human bodies were often studied isolated from the ground and location where they were found. Bioarchaeologists emphasize the context in and around where the remains are found, using a biocultural approach that studies humans through an understanding of the interconnectedness of biology, culture, and environment.
Forensic Anthropology
Forensic anthropologists use many of the same techniques as bioarchaeologists to develop a biological profile for unidentified individuals, including estimating sex, age at death, height, ancestry, or other unique identifying features such as skeletal trauma or diseases. They may also go to a crime or accident scene to assist in the search and recovery of human remains, aiding law enforcement teams (Figure 1.14). The popular television show Bones told the fictional story of a forensic anthropologist, Dr. Temperance Brennan, who brilliantly interpreted clues from victims’ bones to help solve crimes. While the show includes forensic anthropology techniques and responsibilities, it also includes many inaccuracies. For example, forensic anthropologists do not collect trace evidence like hair or fibers, run DNA tests, carry weapons, or solve criminal cases.

Forensic anthropology is considered an applied area of biological anthropology, because it involves a practical application of anthropological theories, methods, and findings to solve real-world problems. While some forensic anthropologists are academics that work for colleges and universities, others are employed by public safety and law agencies.
Human Biology
Many biological anthropologists do work that falls under the label of “human biology.” This type of research explores how the human body is affected by different physical environments, cultural influences, and nutrition. These include studies of human variation or the physiological differences among humans around the world. Some of these anthropologists study human adaptations to extreme environments, which includes physiological responses and genetic advantages to help them survive. Others are interested in how nutrition and disease affect human growth and development. Biological anthropologists engage in a wide range of research that spans the breadth of human biological diversity.
The six subfields of biological anthropology—primatology, paleoanthropology, molecular anthropology, bioarchaeology, forensic anthropology, and human biology—all help us to understand what it means to be biologically human. From molecular analyses of our cells to studies of our changing skeleton, to research on our nonhuman primate cousins, biological anthropology assists in answering the central question of anthropology: What does it mean to be human? Despite their different foci, all biological anthropologists share a commitment to using a scientific approach to study how we became the complex, adaptable species we are today.
Key Terms
Belief: A firmly held opinion or conviction typically based on spiritual apprehension rather than empirical proof.
Cultural relativism: The anthropological practice of suspending judgment and seeking to understand another culture on its own terms sympathetically enough so that the culture appears to be a coherent and meaningful design for living.
Holism: The idea that the parts of a system interconnect and interact to make up the whole.
Hominins: Species that are regarded as human, directly ancestral to humans, or very closely related to humans.
Human adaptation: The ways in which human bodies, people, or cultures change, often in ways better suited to the environment or social context.
Human variation: The range of forms of any human characteristic, such as body shape or skin color.
Hypothesis: Explanation of observed facts; details how and why observed phenomena are the way they are. Scientific hypotheses rely on empirical evidence, are testable, and are able to be refuted.
Indigenous: Refers to people who are the original settlers of a given region and have deep ties to that place. Also known as First Peoples, Aboriginal Peoples, or Native Peoples, these populations are in contrast to other groups who have settled, occupied, or colonized the area more recently.
Law: A prediction about what will happen given certain conditions; typically mathematical.
Sapir-Whorf hypothesis: The principle that the language you speak allows you to think about some things and not other things. This is also known as the linguistic relativity hypothesis.
Subdisciplines: The four major areas that make up the discipline of anthropology: biological anthropology, cultural anthropology, archaeology, and linguistic anthropology. Applied anthropology is sometimes considered to be a fifth subdiscipline.
Subfield: In this textbook, subfield refers to the different specializations within biological anthropology, including primatology, paleoanthropology, molecular anthropology, bioarchaeology, forensic anthropology, and human biology.
For Further Exploration
American Anthropological Association website.
American Association of Biological Anthropologists website.
8, 2023
References
Binford, Leigh. 2016. The El Mozote Massacre: Human Rights and Global Implications. Revised and expanded edition. Tucson: University of Arizona Press.
Estrada, Alejandro, Paul A. Garber, Anthony B. Rylands, Christian Roos, Eduardo Fernandez-Duque, Anthony Di Fiore, K. Anne-Isola Nekaris, et al. 2017. “Impending Extinction Crisis of the World’s Primates: Why Primates Matter.” Science Advances 3(229): 1–16.
Farmer, Paul. 2006. AIDS and Accusation: Haiti and the Geography of Blame. Berkeley: University of California Press.
Farmer, Paul, Matthew Basilico, Vanessa Kerry, Madeleine Ballard, Anne Becker, Gene Bukhman, Ophelia Dahl, et al. 2013. “Global Health Priorities for the Early Twenty-first Century.” In Reimagining Global Health: An Introduction, edited by Paul Farmer, Jim Yong Kim, Arthur Kleinman, and Matthew Basilico, 302–339. Berkeley: University of California Press.
Kenyon, Kathleen. 1979. Archaeology in the Holy Land. New York: W.W. Norton.
Malotki, Ekkehart. 1983. Hopi Time: A Linguistic Analysis of the Temporal Concepts in the Hopi Language. Berlin: De Gruyter.
Mead, Margaret. 1928. Coming of Age in Samoa. Oxford: Morrow.
Ochs, Elinor and Bambi Schieffelin. 2017. “Language Socialization: An Historical Overview.” In Encyclopedia of Language and Education, Volume 8, edited by Patricia Duff, 3-16. New York: Springer.
Rathje, William and Cullen Murphy. 1992. “Five Major Myths about Garbage, and Why They’re Wrong.” Smithsonian 23, no. 4: 113-122.
TANN. 2018. “Mexican Anthropologists Put Face on Nearly 14,000-Year-Old Woman.” Archaeology News Network, August 19, 2018. Accessed on November 16, 2022.
Whorf, Benjamin. 1956. Language, Thought, and Reality. Cambridge: MIT Press.
Acknowledgment
We are grateful to the anonymous reviewers for their many insightful comments and suggestions.
Keith Chan, Ph.D., Grossmont-Cuyamaca Community College District and MiraCosta College
Student contributors to this chapter: Lily Berruyer, Lyn Loytchenko, and Sarah Cupidio
This chapter is a revision from "Chapter 12: Modern Homo sapiens” by Keith Chan. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Identify the skeletal and behavioral traits that represent modern Homo sapiens.
- Critically evaluate different types of evidence for the origin of our species in Africa and our expansion around the world.
- Understand how the human lifestyle changed when people transitioned from foraging to agriculture.
- Hypothesize how human evolutionary trends may continue into the future.
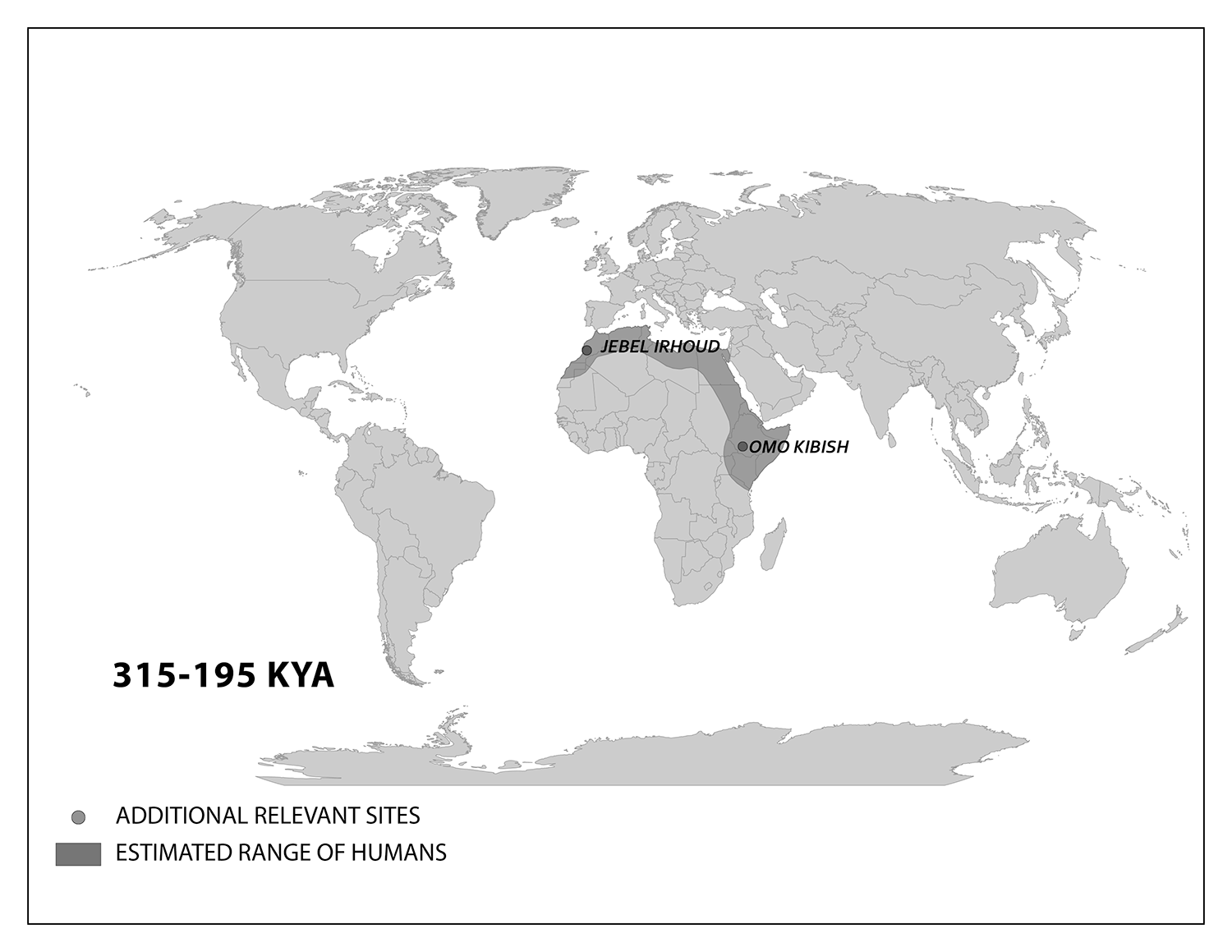
The walls of a pink limestone cave in the hillside of Jebel Irhoud jutted out of the otherwise barren landscape of the Moroccan desert (Figure 13.1). Miners had excavated the cave in the 1960s, revealing some fossils. In 2007, a re-excavation of the site became a momentous occasion for science. A fossil cranium unearthed by a team of researchers was barely visible to the untrained eye. Just the fossil’s robust brows were peering out of the rock. This research team from the Max Planck Institute for Evolutionary Anthropology was the latest to explore the ancient human presence in this part of North Africa after a find by miners in 1960. Excavating near the first discovery, the researchers wanted to learn more about how Homo sapiens lived far from East Africa, where we thought our species originated.

The scientists were surprised when they analyzed the cranium, named Irhoud 10, and other fossils. Statistical comparisons with other human crania concluded that the Irhoud face shapes were typical of recent modern humans while the braincases matched ancient modern humans. Based on the findings of other scientists, the team expected these modern Homo sapiens fossils to be around 200,000 years old. Instead, dating revealed that the cranium had been buried for around 315,000 years.
Together, the modern-looking facial dimensions and the older date reshaped the interpretation of our species: modern Homo sapiens. Some key evolutionary changes from the archaic Homo sapiens (described in Chapter 12) to our species today happened 100,000 years earlier than we had thought and across the vast African continent rather than concentrated in its eastern region.
This revelation in the study of modern Homo sapiens is just one of the latest in this continually advancing area of biological anthropology. Researchers today are still discovering amazing fossils and ingenious ways to collect data and test hypotheses about our past. Through the collective work of many scientists, we are building an overall theory of modern human origins.
Defining Modernity
What defines modern Homo sapiens when compared to archaic Homo sapiens? Modern humans, like you and me, have a set of derived traits that are not seen in archaic humans or any other hominin. As with other transitions in hominin evolution, such as increasing brain size and bipedal ability, modern traits do not appear fully formed or all at once. In other words, the first modern Homo sapiens was not just born one day from archaic parents. The traits common to modern Homo sapiens appeared in a mosaic manner: gradually and out of sync with one another. There are two areas to consider when tracking the complex evolution of modern human traits. One is the physical change in the skeleton. The other is behavior inferred from the size and shape of the cranium and material culture evidence.
Skeletal Traits
The skeleton of modern Homo sapiens is less robust than that of archaic Homo sapiens. In other words, the modern skeleton is gracile, meaning that the structures are thinner and smoother. Differences related to gracility in the cranium are seen in the braincase, the face, and the mandible. There are also broad differences in the rest of the skeleton.
Cranial Traits
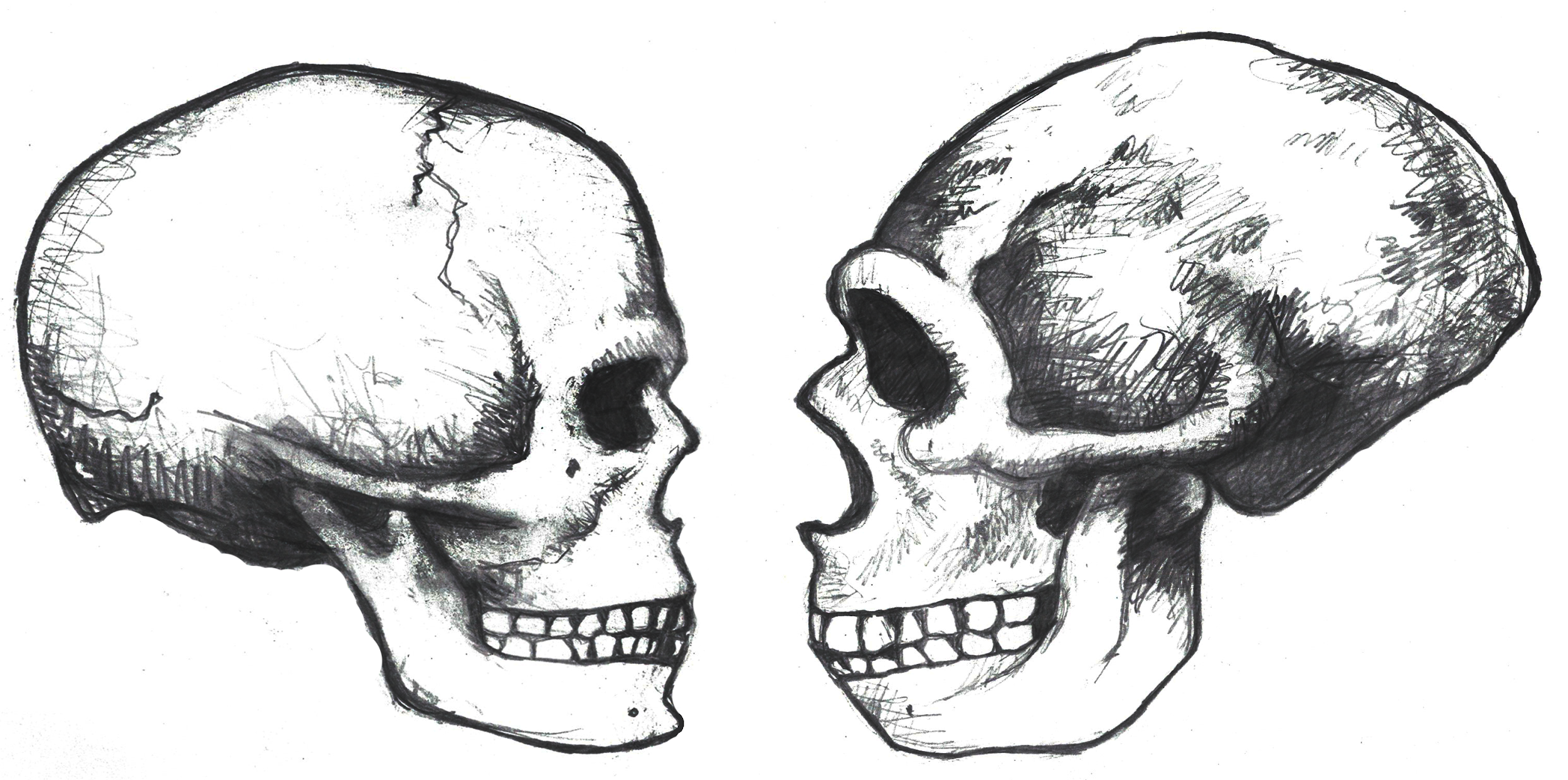
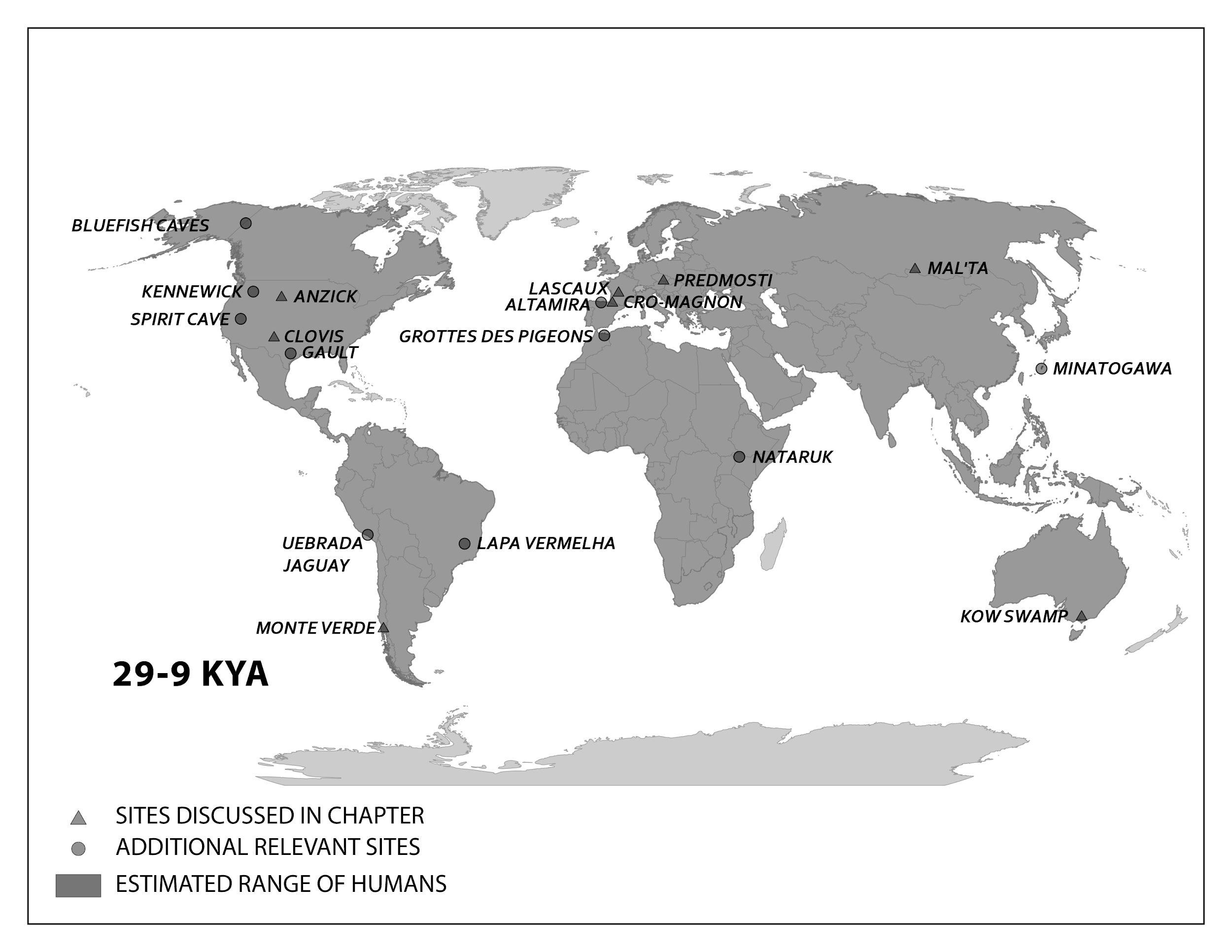
Several elements of the braincase differ between modern and archaic Homo sapiens. Overall, the shape is much rounder, or more globular, on a modern skull (Lieberman, McBratney, and Krovitz 2002; Neubauer, Hublin, and Gunz 2018; Pearson 2008; Figure 13.2). You can feel the globularity of your own modern human skull. Feel the height of your forehead with the palm of your hand. Viewed from the side, the tall vertical forehead of a modern Homo sapiens stands out when compared to the sloping archaic version. This is because the frontal lobe of the modern human brain is larger than the one in archaic humans, and the skull has to accommodate the expansion. The vertical forehead reduces a trait that is common to all other hominins: the brow ridge or supraorbital torus. The parietal lobes of the brain and the matching parietal bones on either side of the skull both bulge outward more in modern humans. At the back of the skull, the archaic occipital bun is no longer present. Instead, the occipital region of the modern human cranium has a derived tall and smooth curve, again reflecting the globular brain inside.
The trend of shrinking face size across hominins reaches its extreme with our species as well. The facial bones of a modern Homo sapiens are extremely gracile compared to all other hominins (Lieberman, McBratney, and Krovitz 2002). Continuing a trend in hominin evolution, technological innovations kept reducing the importance of teeth in reproductive success (Lucas 2007). As natural selection favored smaller and smaller teeth, the surrounding bone holding these teeth also shrank.
Related to smaller teeth, the mandible is also gracile in modern humans when compared to archaic humans and other hominins. Interestingly, our mandibles have pulled back so far from the prognathism of earlier hominins that we gained an extra structure at the most anterior point, called the mental eminence. You know this structure as the chin. At the skeletal level, it resembles an upside-down “T” at the centerline of the mandible (Pearson 2008). Looking back at archaic humans, you will see that they all lack a chin. Instead, their mandibles curve straight back without a forward point. What is the chin for and how did it develop? Flora Gröning and colleagues (2011) found evidence of the chin’s importance by simulating physical forces on computer models of different mandible shapes. Their results showed that the chin acts as structural support to withstand strain on the otherwise gracile mandible.
Postcranial Gracility
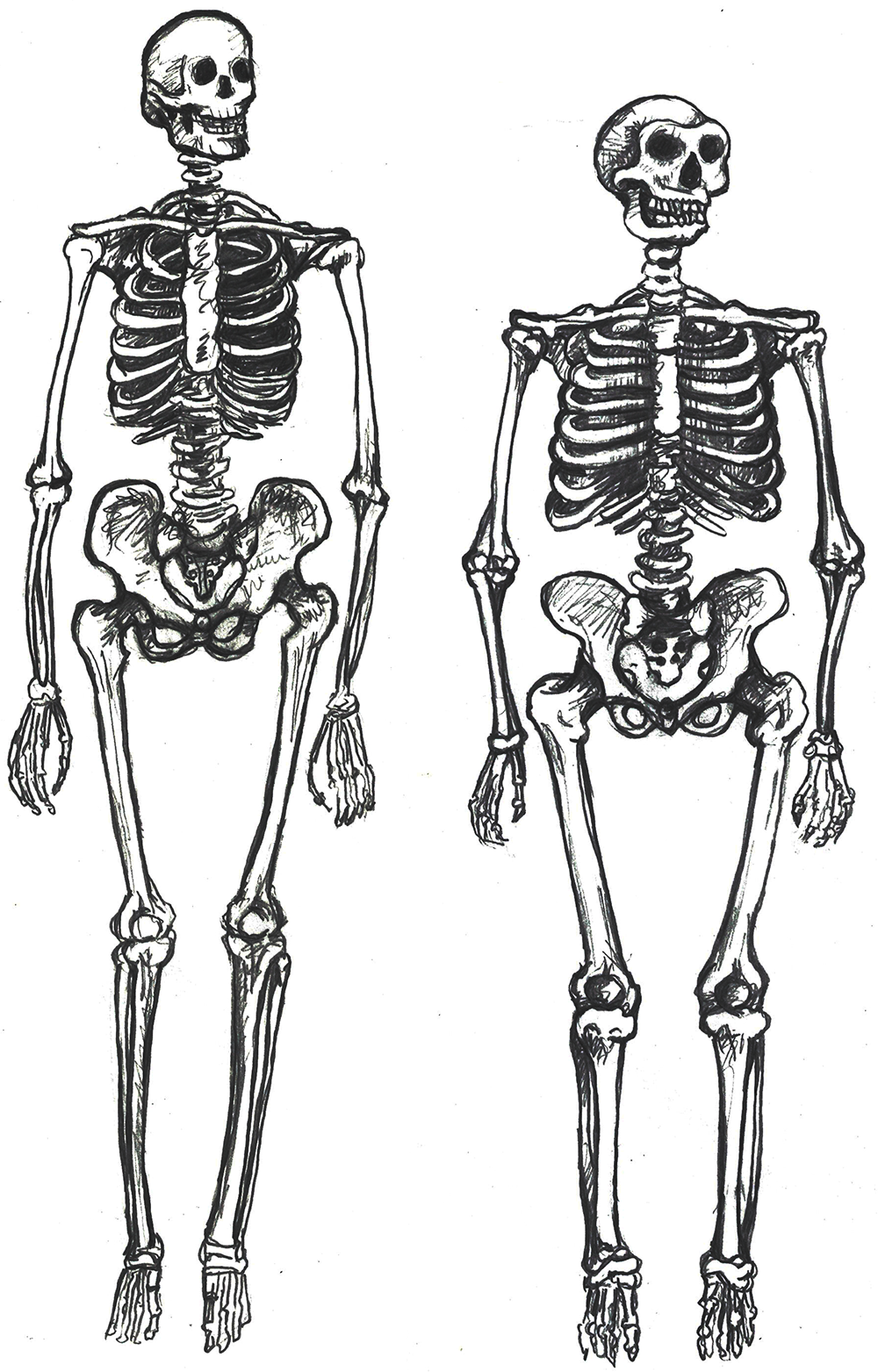
The rest of the modern human skeleton is also more gracile than its archaic counterpart. The differences are clear when comparing a modern Homo sapiens with a cold-adapted Neanderthal (Sawyer and Maley 2005), but the trends are still present when comparing modern and archaic humans within Africa (Pearson 2000). Overall, a modern Homo sapiens postcranial skeleton has thinner cortical bone, smoother features, and more slender shapes when compared to archaic Homo sapiens (Figure 13.3). Comparing whole skeletons, modern humans have longer limb proportions relative to the length and width of the torso, giving us lankier outlines.
Why is our skeleton so gracile compared to those of other hominins? Natural selection can drive the gracilization of skeletons in several ways (Lieberman 2015). A slender frame is believed to be adapted for the efficient long-distance running ability that started with Homo erectus. Furthermore, it is argued that slenderness is a genetic adaptation for cooling an active body in hotter climates, which aligns with the ample evidence that Africa was the home continent of our species.
Behavioral Modernity
Aside from physical differences in the skeleton, researchers have also uncovered evidence of behavioral changes associated with increased cultural complexity from archaic to modern humans. How did cultural complexity develop? Two investigations into this question are archaeology and the analysis of reconstructed brains.
Archaeology tells us much about the behavioral complexity of past humans by interpreting the significance of material culture. In terms of advanced culture, items created with an artistic flair, or as decoration, speak of abstract thought processes (Figure 13.4). The demonstration of difficult artistic techniques and technological complexity hints at social learning and cooperation as well. According to paleoanthropologist John Shea (2011), one way to track the complexity of past behavior through artifacts is by measuring the variety of tools found together. The more types of tools constructed with different techniques and for different purposes, the more modern the behavior. Researchers are still working on an archaeological way to measure cultural complexity that is useful across time and place.

The interpretation of brain anatomy is another promising approach to studying the evolution of human behavior. When looking at investigations on this topic in modern Homo sapiens brains, researchers found a weak association between brain size and test-measured intelligence (Pietschnig et al. 2015). Additionally, they found no association between intelligence and biological sex. These findings mean that there are more significant factors that affect tested intelligence than just brain size. Since the sheer size of the brain is not useful for weighing intelligence within a species, paleoanthropologists are instead investigating the differences in certain brain structures. The differences in organization between modern Homo sapiens brains and archaic Homo sapiens brains may reflect different cognitive priorities that account for modern human culture. As with the archaeological approach, new discoveries will refine what we know about the human brain and apply that knowledge to studying the distant past.
Taken together, the cognitive abilities in modern humans may have translated into an adept use of tools to enhance survival. Researchers Patrick Roberts and Brian A. Stewart (2018) call this concept the generalist-specialist niche: our species is an expert at living in a wide array of environments, with populations culturally specializing in their own particular surroundings. The next section tracks how far around the world these skeletal and behavioral traits have taken us.
First Africa, Then the World
What enabled modern Homo sapiens to expand its range further in 300,000 years than Homo erectus did in 1.5 million years? The key is the set of derived biological traits from the last section. It is theorized that the gracile frame and neurological anatomy allowed modern humans to survive and even flourish in the vastly different environments they encountered. Based on multiple types of evidence, the source of all of these modern humans was Africa. Instead of originating from just one location, evidence shows that modern Homo sapiens evolution occurred in a complex gene flow network across Africa, a concept called African multiregionalism (Scerri et al. 2018).
This section traces the origin of modern Homo sapiens and the massive expansion of our species across all of the continents (except Antarctica) by 12,000 years ago. While modern Homo sapiens first shared geography with archaic humans, modern humans eventually spread into lands where no human had gone before. Figure 13.5 shows the broad routes that our species took expanding around the world. I encourage you to make your own timeline with the dates in this part to see the overall trends.
Modern Homo sapiens Biology and Culture in Africa
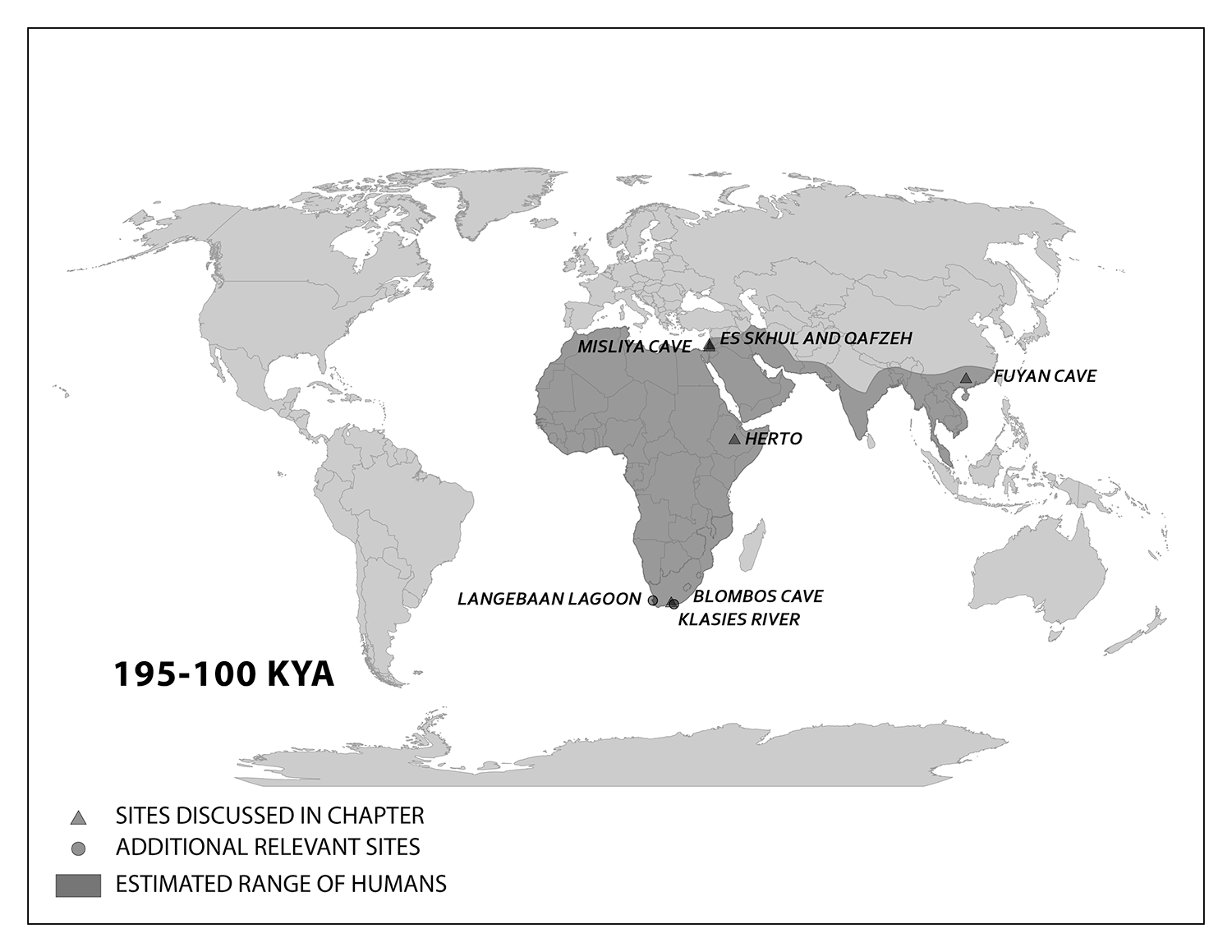
We start with the ample fossil evidence supporting the theory that modern humans originated in Africa during the Middle Pleistocene, having evolved from African archaic Homo sapiens. The earliest dated fossils considered to be modern actually have a mosaic of archaic and modern traits, showing the complex changes from one type to the other. Experts have various names for these transitional fossils, such as Early Modern Homo sapiens or Early Anatomically Modern Humans. However they are labeled, the presence of some modern traits means that they illustrate the origin of the modern type. Three particularly informative sites with fossils of the earliest modern Homo sapiens are Jebel Irhoud, Omo, and Herto.
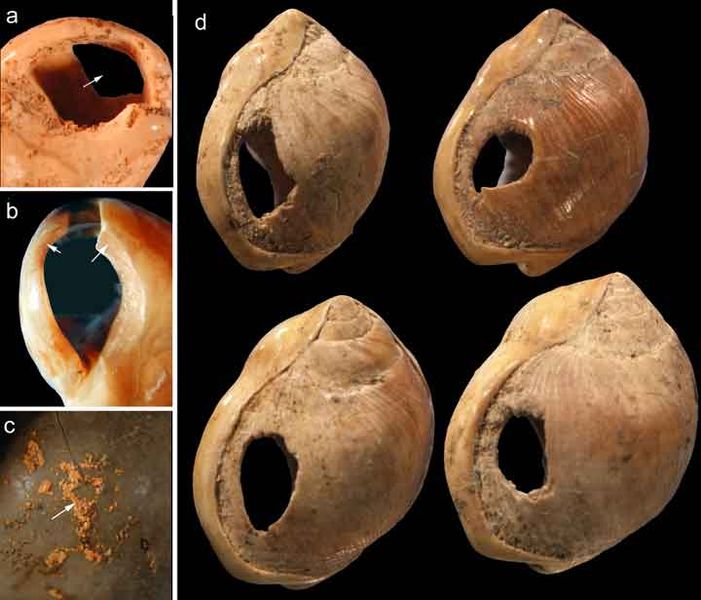
Recall from the start of the chapter that the most recent finds at Jebel Irhoud are now the oldest dated fossils that exhibit some facial traits of modern Homo sapiens. Besides Irhoud 10, the cranium that was dated to 315,000 years ago (Hublin et al. 2017; Richter et al. 2017), there were other fossils found in the same deposit that we now know are from the same time period. In total there are at least five individuals, representing life stages from childhood to adulthood. These fossils form an image of high variation in skeletal traits. For example, the skull named Irhoud 1 has a primitive brow ridge, while Irhoud 2 and Irhoud 10 do not (Figure 13.6). The braincases are lower than what is seen in the modern humans of today but higher than in archaic Homo sapiens. The teeth also have a mix of archaic and modern traits that defy clear categorization into either group.
Research separated by nearly four decades uncovered fossils and artifacts from the Kibish Formation in the Lower Omo Valley in Ethiopia. These Omo Kibish hominins were represented by braincases and fragmented postcranial bones of three individuals found kilometers apart, dating back to around 233,000 years ago (Day 1969; McDougall, Brown, and Fleagle 2005; Vidal et al. 2022). One interesting finding was the variation in braincase size between the two more-complete specimens: while the individual named Omo I had a more globular dome, Omo II had an archaic-style long and low cranium.
Also in Ethiopia, a team led by Tim White (2003) excavated numerous fossils at Herto. There were fossilized crania of two adults and a child, along with fragments of more individuals. The dates ranged between 160,000 and 154,000 years ago. The skeletal traits and stone-tool assemblage were both intermediate between the archaic and modern types. Features reminiscent of modern humans included a tall braincase and thinner zygomatic (cheek) bones than those of archaic humans (Figure 13.7). Still, some archaic traits persisted in the Herto fossils, such as the supraorbital tori. Statistical analysis by other research teams concluded that at least some cranial measurements fit just within the modern human range (McCarthy and Lucas 2014), favoring categorization with our own species.

The timeline of material culture suggests a long period of relying on similar tools before a noticeable diversification of artifacts types. Researchers label the time of stable technology shared with archaic types the Middle Stone Age, while the subsequent time of diversification in material culture is called the Later Stone Age.
In the Middle Stone Age, the sites of Jebel Irhoud, Omo, and Herto all bore tools of the same flaked style as archaic assemblages, even though they were separated by almost 150,000 years. The consistency in technology may be evidence that behavioral modernity was not so developed. No clear signs of art dating back this far have been found either. Other hypotheses not related to behavioral modernity could explain these observations. The tool set may have been suitable for thriving in Africa without further innovation. Maybe works of art from that time were made with media that deteriorated or perhaps such art was removed by later humans.
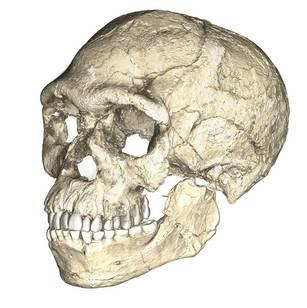
Evidence of what Homo sapiens did in Africa from the end of the Middle Stone Age to the Later Stone Age is concentrated in South African cave sites that reveal the complexity of human behavior at the time. For example, Blombos Cave, located along the present shore of the Cape of Africa facing the Indian Ocean, is notable for having a wide variety of artifacts. The material culture shows that toolmaking and artistry were more complex than previously thought for the Middle Stone Age. In a layer dated to 100,000 years ago, researchers found two intact ochre-processing kits made of abalone shells and grinding stones (Henshilwood et al. 2011). Marine snail shell beads from 75,000 years ago were also excavated (Figure 13.8; d’Errico et al. 2005). Together, the evidence shows that the Middle Stone Age occupation at Blombos Cave incorporated resources from a variety of local environments into their culture, from caves (ochre), open land (animal bones and fat), and the sea (abalone and snail shells). This complexity shows a deep knowledge of the region’s resources and their use—not just for survival but also for symbolic purposes.
On the eastern coast of South Africa, Border Cave shows new African cultural developments at the start of the Later Stone Age. Paola Villa and colleagues (2012) identified several changes in technology around 43,000 years ago. Stone-tool production transitioned from a slower process to one that was faster and made many microliths, small and precise stone tools. Changes in decorations were also found across the Later Stone Age transition. Beads were made from a new resource: fragments of ostrich eggs shaped into circular forms resembling present-day breakfast cereal O’s (d’Errico et al. 2012). These beads show a higher level of altering one’s own surroundings and a move from the natural to the abstract in terms of design.
Expansion into the Middle East and Asia
While modern Homo sapiens lived across Africa, some members eventually left the continent. These pioneers could have used two connections to the Middle East or West Asia. From North Africa, they could have crossed the Sinai Peninsula and moved north to the Levant, or eastern Mediterranean. Finds in that region show an early modern human presence. Other finds support the Southern Dispersal model, with a crossing from East Africa to the southern Arabian Peninsula through the Straits of Bab-el-Mandeb. It is tempting to think of one momentous event in which people stepped off Africa and into the Middle East, never to look back. In reality, there were likely multiple waves of movement producing gene flow back and forth across these regions as the overall range pushed east. The expanding modern human population could have thrived by using resources along the southern coast of the Arabian Peninsula to South Asia, with side routes moving north along rivers. The maximum range of the species then grew across Asia.
Modern Homo sapiens in the Middle East
Geographically, the Middle East is the ideal place for the African modern Homo sapiens population to inhabit upon expanding out of their home continent. In the Eastern Mediterranean coast of the Levant, there is a wealth of skeletal and material culture linked to modern Homo sapiens. Recent discoveries from Saudi Arabia further add to our view of human life just beyond Africa.
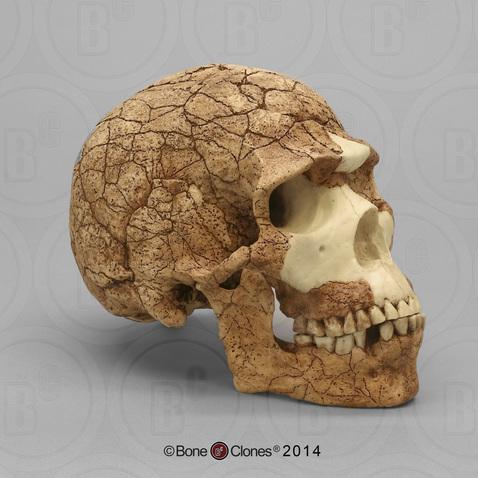
The Caves of Mount Carmel in present-day Israel have preserved skeletal remains and artifacts of modern Homo sapiens, the first-known group living outside Africa. The skeletal presence at Misliya Cave is represented by just part of the left upper jaw of one individual, but it is notable for being dated to a very early time, between 194,000 and 177,000 years ago (Hershkovitz et al. 2018). Later, from 120,000 to 90,000 years ago, fossils of multiple individuals across life stages were found in the caves of Es-Skhul and Qafzeh (Shea and Bar-Yosef 2005). The skeletons had many modern Homo sapiens traits, such as globular crania and more gracile postcranial bones when compared to Neanderthals. Still, there were some archaic traits. For example, the adult male Skhul V also possessed what researchers Daniel Lieberman, Osbjorn Pearson, and Kenneth Mowbray (2000) called marked or clear occipital bunning. Also, compared to later modern humans, the Mount Carmel people were more robust. Skhul V had a particularly impressive brow ridge that was short in height but sharply jutted forward above the eyes (Figure 13.9). The high level of preservation is due to the intentional burial of some of these people. Besides skeletal material, there are signs of artistic or symbolic behavior. For example, the adult male Skhul V had a boar’s jaw on his chest. Similarly, Qafzeh 11, a juvenile with healed cranial trauma, had an impressive deer antler rack placed over his torso (Figure 13.10; Coqueugniot et al. 2014). Perforated seashells colored with ochre, mineral-based pigment, were also found in Qafzeh (Bar-Yosef Mayer, Vandermeersch, and Bar-Yosef 2009).

One remaining question is, what happened to the modern humans of the Levant after 90,000 years ago? Another site attributed to our species did not appear in the region until 47,000 years ago. Competition with Neanderthals may have accounted for the disappearance of modern human occupation since the Neanderthal presence in the Levant lasted longer than the dates of the early modern Homo sapiens. John Shea and Ofer Bar-Yosef (2005) hypothesized that the Mount Carmel modern humans were an initial expansion from Africa that failed. Perhaps they could not succeed due to competition with the Neanderthals who had been there longer and had both cultural and biological adaptations to that environment.
Modern Homo sapiens of China
A long history of paleoanthropology in China has found ample evidence of modern human presence. Four notable sites are the caves at Fuyan, Liujiang, Tianyuan, and Zhoukoudian. In the distant past, these caves would have been at least seasonal shelters that unintentionally preserved evidence of human presence for modern researchers to discover.
At Fuyan Cave in Southern China, paleoanthropologists found 47 adult teeth associated with cave formations dated to between 120,000 and 80,000 years ago (Liu et al. 2015). It is currently the oldest-known modern human site in China, though other researchers question the validity of the date range (Michel et al. 2016). The teeth have the small size and gracile features of modern Homo sapiens dentition.
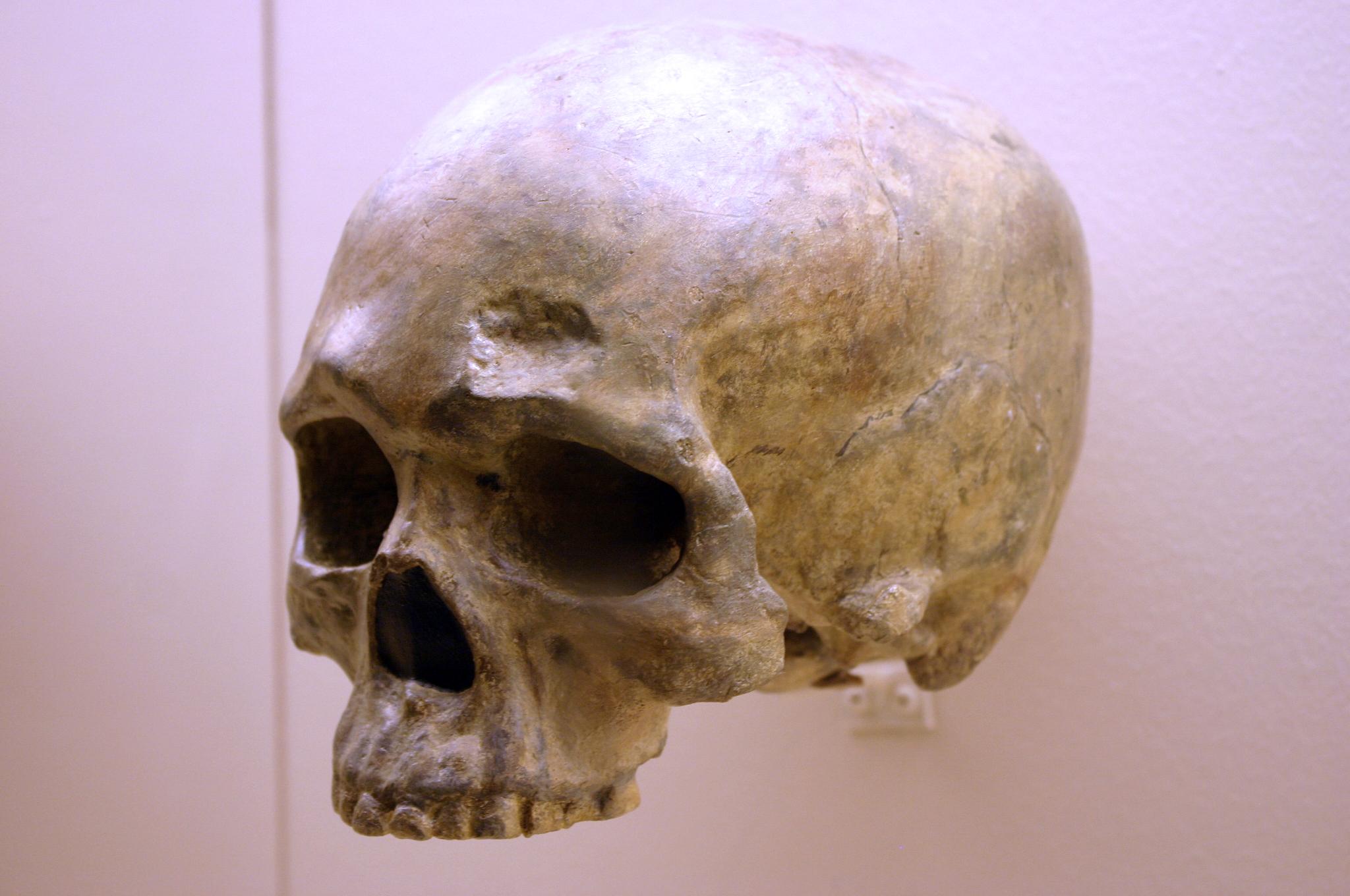
The fossil Liujiang (or Liukiang) hominin (67,000 years ago) has derived traits that classified it as a modern Homo sapiens, though primitive archaic traits were also present. In the skull, which was found nearly complete, the Liujiang hominin had a taller forehead than archaic Homo sapiens but also had an enlarged occipital region (Figure 13.11; Brown 1999; Wu et al. 2008). Other parts of the skeleton also had a mix of modern and archaic traits: for example, the femur fragments suggested a slender length but with thick bone walls (Woo 1959).
Another Chinese site to describe here is the one that has been studied the longest. In the Zhoukoudian Cave system (Figure 13.12), where Homo erectus and archaic Homo sapiens have also been found, there were three crania of modern Homo sapiens. These crania, which date to between 34,000 and 10,000 years ago, were all more globular than those of archaic humans but still lower and longer than those of later modern humans (Brown 1999; Harvati 2009). When compared to one another, the crania showed significant differences from one another. Comparison of cranial measurements to other populations past and present found no connection with modern East Asians, again showing that human variation was very different from what we see today.

Crossing to Australia
Expansion of the first modern human Asians, still following the coast, eventually entered an area that researchers call Sunda before continuing on to modern Australia. Sunda was a landmass made up of the modern-day Malay Peninsula, Sumatra, Java, and Borneo. Lowered sea levels connected these places with land bridges, making them easier to traverse. Proceeding past Sunda meant navigating Wallacea, the archipelago that includes the Indonesian islands east of Borneo. In the distant past, there were many megafauna, large animals that migrating humans would have used for food and materials (such as utilizing animals’ hides and bones). Further southeast was another landmass called Sahul, which included New Guinea and Australia as one contiguous continent. Based on fossil evidence, this land had never seen hominins or any other primates before modern Homo sapiens arrived. Sites along this path offer clues about how our species handled the new environment to live successfully as foragers.

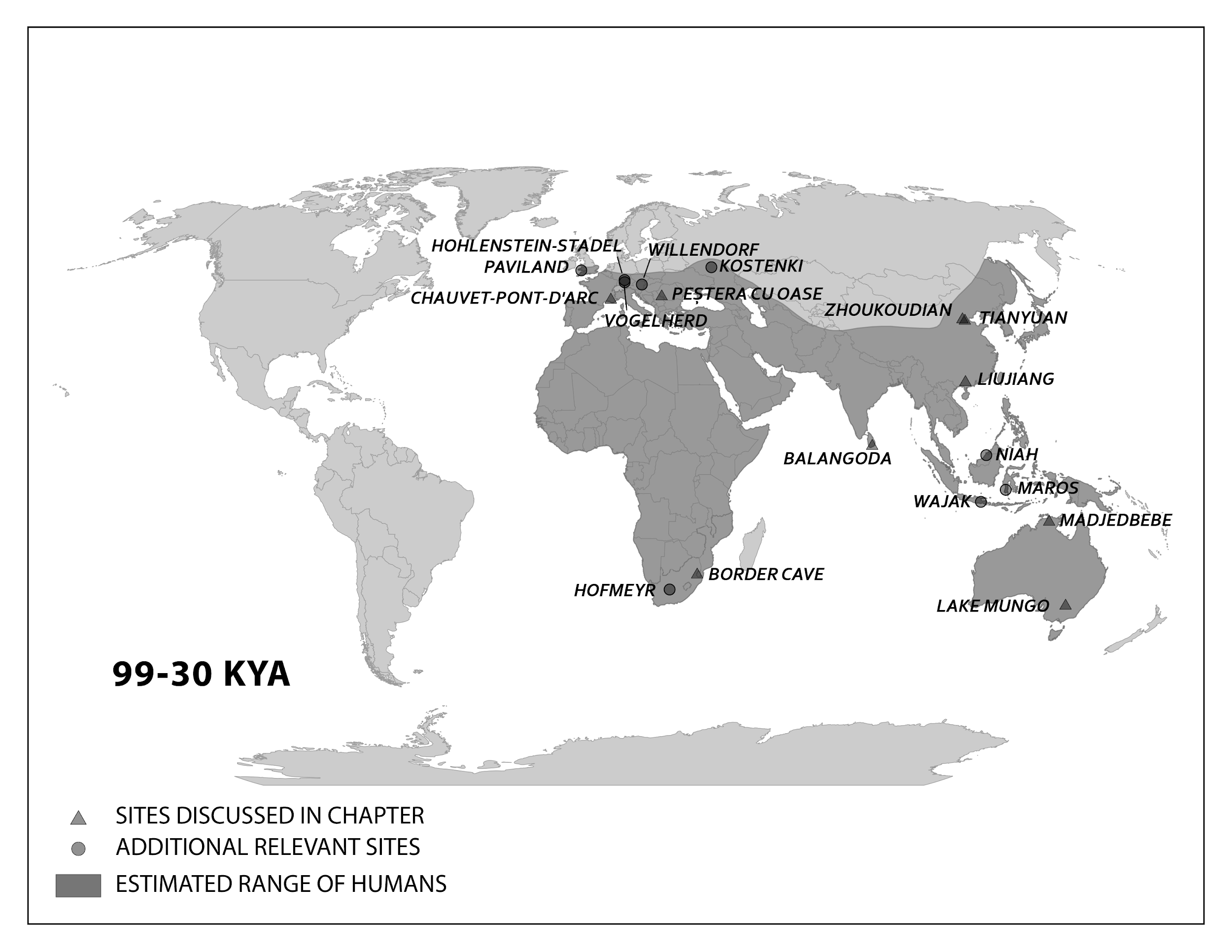
The skeletal remains at Lake Mungo, land traditionally owned by Mutthi Mutthi, Ngiampaa, and Paakantji peoples, are the oldest known in the continent. The now-dry lake was one of a series located along the southern coast of Australia in New South Wales, far from where the first people entered from the north (Barbetti and Allen 1972; Bowler et al. 1970). Two individuals dating to around 40,000 years ago show signs of artistic and symbolic behavior, including intentional burial. The bones of Lake Mungo 1 (LM1), an adult female, were crushed repeatedly, colored with red ochre, and cremated (Bowler et al. 1970). Lake Mungo 3 (LM3), a tall, older male with a gracile cranium but robust postcranial bones, had his fingers interlocked over his pelvic region (Brown 2000).
Kow Swamp, within traditional Yorta Yorta land also in southern Australia, contained human crania that looked distinctly different from the ones at Lake Mungo (Durband 2014; Thorne and Macumber 1972). The crania, dated between 9,000 and 20,000 years ago, had extremely robust brow ridges and thick bone walls, but these were paired with globular features on the braincase (Figure 13.13).
While no fossil humans have been found at the Madjedbebe rock shelter in the North Territory of Australia, more than 10,000 artifacts found there show both behavioral modernity and variability (Clarkson et al. 2017). They include a diverse array of stone tools and different shades of ochre for rock art, including mica-based reflective pigment (similar to glitter). These impressive artifacts are as far back as 56,000 years old, providing the date for the earliest-known presence of humans in Australia.
From the Levant to Europe
The first modern human expansion into Europe occurred after other members of our species settled in East Asia and Australia. As the evidence from the Levant suggests, modern human movement to Europe may have been hampered by the presence of Neanderthals. It is suggested that another obstacle was the colder climate, which was incompatible with the biology of modern Homo sapiens from Africa, as they were adapted to high temperatures and ultraviolet radiation. Still, by 40,000 years ago, modern Homo sapiens had a detectable presence. This time was also the start of the Later Stone Age or Upper Paleolithic, when there was an expansion in cultural complexity. There is a wealth of evidence from this region due to a Western bias in research, the proximity of these findings to Western scientific institutions, and the desire of Western scientists to explore their own past.
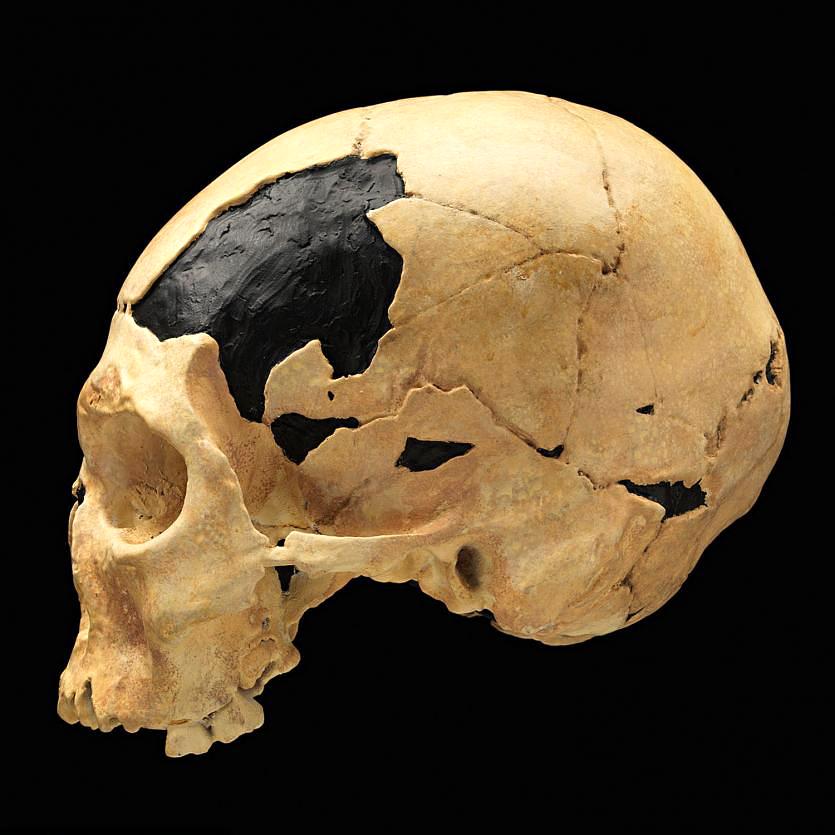
In Romania, the site of Peștera cu Oase (Cave of Bones) had the oldest-known remains of modern Homo sapiens in Europe, dated to around 40,000 years ago (Trinkaus et al. 2003a). Among the bones and teeth of many animals were the fragmented cranium of one person and the mandible of another (the two bones did not fit each other). Both bones have modern human traits similar to the fossils from the Middle East, but they also had Neanderthal traits. Oase 1, the mandible, had a mental eminence but also extremely large molars (Trinkaus et al. 2003b). This mandible has yielded DNA that surprisingly is equally similar to DNA from present-day Europeans and Asians (Fu et al. 2015). This means that Oase 1 was not the direct ancestor of modern Europeans. The Oase 2 cranium has the derived traits of reduced brow ridges along with archaic wide zygomatic cheekbones and an occipital bun (Figure 13.14; Rougier et al. 2007).
Dating to around 26,000 years ago, Předmostí near Přerov in the Czech Republic was a site where people buried over 30 individuals along with many artifacts. Eighteen individuals were found in one mass burial area, a few covered by the scapulae of woolly mammoths (Germonpré, Lázničková-Galetová, and Sablin 2012). The Předmostí crania were more globular than those of archaic humans but tended to be longer and lower than in later modern humans (Figure 13.15; Velemínská et al. 2008). The height of the face was in line with modern residents of Central Europe. There was also skeletal evidence of dog domestication, such as the presence of dog skulls with shorter snouts than in wild wolves (Germonpré, Lázničková-Galetová, and Sablin et al. 2012). In total, Předmostí could have been a settlement dependent on mammoths for subsistence and the artificial selection of early domesticated dogs.
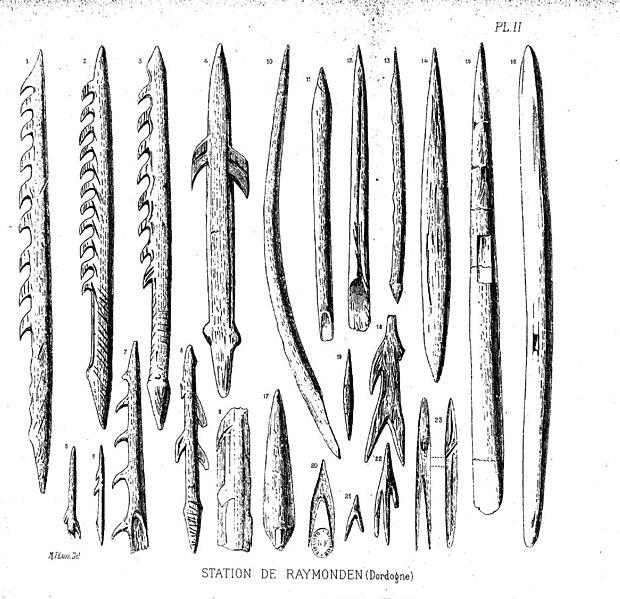
The sequence of modern Homo sapiens technological change in the Later Stone Age has been thoroughly dated and labeled by researchers working in Europe. Among them, the Gravettian tradition of 33,000 years to 21,000 years ago is associated with most of the known curvy female figurines, often assumed to be “Venus” figures. Hunting technology also advanced in this time with the first known boomerang, atlatl (spear thrower), and archery. The Magdalenian tradition spread from 17,000 to 12,000 years ago. This culture further expanded on fine bone tool work, including barbed spearheads and fishhooks (Figure 13.16).
Among the many European sites dating to the Later Stone Age, the famous cave art sites deserve mention. Chauvet-Pont-d'Arc Cave in southern France dates to separate Aurignacian occupations 31,000 years ago and 26,000 years ago. Over a hundred art pieces representing 13 animal species are preserved, from commonly depicted deer and horses to rarer rhinos and owls. Another French cave with art is Lascaux, which is several thousand years younger at 17,000 years ago in the Magdalenian period. At this site, there are over 6,000 painted figures on the walls and ceiling (Figure 13.17). Scaffolding and lighting must have been used to make the paintings on the walls and ceiling deep in the cave. Overall, visiting Lascaux as a contemporary must have been an awesome experience: trekking deeper in the cave lit only by torches giving glimpses of animals all around as mysterious sounds echoed through the galleries.

Special Topic: Cannibalism and Culture - Mortuary Practices in Modern Homo sapiens
Within a 2017 publication in the Journal of Archaeological Method and Theory, Saladié and Rodríguez-Hidalgo bring light to traces of early cannibalism in western Eurasia, arguing that context-specific cannibalistic practices were present throughout the Pleistocene and increased notably from the end of the Upper Palaeolithic and onward (Saladié & Rodriguez-Hidalgo, 2017). While early hominins and Neandertals are recognized in this research, the authors highlight the presence of these mortuary practices in a cluster of Homo sapiens sites. More recent research uncovers similar findings that back these claims as well, where human bones in Herto Ethiopia, Maszycka Cave Poland, and Gough’s Cave in the United Kingdom show anthropogenic defleshing and other modifications which have been interpreted as cannibalism (Pobiner, Et al. 2023). These findings suggest that cannibalistic behaviours formed a recurring aspect of modern human behaviour in certain ecological and cultural contexts.

A significant example comes from the Neolithic levels of Fontbrégua Cave in southeastern France, where Paola Villa and colleagues compared clusters of human bones with the remains of wild and domestic animals from the same sediments. The study found that human bodies were butchered, processed, and most likely eaten in a way that parallels animal carcass treatment, including the placement and timing of cut marks, dismemberment sequences, and perimortem fractures to open marrow cavities (Villa, Et al. 1986). As the assemblage comes from a primary depositional context with pristine preservation and careful excavation, the authors suggest that cannibalism is the only satisfactory explanation for the pattern of cut marks and breakage seen on the human bones.
More recent work has furthered this hypothesis for Late Upper Palaeolithic Homo sapiens, especially in Magdalenian contexts. In a 2023 study, researchers combined archeological and genetic evidence from fifty-nine Magdalenian sites, concluding that this specific culture shows an unusually high frequency of cannibalistic cases compared to earlier and later hominin groups; so much so that they identify “primary burial and cannibalism” as the two main mortuary expressions (Marsh & Bello, 2023). Additionally, new analyses from Maszycka Cave in Poland have described cut and broken human bones in patterns consistent with human consumption, reinforcing this cannibalism hypothesis (Marginedas & Saladié, 2025). Together, these studies suggest that for some Magdalenian groups, mortuary cannibalism was a habitual way of disposing of the dead, not just a one-off crisis response. At the same time, researchers emphasize that not every modified skeleton indicates blatant consumption of the dead and that ritual or symbolic perspectives must also be considered. Previously noted in chapter 12, Ullrich’s survey of European mortuary practices presents frequent manipulations of corpses from the Palaeolithic through the Hallstatt period. Said manipulations include cut marks, dismemberment, skull fracturing, and marrow extraction (Ullrich, 2005, p. 258), which Ullrich interprets as cannibalistic rites embedded in cult ceremonies rather than everyday subsistence. In the author’s view, some Palaeolithic groups may have believed that consuming members of their community allowed them to take on the strengths,

abilities, or even mental aspects of their dead member; the act of cannibalism may have functioned as a form of appropriating physical and mental powers rather than simply obtaining calories. With that, Neolithic assemblages show how careful taphonomic work can distinguish cuts linked to interpersonal violence from those associated with systemic butchery (Marginedas & Saladié, 2025). The findings seek to highlight that some Homo sapiens populations combined ritual, mortuary, and nutritional motives when processing human remains.
These past and contemporary findings are significant for future anthropology students as they shed light on biological anthropology methodology and interpretation. Archaeologists are able to distinguish between occasions when humans were handled like any other carcass and times when they were handled in more symbolic ways thanks to factors like cut mark orientation, the timing of bone breakage, and direct comparison with animal remains. Readers interested in exploring this topic further should investigate the context-specific motivations for cannibalism, like nutritional stress, warfare, funerary sites, or ritual power appropriation.
Similar archeological techniques have been used to explore behaviours of cannibalism among Indigenous Huron-Wendat populations in 1651 Canada, where human bones exhibit cutmarks, perimortem fractures, and thermal modifications (Spence & Jackson, 2014). These shared taphonomic signatures across continents reveal recurring practices under ecological stress, bridging prehistoric Europe to North American contexts. Engaging with such analytical techniques opens up new ways that archeologists and anthropologists can investigate the variability in human behaviour, from nutritional crises to mortuary rituals.
Peopling of the Americas
By 25,000 years ago, our species was the only member of Homo left on Earth. Gone were the Neanderthals, Denisovans, Homo naledi, and Homo floresiensis. The range of modern Homo sapiens kept expanding eastward into—using the name given to this area by Europeans much later—the Western Hemisphere. This section will address what we know about the peopling of the Americas, from the first entry to these continents to the rapid spread of Indigenous Americans across its varied environments.
While evidence points to an ancient land bridge called Beringia that allowed people to cross from what is now northeastern Siberia into modern-day Alaska, what people did to cross this land bridge is still being investigated. For most of the 20th century, the accepted theory was the Ice-Free Corridor model. It stated that northeast Asians (East Asians and Siberians) first expanded across Beringia inland through a passage between glaciers that opened into the western Great Plains of the United States, just east of the Rocky Mountains, around 13,000 years ago (Swisher et al. 2013). While life up north in the cold environment would have been harsh, migrating birds and an emerging forest might have provided sustenance as generations expanded through this land (Potter et al. 2018).
However, in recent decades, researchers have accumulated evidence against the Ice-Free Corridor model. Archaeologist K. R. Fladmark (1979) brought the alternate Coastal Route model into the archaeological spotlight; researcher Jon M. Erlandson has been at the forefront of compiling support for this theory (Erlandson et al. 2015). The new focus is the southern edge of the land bridge instead of its center: About 16,000 years ago, members of our species expanded along the coastline from northeast Asia, east through Beringia, and south down the Pacific Coast of North America while the inland was still sealed off by ice. The coast would have been free of ice at least part of the year, and many resources would have been found there, such as fish (e.g., salmon), mammals (e.g., whales, seals, and otters), and plants (e.g., seaweed).
South through the Americas
When the first modern Homo sapiens reached the Western Hemisphere, the spread through the Americas was rapid. Multiple migration waves crossed from North to South America (Posth et al. 2018). Our species took advantage of the lack of hominin competition and the bountiful resources both along the coasts and inland. The Americas had their own wide array of megafauna, which included woolly mammoths (Figure 13.20), mastodons, camels, horses, ground sloths, giant tortoises, and—a favorite of researchers—a two-meter-tall beaver. The reason we cannot see these amazing animals today may be that resources gained from these fauna were crucial to the survival for people over 12,000 years ago (Araujo et al. 2017). Several sites are notable for what they add to our understanding of the distant past in the Americas, including interactions with megafauna and other elements of the environment.

A 2019 discovery may allow researchers to improve theories about the peopling of the Americas. In White Sands National Park, New Mexico, 60 human footprints have been astonishingly dated to around 22,000 years ago (Bennett et al. 2021). This date and location do not match either the Ice-Free Corridor or Coastal Route models. Researchers are now working to verify the find and adjust previous models to account for the new evidence. This groundbreaking find is sparking new theories; it is another example of the fast pace of research performed on our past.
Monte Verde is a landmark site that shows that the human population had expanded down the whole vertical stretch of the Americas to Chile by 14,600 years ago. The site has been excavated by archaeologist Tom D. Dillehay and his team (2015). The remains of nine distinct edible species of seaweed at the site shows familiarity with coastal resources and relates to the Coastal Route model by showing a connection between the inland people and the sea.

Named after the town in New Mexico, the Clovis stone-tool style is the first example of a widespread culture across much of North America, between 13,400 and 12,700 years ago (Miller, Holliday, and Bright 2013). Clovis points were fluted with two small projections, one on each end of the base, facing away from the head (Figure 13.21). The stone points found at this site match those found as far as the Canadian border and northern Mexico, and from the west coast to the east coast of the United States. Fourteen Clovis sites also contained the remains of mammoths or mastodons, suggesting that hunting megafauna with these points was an important part of life for the Clovis people. After the spread of the Clovis style, it diversified into several regional styles, keeping some of the Clovis form but also developing their own unique touches.
The Big Picture: The Assimilation Hypothesis
How do researchers make sense of all of these modern Homo sapiens discoveries that cover over 300,000 years of time and stretch across every continent except Antarctica? How was modern Homo sapiens related to archaic Homo sapiens?
The Assimilation hypothesis proposes that modern Homo sapiens evolved in Africa first and expanded out but also interbred with the archaic Homo sapiens they encountered outside Africa (Figure 13.22). This hypothesis is powerful since it explains why Africa has the oldest modern human fossils, why early modern humans found in Europe and Asia bear a resemblance to the regional archaics, and why traces of archaic DNA can be found in our genomes today (Dannemann and Racimo 2018; Reich et al. 2010; Reich et al. 2011; Slatkin and Racimo 2016; Smith et al. 2017; Wall and Yoshihara Caldeira Brandt 2016).
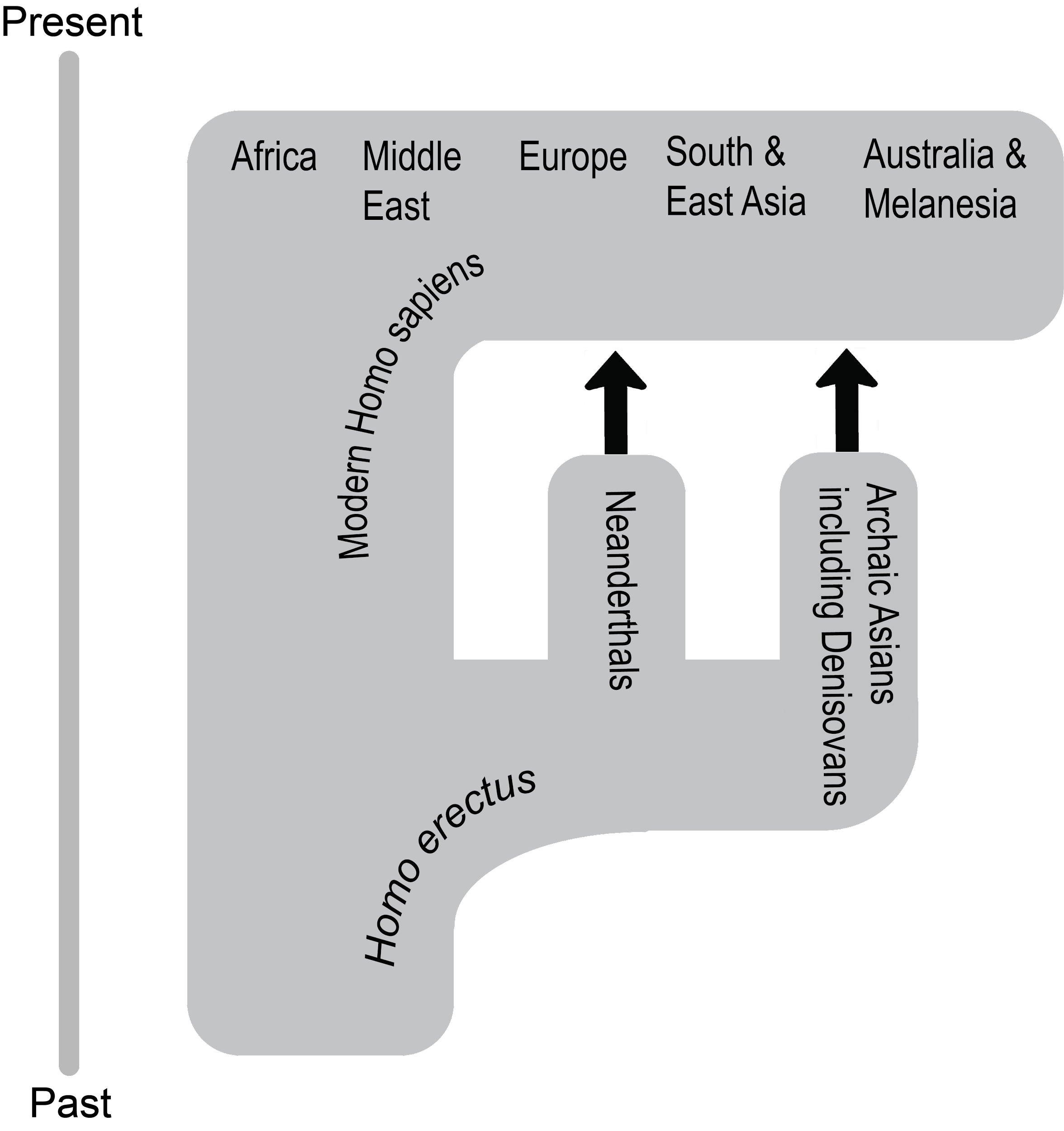
While researchers have produced a model that satisfies the data, there are still a lot of questions for paleoanthropologists to answer regarding our origins. What were the patterns of migration in each part of the world? Why did the archaic humans go extinct? In what ways did archaic and modern humans interact? The definitive explanation of how our species started and what our ancestors did is still out there to be found. You are now in a great place to welcome the next discovery about our distant past—maybe you’ll even contribute to our understanding as well.
The Chain Reaction of Agriculture
While it may be hard to imagine today, for most of our species’ existence we were nomadic: moving through the landscape without a singular home. Instead of a refrigerator or pantry stocked with food, we procured nutrition and other resources as needed based on what was available in the environment. This section gives an overview of how the foraging lifestyle enabled the expansion of our species and how the invention of a new way of life caused a chain reaction of cultural change.
The Foraging Tradition
There are a variety of possible subsistence strategies, or methods of finding sustenance and resources. To understand our species is to understand the subsistence strategy of foraging, or the search for resources in the environment. While most (but not all) humans today live in cultures that practice agriculture (whereby we greatly shape the environment to mass produce what we need), we have spent far more time as nomadic foragers than as settled agriculturalists. As such, it has been suggested that our traits have evolved to be primarily geared toward foraging. For instance, our efficient bipedalism allows persistence-hunting across long distances as well as movement from resource to resource.
How does human foraging, also known as hunting and gathering, work? Anthropologists have used all four fields to answer this question (see Ember n.d.). Typically, people formed bands, or kin-based groups of around 50 people or less (rarely over 100). A band’s organization would be egalitarian, with a flexible hierarchy based on an individual’s age, level of experience, and relationship with others. Everyone would have a general knowledge of the skills assigned to their gender roles, rather than specializing in different occupations. A band would be able to move from place to place in the environment, using knowledge of the area to forage (Figure 13.23). In varied environments—from savannas to tropical forests, deserts, coasts, and the Arctic circle—people found sustenance needed for survival.

Humans made extensive use of the foraging subsistence strategy, but this lifestyle did have limitations. The ease of foraging depended on the richness of the environment. Due to the lack of storage, resources had to be dependably found when needed. While a bountiful environment would require just a few hours of foraging a day and could lead to a focus on one location, the level and duration of labor increased greatly in poor or unreliable environments. Labor was also needed to process the acquired resources, which contributed to the foragers’ daily schedule (Crittenden and Schnorr 2017).
The adaptations to foraging found in modern Homo sapiens may explain why our species became so successful both within Africa and in the rapid expansion around the world. Overcoming the limitations, each generation at the edge of our species’s range would have found it beneficial to expand a little further, keeping contact with other bands but moving into unexplored territory where resources were more plentiful. The cumulative effect would have been the spread of modern Homo sapiens across continents and hemispheres.
Why Agriculture?
After hundreds of thousands of years of foraging, some groups of people around 12,000 years ago started to practice agriculture. This transition, called the Neolithic Revolution, occurred at the start of the Holocene epoch. While the reasons for this global change are still being investigated, two likely co-occurring causes are a growing human population and natural global climate change.
Overcrowding could have affected the success of foraging in the environment, leading to the development of a more productive subsistence strategy (Cohen 1977). Foraging works best with low population densities since each band needs a lot of space to support itself. If too many people occupy the same environment, they deplete the area faster. The high population could exceed the carrying capacity, or number of people a location can reliably support. Reaching carrying capacity on a global level due to growing population and limited areas of expansion would have been an increasingly pressing issue after the expansion through the major continents by 14,600 years ago.
A changing global climate immediately preceded the transition to agriculture, so researchers have also explored a connection between the two events. Since the Last Glacial Maximum of 23,000 years ago, the Earth slowly warmed. Then, from 13,000 to 11,700 years ago, the temperature in most of the Northern Hemisphere dropped suddenly in a phenomenon called the Younger Dryas. Glaciers returned in Europe, Asia, and North America. In Mesopotamia, which includes the Levant, the climate changed from warm and humid to cool and dry. The change would have occurred over decades, disrupting the usual nomadic patterns and subsistence of foragers around the world. The disruption to foragers due to the temperature shift could have been a factor in spurring a transition to agriculture. Researchers Gregory K. Dow and colleagues (2009) believe that foraging bands would have clustered in the new resource-rich places where people started to direct their labor to farming the limited area. After the Younger Dryas ended, people expanded out of the clusters with their agricultural knowledge (Figure 13.24).
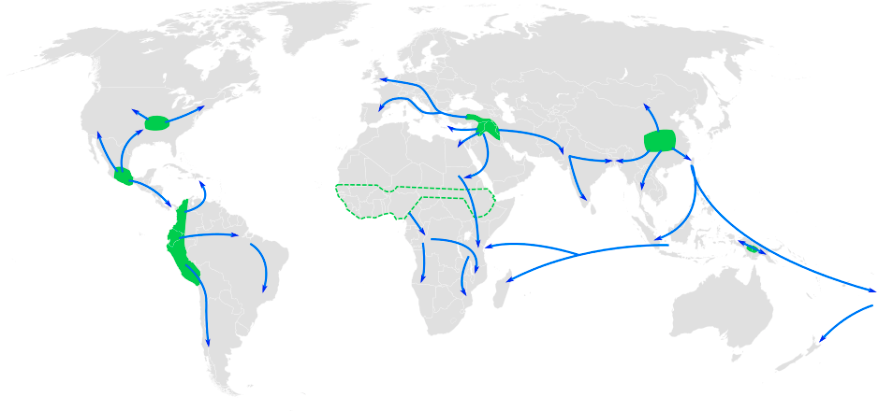
The double threat of the limitation of human continental expansion and the sudden global climate change may have placed bands in peril as more populations outpaced their environment’s carrying capacity. Not only had a growing population may have led to increased competition with other bands, but environments worldwide had shifted to create more uncertainty. As such, it has been proposed that as people in different areas around the world faced this unpredictable situation, they became the independent inventors of agriculture.
Agriculture around the World
Due to global changes to the human experience starting from 12,000 years ago, it has been suggested that cultures with no knowledge of each other turned toward intensely farming their local resources (see Figure 13.24). It is proposed that the first farmers engaged in artificial selection of their domesticates to enhance useful traits over generations. The switch to agriculture took time and effort with no guarantee of success and constant challenges (e.g. fires, droughts, diseases, and pests). The regions with the most widespread impact in the face of these obstacles became the primary centers of agriculture (Figure 13.25; Fuller 2010):
- Mesopotamia: The Fertile Crescent from the Tigris and Euphrates rivers through the Levant was where bands started to domesticate plants and animals around 12,000 years ago. The connection between the development of agriculture and the Younger Dryas was especially strong here. Farmed crops included wheat, barley, peas, and lentils. This was also where cattle, pigs, sheep, and goats were domesticated.
- South and East Asia: Multiple regions across this land had varieties of rice, millet, and soybeans by 10,000 years ago. Pigs were farmed with no connection to Mesopotamia. Chickens were also originally from this region, bred for fighting first and food second.
- New Guinea: Agriculture started here 10,000 years ago. Bananas, sugarcane, and taro were native to this island. Sweet potatoes were brought back from voyages to South America around the year C.E. 1000. No known animal farming occurred here.
- Mesoamerica: Agriculture from Central Mexico to northern South America also occurred from 10,000 years ago; it was also only plant based. Maize was a crop bred from teosinte grass, which has become one of the global staples. Beans, squash, and avocados were also grown in this region.
- The Andes: Starting around 8,000 years ago, local domesticated plants started with squash but later included potatoes, tomatoes, beans, and quinoa. Maize was brought down from Mesoamerica. The main farm animals were llamas, alpacas, and guinea pigs.
- Sub-Saharan Africa: This region went through a change 5,000 years ago called the Bantu expansion. The Bantu agriculturalists were established in West Central Africa and then expanded south and east. Native varieties of rice, yams, millet, and sorghum were grown across this area. Cattle were also domesticated here.
- Eastern North America: This region was the last major independent agriculture center, from 4,000 years ago. Squash and sunflower are the produce from this region that are most known today, though sumpweed and pitseed goosefoot were also farmed. Hunting was still the main source of animal products.

By 5,000 years ago, our species was well within the Neolithic Revolution. Agriculturalists spread to neighboring parts of the world with their domesticates, further expanding the use of this subsistence strategy. From this point, the human species changed from being primarily foragers to primarily agriculturalists with skilled control of their environments. The planet changed from mostly unaffected by human presence to being greatly transformed by humans. The revolution took millennia, but it was a true revolution as our species’ lifestyle was dramatically reshaped.
Cultural Effects of Agriculture
The worldwide adoption of agriculture altered the course of human culture and history forever. The core change in human culture due to agriculture is the move toward not moving: rather than live a nomadic lifestyle, farmers had to remain in one area to tend to their crops and livestock. The term for living bound to a certain location is sedentarism. This led to new aspects of life that were uncommon among foragers: the construction of permanent shelters and agricultural infrastructure, such as fields and irrigation, plus the development of storage technology, such as pottery, to preserve extra resources in case of future instability.

The high productivity of successful agriculture sparked further changes (Smith 2009). It is argued that since successful agriculture produced a much greater amount of food and other resources per unit of land compared to foraging, the population growth rate skyrocketed. The surplus of a bountiful harvest also provided insurance for harder times, reducing the risk of famine and bringing change to society as well. With a few farming households producing enough food to feed many others, people could start specializing in roles such as craftspeople, traders, religious figures, and artists, spurring innovation in these areas as people could now devote time and effort toward specific skills. These interdependent people would settle an area together for convenience, and over time, the growth of these settlements led to urbanization, the founding of cities that became the foci of human interaction (Figure 13.26).
The formation of cities led to new issues that sparked the growth of further specializations, called institutions. These are cultural constructs that exist beyond the individual and have wide control over a population. Leadership of these cities became hierarchical with different levels of rank and control. The stratification of society increased social inequality between those with more or less power over others. Under leadership, people built impressive monumental architecture, such as pyramids and palaces, that embodied the wealth and power of these early cities. Alliances could unite cities, forming the earliest states. In several regions of the world, state organization expanded into empires, wide-ranging political entities that covered a variety of cultures.
Urbanization brought new challenges as well. The concentration of sedentary peoples was ideal for infectious diseases to thrive since they could jump from person to person and even from livestock to person (Armelagos, Brown, and Turner 2005). While successful agriculture provided a large surplus of food to thwart famine, the food produced offered less diverse food sources than foragers’ diets (Cohen and Armelagos 1984; Cohen and Crane-Kramer 2007). This shift in nutrition caused other diseases to flourish among those who adopted farming, such as dental cavities and malocclusion (the misalignment of teeth caused by soft, agricultural diets). The need to extract “wisdom teeth” or third molars seen in agricultural cultures today stems from this misalignment between the environment our ancestors adapted to and our lifestyles today.
As the new disease trends show, the adoption of agriculture and the ensuing cultural changes were not entirely positive. It is also important to note that this is not an absolutely linear progression of human culture from simple to complex. In many cases, empires have collapsed and, in some cases, cities dispersed to low-density bands that rejected institutions. However, a global trend has emerged since the adoption of agriculture, wherein population and social inequality have increased, leading to the massive and influential nation-states of today.
The rise of states in Europe has a direct impact on many of this book’s topics. Science started as a European cultural practice by the upper class that became a standardized way to study the world. Education became an institution to provide a standardized path toward producing and gaining knowledge. The scientific study of human diversity, embroiled in the race concept that still haunts us today, was connected to the European slave trade and colonialism.
Also starting in Europe, the Industrial Revolution of the 19th century turned cities into centers of mass manufacturing and spurred the rapid development of inventions (Figure 13.27). In the technologically interconnected world of today, human society has reached a new level of complexity with globalization. In this system, goods are mass-produced and consumed in different parts of the world, weakening the reliance on local farms and factories. The imbalanced relationship between consumers and producers of goods further increases economic inequality.

As states based on agriculture and industry keep exerting influence on humanity today, there are people, like the Hadzabe of Tanzania, who continue to live a lifestyle centered on foraging. Due to the overwhelming force that agricultural societies exert, foragers today have been marginalized to live in the least habitable parts of the world—the areas that are not conducive to farming, such as tropical rainforests, deserts, and the Arctic (Headland et al. 1989). Foragers can no longer live in the abundant environments that humans would have enjoyed before the Neolithic Revolution. Interactions with agriculturalists are typically imbalanced, with trade and other exchanges heavily favoring the larger group. One of anthropology’s important roles today is to intelligently and humanely manage equitable interactions between people of different backgrounds and levels of influence.
Special Topic: Indigenous Land Management
Insight into the lives of past modern humans has evolved as researchers revise previous theories and establish new connections with Indigenous knowledge holders.
The outdated view of foraging held that people lived off of the land without leaving an impact on the environment. Accompanying this idea was anthropologist Marshall Sahlins’s (1968) proposal that foragers were the “original affluent society” since they were meeting basic needs and achieving satisfaction with less work hours than agriculturalists and city-dwellers. This view countered an earlier idea that foragers were always on the brink of starvation. Sahlins’s theory took hold in the public eye as an attractive counterpoint to our busy contemporary lives in which we strive to meet our endless wants.
A fruitful type of study involving researchers collaborating with Indigenous experts has found that foragers did not just live off the land with minimal effort nor were they barely surviving in unchanging environments. Instead, they shaped the landscape to their needs using labor and strategies that were more subtle than what European colonizers and subsequent researchers were used to seeing. Research from two regions shows the latest developments in understanding Indigenous land management.
In British Columbia, Canada, the bridging of scientific and Indigenous perspectives has shown that the forests of the region are not untouched wilderness but, rather, have been crafted by Indigenous peoples thousands of years ago. Forest gardens adjacent to archaeological sites show higher plant diversity than unmanaged places even after 150 years (Armstrong et al. 2021). On the coast, 3,500-year-old archaeological sites are evidence of constructed clam gardens, according to Indigenous experts (Lepofsky et al. 2015). Another project, in consultation with Elders of the T’exelc (William Lakes First Nation) in British Columbia, introduced researchers to explanations of how forests were managed before the practice was disrupted by European colonialism (Copes-Gerbitz et al. 2021). Careful management of controlled fires reduced the density of the forest to favor plants such as raspberries and allow easier movement through the landscape.
Similarly, the study of landscapes in Australia, in consultation with Aboriginal Australians today, shows that areas previously considered wilderness by scientists were actually the result of controlling fauna and fires. The presence of grasslands with adjacent forests were purposely constructed to attract kangaroos for hunting (Gammage 2008). People also managed other animal and insect life, from emus to caterpillars. In Tasmania, a shift from productive grassland to wildfire-prone rainforest occurred after Aboriginal Australian land management was replaced by British colonial rule (Fletcher, Hall, and Alexander 2021). The site of Budj Bim of the Gunditjmara people has archaeological features of aquaculture, or the farming of fish, that date back 6,600 years (McNiven et al. 2012; McNiven et al. 2015). These examples show that Indigenous knowledge of how to manipulate the environment may be invaluable at the state level, such as by creating an Aboriginal ranger program to guide modern land management.
The Future of Humanity
A common question stemming from understanding human evolution is: What will the genetic and biological traits of our species be hundreds of thousands of years in the future? When faced with this question, people tend to think of directional selection. Maybe our braincases will be even larger, resembling the large-headed and small-bodied aliens of science fiction (Figure 13.28). Or, our hands could be specialized for interacting with our touch-based technology with less risk of repetitive injury. These ideas do not stand up to scrutiny. Since natural selection is based on adaptations that increase reproductive success, any directional change must be due to a higher rate of producing successful offspring compared to other alleles. Larger brains and more agile fingers would be convenient to possess, but they do not translate into an increase in the underlying allele frequencies.
Scientists are hesitant to professionally speculate on the unknowable, and we will never know what is in store for our species one thousand or one million years from now, but there are two trends in human evolution that may carry on into the future: increased genetic variation and a reduction in regional differences.
Rather than a directional change, genetic variation in our species could expand. Our technology can protect us from extreme environments and pathogens, even if our biological traits are not tuned to handle these stressors. The rapid pace of technological advancement means that biological adaptations will become less and less relevant to reproductive success, so nonbeneficial genetic traits will be more likely to remain in the gene pool. Biological anthropologist Jay T. Stock (2008) views environmental stress as needing to defeat two layers of protection before affecting our genetics. The first layer is our cultural adaptations. Our technology and knowledge can reduce pressure on one’s genotype to be “just right” to pass to the next generation. The second defense is our flexible physiology, such as our acclimatory responses. Only stressors not handled by these powerful responses would then cause natural selection on our alleles. These shields are already substantial, and cultural adaptations will only keep increasing in strength.
The increasing ability to travel far from one’s home region means that there will be a mixing of genetic variation on a global level in the future of our species. In recent centuries, gene flow of people around the world has increased, creating admixture in populations that had been separated for tens of thousands of years. For skin color, this means that populations all around the world could exhibit the whole range of skin colors, rather than the current pattern of decreasing melanin pigment farther from the equator. The same trend of intermixing would apply to all other traits, such as blood types. While our genetics will become more varied, the variation will be more intermixed instead of regionally isolated.
Our distant descendants will not likely be dextrous ultraintellectuals; more likely, they will be a highly variable and mobile species supported by novel cultural adaptations that make up for any inherited biological limitations. Technology may even enable the editing of DNA directly, changing these trends. With the uncertainty of our future, these are just the best-educated guesses for now. Our future is open and will be shaped little by little by the environment, our actions, and the actions of our descendants.
Hominin Species Summary
|
Hominin |
Modern Homo sapiens |
|
Dates |
315,000 years ago to present |
|
Region(s) |
Starting in Africa, then expanding around the world |
|
Famous discoveries |
Cro-Magnon individuals, discovered 1868 in Dordogne, France. Otzi the Ice Man, discovered 1991 in the Alps between Austria and Italy. Kennewick man, discovered 1996 in Washington state. |
|
Brain size |
1400 cc average |
|
Dentition |
Extremely small with short cusps. |
|
Cranial features |
An extremely globular brain case and gracile features throughout the cranium. The mandibular symphysis forms a chin at the anterior-most point. |
|
Postcranial features |
Gracile skeleton adapted for efficient bipedal locomotion at the expense of the muscular strength of most other large primates. |
|
Culture |
Extremely extensive and varied culture with many spoken and written languages. Art is ubiquitous. Technology is broad in complexity and impact on the environment. |
|
Other |
The only living hominin. Chimpanzees and bonobos are the closest living relatives. |
Summary
Modern Homo sapiens is the species that took the hominin lifestyle the furthest to become the only living member of that lineage. The largest factor that allowed us to persist while other hominins went extinct was likely our advanced ability to culturally adapt to a wide variety of environments. Our species, with its skeletal and behavioral traits, was well-suited to be generalist-specialists who successfully foraged across most of the world’s environments. The biological basis of this adaptation was our reorganized brain that facilitated innovation in cultural adaptations and intelligence for leveraging our social ties and finding ways to acquire resources from the environment. As the brain’s ability increased, it shaped the skull by reducing the evolutionary pressure to have large teeth and robust cranial bones to produce the modern Homo sapiens face.
Our ability to be generalist-specialists is seen in the geographical range that modern Homo sapiens covered in 300,000 years. In Africa, our species formed from multiregional gene flow that loosely connected archaic humans across the continent. People then expanded out to the rest of the continental Eurasia and even further to the Americas.
For most of our species’s existence, foraging was the general subsistence strategy within which people specialized to culturally adapt to their local environment. With omnivorousness and mobility, people found ways to extract and process resources, shaping the environment in return. When resource uncertainty hit the species, people around the world focused on agriculture to have a firmer control of sustenance. The new strategy shifted human history toward exponential growth and innovation, leading to our high dependence on cultural adaptations today.
While a cohesive image of our species has formed in recent years, there is still much to learn about our past. The work of many driven researchers shows that there are amazing new discoveries made all the time that refine our knowledge of human evolution. Technological innovations such as DNA analysis enable scientists to approach lingering questions from new angles. The answers we get allow us to ask even more insightful questions that will lead us to the next revelation. Like the pink limestone strata at Jebel Irhoud, previous effort has taken us so far and you are now ready to see what the next layer of discovery holds.
Review Questions
- What are the skeletal and behavioral traits that define modern Homo sapiens? What are the evolutionary explanations for its presence?
- What are some creative ways that researchers have learned about the past by studying fossils and artifacts?
- How do the discoveries mentioned in “First Africa, Then the World” fit the Assimilation model?
- What is foraging? What adaptations do we have for this subsistence strategy? Could you train to be a skilled forager?
- What are aspects of your life that come from dependence on agriculture and its cultural effects? Where did the ingredients of your favorite foods originate from?
Key Terms
African multiregionalism: The idea that modern Homo sapiens evolved as a complex web of small regional populations with sporadic gene flow among them.
Agriculture: The mass production of resources through farming and domestication.
Aquaculture: The farming of fish using techniques such as trapping, channels, and artificial ponds.
Assimilation hypothesis: Current theory of modern human origins stating that the species evolved first in Africa and interbred with archaic humans of Europe and Asia.
Atlatl: A handheld spear thrower that increased the force of thrown projectiles.
Band: A small group of people living together as foragers.
Beringia: Ancient landmass that connected Siberia and Alaska. The ancestors of Indigenous Americans would have crossed this area to reach the Americas.
Carrying capacity: The amount of organisms that an environment can reliably support.
Coastal Route model: Theory that the first Paleoindians crossed to the Americas by following the southern coast of Beringia.
Early Modern Homo sapiens, Early Anatomically Modern Human: Terms used to refer to transitional fossils between archaic and modern Homo sapiens that have a mosaic of traits. Humans like ourselves, who mostly lack archaic traits, are referred to as Late Modern Homo sapiens and simply Anatomically Modern Humans.
Egalitarian: Human organization without strict ranks. Foraging societies tend to be more egalitarian than those based on other subsistence strategies.
Foraging: Lifestyle consisting of frequent movement through the landscape and acquiring resources with minimal storage capacity.
Generalist-specialist niche: The ability to survive in a variety of environments by developing local expertise. Evolution toward this niche may have been what allowed modern Homo sapiens to expand past the geographical range of other human species.
Globalization: A recent increase in the interconnectedness and interdependence of people that is facilitated with long-distance networks.
Globular: Having a rounded appearance. Increased globularity of the braincase is a trait of modern Homo sapiens.
Gracile: Having a smooth and slender quality; the opposite of robust.
Holocene: The epoch of the Cenozoic Era starting around 12,000 years ago and lasting arguably through the present.
Ice-Free Corridor model: Theory that the first Native Americans crossed to the Americas through a passage between glaciers.
Institutions: Long-lasting and influential cultural constructs. Examples include government, organized religion, academia, and the economy.
Last Glacial Maximum: The time 23,000 years ago when the most recent ice age was the most intense.
Later Stone Age: Time period following the Middle Stone Age with a diversification in tool types, starting around 50,000 years ago.
Levant: The eastern coast of the Mediterranean. The site of early modern human expansion from Africa and later one of the centers of agriculture.
Megafauna: Large ancient animals that may have been hunted to extinction by people around the world.
Mental eminence: The chin on the mandible of modern H. sapiens. One of the defining traits of our species.
Microlith: Small stone tool found in the Later Stone Age; also called a bladelet.
Middle Stone Age: Time period known for Mousterian lithics that connects African archaic to modern Homo sapiens.
Monumental architecture: Large and labor-intensive constructions that signify the power of the elite in a sedentary society. A common type is the pyramid, a raised crafted structure topped with a point or platform.
Mosaic: Composed from a mix or composite of traits.
Neolithic Revolution: Time of rapid change to human cultures due to the invention of agriculture, starting around 12,000 years ago.
Ochre: Iron-based mineral pigment that can be a variety of yellows, reds, and browns. Used by modern human cultures worldwide since at least 80,000 years ago.
Sahul: Ancient landmass connecting New Guinea and Australia.
Sedentarism: Lifestyle based on having a stable home area; the opposite of nomadism.
Southern Dispersal model: Theory that modern H. sapiens expanded from East Africa by crossing the Red Sea and following the coast east across Asia.
Subsistence strategy: The method an organism uses to find nourishment and other resources.
Sunda: Ancient Asian landmass that incorporated modern Southeast Asia.
Supraorbital torus: The bony brow ridge across the top of the eye orbits on many hominin crania.
Upper Paleolithic: Time period considered synonymous with the Later Stone Age.
Urbanization: The increase of population density as people settled together in cities.
Wallacea: Archipelago southeast of Sunda with different biodiversity than Asia.
Younger Dryas: The rapid change in global climate—notably a cooling of the Northern Hemisphere—13,000 years ago.
For Further Exploration
Websites
First-person virtual tour of Lascaux cave with annotated cave art: Ministère de la Culture and Musée d’Archéologie Nationale. “Visit the cave” Lascaux website.
Online anthropology magazine articles related to paleoanthropology and human evolution: SAPIENS. “Evolution.” SAPIENS website.
Various presentations of information about hominin evolution: Smithsonian Institution. “What does it mean to be human?” Smithsonian National Museum of Natural History website.
Magazine-style articles on archaeology and paleoanthropology: ThoughtCo. “Archaeology.” ThoughtCo. Website.
Database of comparisons across hominins and primates: University of California, San Diego. “MOCA Domains.” Center for Academic Research & Training in Anthropogeny website.
Books
Engaging book that covers human-made changes to the environment with industrialization and globalization: Kolbert, Elizabeth. 2014. The Sixth Extinction: An Unnatural History. New York: Bloomsbury.
Overview of what human life was like among the environmental shifts of the Ice Age: Woodward, Jamie. 2014. The Ice Age: A Very Short Introduction. Oxford: OUP Press.
Articles
Recent review paper about the current state of paleoanthropology research: Stringer, C. 2016. “The Origin and Evolution of Homo sapiens.” Philosophical Transactions of the Royal Society B 371 (1698).
Overview of the history of American paleoanthropology and the many debates that have occurred over the years: Trinkaus, E. 2018. “One Hundred Years of Paleoanthropology: An American Perspective.” American Journal of Physical Anthropology 165 (4): 638–651.
Amazing magazine article that synthesizes hominin evolution and why it is important to study this subject: Wheelwright, Jeff. 2015. “Days of Dysevolution.” Discover 36 (4): 33–39.
Fascinating research on Ötzi, a mummy from 5,000 years ago: Wierer, Ursula, Simona Arrighi, Stefano Bertola, Günther Kaufmann, Benno Baumgarten, Annaluisa Pedrotti, Patrizia Pernter, and Jacques Pelegrin. 2018. “The Iceman’s Lithic Toolkit: Raw Material, Technology, Typology and Use.” PLOS One 13 (6): e0198292. https://doi.org/10.1371/journal.pone.0198292.
Documentaries
PBS NOVA series covering the expansion of modern Homo sapiens and interbreeding with archaic humans: Brown, Nicholas, dir. 2015. First Peoples. Edmonton: Wall to Wall Television. Amazon Prime Video.
PBS NOVA special featuring the footprints found in White Sands National Park: Falk, Bella, dir. 2016. Ice Age Footprints. Boston: Windfall Films. https://www.pbs.org/wgbh/nova/video/ice-age-footprints/.
PBS NOVA special about how modern humans evolved adaptations to different environments. Shows how present-day people live around the world: Thompson, Niobe, dir. 2016. Great Human Odyssey. Edmonton: Clearwater Documentary. https://www.pbs.org/wgbh/nova/evolution/great-human-odyssey.html.
References
Araujo, Bernardo B. A., Luiz Gustavo R. Oliveira-Santos, Matheus S. Lima-Ribeiro, José Alexandre F. Diniz-Filho, and Fernando A. S. Fernandez. 2017. “Bigger Kill Than Chill: The Uneven Roles of Humans and Climate on Late Quaternary Megafaunal Extinctions.” Quaternary International 431: 216–222.
Armelagos, George J., Peter J. Brown, and Bethany Turner. 2005. “Evolutionary, Historical, and Political Economic Perspectives on Health and Disease.” Social Science & Medicine 61 (4): 755–765.
Armstrong, C. G., J. E. D. Miller, A. C. McAlvay, P. M. Ritchie, and D. Lepofsky. 2021. “Historical Indigenous Land-Use Explains Plant Functional Trait Diversity. Ecology and Society 26 (2): 6.
Bar-Yosef Mayer, Daniella E., Bernard Vandermeersch, and Ofer Bar-Yosef. 2009. “Shells and Ochre in Middle Paleolithic Qafzeh Cave, Israel: Indications for Modern Behavior.” Journal of Human Evolution 56 (3): 307–314.
Barbetti, M., and H. Allen. 1972. “Prehistoric Man at Lake Mungo, Australia, by 32,000 Years Bp.” Nature 240 (5375): 46–48.
Bennett, M. R., D. Bustos, J. S. Pigati, K. B. Springer, T. M. Urban, V. T. Holliday, Sally C. Reynolds, et al. (2021). “Evidence of Humans in North America during the Last Glacial Maximum.” Science 373 (6562): 1528–1531.
Bowler, J. M., Rhys Jones, Harry Allen, and A. G. Thorne. 1970. “Pleistocene Human Remains from Australia: A Living Site and Human Cremation from Lake Mungo, Western New South Wales.” World Archaeology 2 (1): 39–60.
Brown, Peter. 1999. “The First Modern East Asians? Another Look at Upper Cave 101, Liujiang and Minatogawa 1.” In Interdisciplinary Perspectives on the Origins of the Japanese, edited by K. Omoto, 105–131. Kyoto: International Research Center for Japanese Studies.
Brown, Peter. 2000. “Australian Pleistocene Variation and the Sex of Lake Mungo 3.” Journal of Human Evolution 38 (5): 743–749.
Clarkson, Chris, Zenobia Jacobs, Ben Marwick, Richard Fullagar, Lynley Wallis, Mike Smith, Richard G. Roberts, et al. 2017. “Human Occupation of Northern Australia by 65,000 Years Ago.” Nature 547 (7663): 306–310.
Cohen, Mark Nathan. 1977. The Food Crisis in Prehistory: Overpopulation and the Origins of Agriculture. New Haven, CT: Yale University Press.
Cohen, Mark Nathan, and George J. Armelagos, eds. 1984. Paleopathology at the Origins of Agriculture. Orlando, FL: Academic Press.
Cohen, Mark Nathan, and Gillian M. M. Crane-Kramer, eds. 2007. Ancient Health: Skeletal Indicators of Agricultural and Economic Intensification. Gainesville, FL: University Press of Florida.
Copes-Gerbitz, K., S. Hagerman, and L. Daniels. 2021. “Situating Indigenous Knowledge for Resilience in Fire-Dependent Social-Ecological Systems.” Ecology and Society 26(4): 25. https://www.ecologyandsociety.org/vol26/iss4/art25/.
Coqueugniot, Hélène, Olivier Dutour, Baruch Arensburg, Henri Duday, Bernard Vandermeersch, and Anne-Marie Tillier. 2014. “Earliest Cranio-Encephalic Trauma from the Levantine Middle Palaeolithic: 3-D Reappraisal of the Qafzeh 11 Skull, Consequences of Pediatric Brain Damage on Individual Life Condition and Social Care.” PLOS ONE 9 (7): e102822.
Crittenden, Alyssa N., and Stephanie L. Schnorr. 2017. “Current Views on Hunter‐Gatherer Nutrition and the Evolution of the Human Diet.” American Journal of Physical Anthropology 162 (S63): 84–109.
d’Errico, Francesco, Lucinda Backwell, Paola Villa, Ilaria Degano, Jeannette J. Lucejko, Marion K. Bamford, Thomas F. G. Higham, Maria Perla Colombini, and Peter B. Beaumont. 2012. “Early Evidence of San Material Culture Represented by Organic Artifacts from Border Cave, South Africa.” Proceedings of the National Academy of Sciences 109 (33): 13214–13219.
d’Errico, Francesco, Christopher Henshilwood, Marian Vanhaeren, and Karen Van Niekerk. 2005. “Nassarius Kraussianus Shell Beads from Blombos Cave: Evidence for Symbolic Behaviour in the Middle Stone Age.” Journal of Human Evolution 48 (1): 3–24.
Dannemann, Michael, and Fernando Racimo. 2018. “Something Old, Something Borrowed: Admixture and Adaptation in Human Evolution.” Current Opinion in Genetics & Development 53: 1–8.
Day, M. H. 1969. “Omo Human Skeletal Remains.” Nature 222: 1135–1138.
Dillehay, Tom D., Carlos Ocampo, José Saavedra, Andre Oliveira Sawakuchi, Rodrigo M. Vega, Mario Pino, Michael B. Collins, et al. 2015. “New Archaeological Evidence for an Early Human Presence at Monte Verde, Chile.” PLOS ONE 10 (11): e0141923. doi:10.1371/journal.pone.0141923.
Dow, Gregory K., Clyde G. Reed, and Nancy Olewiler. 2009. “Climate Reversals and the Transition to Agriculture.” Journal of Economic Growth 14 (1): 27–53.
Durband, Arthur C. 2014. “Brief Communication: Artificial Cranial Modification in Kow Swamp and Cohuna.” American Journal of Physical Anthropology 155 (1): 173–178.
Ember, Carol R. N.d. “Hunter-Gatherers.” Explaining Human Culture. Human Relations Area Files. Accessed March 4, 2023. https://hraf.yale.edu/ehc/summaries/hunter-gatherers.
Erlandson, Jon M., Todd J. Braje, Kristina M. Gill, and Michael H. Graham. 2015. “Ecology of the Kelp Highway: Did Marine Resources Facilitate Human Dispersal from Northeast Asia to the Americas?” The Journal of Island and Coastal Archaeology 10 (3): 392–411.
Fladmark, K. R. 1979. “Routes: Alternate Migration Corridors for Early Man in North America.” American Antiquity 44 (1): 55–69.
Fletcher, M. S., T. Hall, and A. N. Alexandra. 2021. “The Loss of an Indigenous Constructed Landscape Following British Invasion of Australia: An Insight into the Deep Human Imprint on the Australian Landscape.” Ambio 50(1): 138–149.
Fu, Qiaomei, Mateja Hajdinjak, Oana Teodora Moldovan, Silviu Constantin, Swapan Mallick, Pontus Skoglund, Nick Patterson, et al. 2015. “An Early Modern Human from Romania with a Recent Neanderthal Ancestor.” Nature 524 (7564): 216–219.
Fuller, Dorian Q. 2010. “An Emerging Paradigm Shift in the Origins of Agriculture.” General Anthropology 17 (2): 1, 8–11.
Gammage, B. 2008. “Plain Facts: Tasmania under Aboriginal Management.” Landscape Research 33 (2): 241–254.
Germonpré, Mietje, Martina Lázničková-Galetová, and Mikhail V. Sablin. 2012. “Palaeolithic Dog Skulls at the Gravettian Předmostí Site, the Czech Republic.” Journal of Archaeological Science 39 (1): 184–202.
Gröning, Flora, Jia Liu, Michael J. Fagan, and Paul O’Higgins. 2011. “Why Do Humans Have Chins? Testing the Mechanical Significance of Modern Human Symphyseal Morphology with Finite Element Analysis.” American Journal of Physical Anthropology 144 (4): 593–606.
Harvati, Katerina. 2009. “Into Eurasia: A Geometric Morphometric Reassessment of the Upper Cave (Zhoukoudian) Specimens.” Journal of Human Evolution 57 (6): 751–762.
Headland, Thomas N., Lawrence A. Reid, M. G. Bicchieri, Charles A. Bishop, Robert Blust, Nicholas E. Flanders, Peter M. Gardner, Karl L. Hutterer, Arkadiusz Marciniak, and Robert F. Schroeder. 1989. “Hunter-Gatherers and Their Neighbors from Prehistory to the Present.” Current Anthropology 30 (1): 43–66.
Henshilwood, Christopher S., Francesco d’Errico, Karen L. van Niekerk, Yvan Coquinot, Zenobia Jacobs, Stein-Erik Lauritzen, Michel Menu, and Renata García-Moreno. 2011. “A 100,000-Year-Old Ochre-Processing Workshop at Blombos Cave, South Africa.” Science 334 (6053): 219–222.
Hershkovitz, Israel, Gerhard W. Weber, Rolf Quam, Mathieu Duval, Rainer Grün, Leslie Kinsley, Avner Ayalon, et al. 2018. “The Earliest Modern Humans Outside Africa.” Science 359 (6374): 456–459.
Hublin, Jean-Jacques, Abdelouahed Ben-Ncer, Shara E. Bailey, Sarah E. Freidline, Simon Neubauer, Matthew M. Skinner, Inga Bergmann, et al. 2017. “New Fossils from Jebel Irhoud, Morocco, and the Pan-African Origin of Homo sapiens.” Nature 546 (7657): 289–292.
Lepofsky, D., N. F. Smith, N. Cardinal, J. Harper, M. Morris, M., Gitla (Elroy White), Randy Bouchard, et al. 2015. “Ancient Shellfish Mariculture on the Northwest Coast of North America.” American Antiquity 80 (2): 236–259.
Lieberman, Daniel E. 2015. “Human Locomotion and Heat Loss: An Evolutionary Perspective.” Comprehensive Physiology 5 (1): 99–117.
Lieberman, Daniel E., Brandeis M. McBratney, and Gail Krovitz. 2002. “The Evolution and Development of Cranial Form in Homo sapiens.” Proceedings of the National Academy of Sciences 99 (3): 1134–1139.
Lieberman, Daniel E., Osbjorn M. Pearson, and Kenneth M. Mowbray. 2000. “Basicranial Influence on Overall Cranial Shape.” Journal of Human Evolution 38 (2): 291–315.
Liu, Wu, María Martinón-Torres, Yan-jun Cai, Song Xing, Hao-wen Tong, Shu-wen Pei, Mark Jan Sier, Xiao-hong Wu, R. Lawrence Edwards, and Hai Cheng. 2015. “The Earliest Unequivocally Modern Humans in Southern China.” Nature 526 (7575): 696-699.
Lucas, Peter W. 2007. “The Evolution of the Hominin Diet from a Dental Functional Perspective.” In Evolution of the Human Diet: The Known, the Unknown, and the Unknowable, edited by Peter S. Ungar, 31–38 Oxford, UK: Oxford University Press.
Marsh, W. A. & Bello, S. (2023). Cannibalism and burial in the late Upper Palaeolithic: Combining archaeological and genetic evidence. Quaternary Science Reviews, 319, 108309. https://doi.org/10.1016/j.quascirev.2023.108309
McCarthy, Robert C., and Lynn Lucas. 2014. “A Morphometric Reassessment of Bou-Vp-16/1 from Herto, Ethiopia.” Journal of Human Evolution 74: 114–117.
McDougall, Ian, Francis H. Brown, and John G. Fleagle. 2005. “Stratigraphic Placement and Age of Modern Humans from Kibish, Ethiopia.” Nature 433 (7027): 733–736.
McNiven, I. J., J. Crouch, T. Richards, N. Dolby, and G. Jacobsen. 2012. “Dating Aboriginal Stone-Walled Fishtraps at Lake Condah, Southeast Australia.” Journal of Archaeological Science 39 (2): 268–286.
McNiven, I., J. Crouch, T. Richards, K. Sniderman, N. Dolby, and G. Mirring. 2015. “Phased Redevelopment of an Ancient Gunditjmara Fish Trap over the Past 800 Years: Muldoons Trap Complex, Lake Condah, Southwestern Victoria.” Australian Archaeology 81 (1): 44–58.
Michel, Véronique, Hélène Valladas, Guanjun Shen, Wei Wang, Jian-xin Zhao, Chuan-Chou Shen, Patricia Valensi, and Christopher J. Bae. 2016. “The Earliest Modern Homo sapiens in China?” Journal of Human Evolution 101: 101–104.
Miller, D. Shane, Vance T. Holliday, and Jordon Bright. 2013. “Clovis across the Continent.” In Paleoamerican Odyssey, edited by Kelly E. Graf, Caroline V. Ketron, and Michael R. Waters, 207–220. College Station: Texas A&M University Press.
Neubauer, Simon, Jean-Jacques Hublin, and Philipp Gunz. 2018. “The Evolution of Modern Human Brain Shape.” Science Advances 4 (1): eaao5961. https://doi.org/10.1126/sciadv.aao5961.
Pearson, Osbjorn M. 2000. “Postcranial Remains and the Origin of Modern Humans.” Evolutionary Anthropology 9: 229–247.
Pearson, Osbjorn M. 2008. “Statistical and Biological Definitions of ‘Anatomically Modern’ Humans: Suggestions for a Unified Approach to Modern Morphology.” Evolutionary Anthropology: Issues, News, and Reviews 17 (1): 38–48.
Pobiner, B., Pante, M. & Keevil, T. (2023). Early Pleistocene cut marked hominin fossil from Koobi Fora, Kenya. Sci Rep, 13, 9896. https://doi.org/10.1038/s41598-023-35702-7
Pietschnig, Jakob, Lars Penke, Jelte M. Wicherts, Michael Zeiler, and Martin Voracek. 2015. “Meta-Analysis of Associations between Human Brain Volume and Intelligence Differences: How Strong Are They and What Do They Mean?” Neuroscience & Biobehavioral Reviews 57: 411–432.
Posth, Cosimo, Nathan Nakatsuka, Iosif Lazaridis, Pontus Skoglund, Swapan Mallick, Thiseas C. Lamnidis, Nadin Rohland, et al. 2018. “Reconstructing the Deep Population History of Central and South America.” Cell 175 (5): 1185–1197.
Potter, Ben A., James F. Baichtal, Alwynne B. Beaudoin, Lars Fehren-Schmitz, C. Vance Haynes, Vance T. Holliday, Charles E. Holmes, et al. 2018. “Current Evidence Allows Multiple Models for the Peopling of the Americas.” Science Advances 4 (8): eaat5473. https://doi.org/10.1126/sciadv.aat5473.
Reich, David, Richard E. Green, Martin Kircher, Johannes Krause, Nick Patterson, Eric Y. Durand, Bence Viola, et al. 2010. “Genetic History of an Archaic Hominin Group from Denisova Cave in Siberia.” Nature 468 (7327): 1053–1060.
Reich, David, Nick Patterson, Martin Kircher, Frederick Delfin, Madhusudan R. Nandineni, Irina Pugach, Albert Min-Shan Ko, et al. 2011. “Denisova Admixture and the First Modern Human Dispersals into Southeast Asia and Oceania.” American Journal of Human Genetics 89 (4): 516–528.
Richter, Daniel, Rainer Grün, Renaud Joannes-Boyau, Teresa E. Steele, Fethi Amani, Mathieu Rué, Paul Fernandes, et al. 2017. “The Age of the Hominin Fossils from Jebel Irhoud, Morocco, and the Origins of the Middle Stone Age.” Nature 546 (7657): 293–296.
Roberts, Patrick, and Brian A. Stewart. 2018. “Defining the ‘Generalist-Specialist’ Niche for Pleistocene Homo sapiens.” Nature Human Behaviour 2: 542–550.
Rougier, Helene, Ştefan Milota, Ricardo Rodrigo, Mircea Gherase, Laurenţiu Sarcinǎ, Oana Moldovan, João Zilhão, et al. 2007. “Peştera Cu Oase 2 and the Cranial Morphology of Early Modern Europeans.” Proceedings of the National Academy of Sciences 104 (4): 1165–1170.
Sahlins, Marshall. 1968. “Notes on the Original Affluent Society.” In Man the Hunter, edited by R. B. Lee and I. DeVore, 85–89. New York: Aldine Publishing Company.
Saladié, P. & Rodríguez-Hidalgo, A. (2017). Archaeological Evidence for Cannibalism in Prehistoric Western Europe: from Homo antecessor to the Bronze Age. Journal of Archaeological Method and Theory 24, 1034–1071. https://doi.org/10.1007/s10816-016-9306-y
Sawyer, G. J., and Blaine Maley. 2005. “Neanderthal Reconstructed.” The Anatomical Record (Part B: New Anat.) 283 (1): 23–31.
Scerri, Eleanor M. L., Mark G. Thomas, Andrea Manica, Philipp Gunz, Jay T. Stock, Chris Stringer, Matt Grove, et al. 2018. “Did Our Species Evolve in Subdivided Populations Across Africa, and Why Does It Matter?” Trends in Ecology & Evolution 33 (8): 582–594.
Shea, John J. 2011. “Refuting a Myth about Human Origins.” American Scientist 99 (2): 128–135.
Shea, John J., and Ofer Bar-Yosef. 2005. “Who Were the Skhul/Qafzeh People? An Archaeological Perspective on Eurasia’s Oldest Modern Humans.” Journal of the Israel Prehistoric Society 35: 451–468.
Slatkin, Montgomery, and Fernando Racimo. 2016. “Ancient DNA and Human History.” Proceedings of the National Academy of Sciences 113 (23): 6380–6387.
Smithsonian Institution. (2023, June 26). Humans’ evolutionary relatives butchered one another 1.45 million years ago. Smithsonian Institution. https://www.si.edu/newsdesk/releases/humans-evolutionary-relatives-butchered-one-another-145-million-years-ago
Smith, Fred H., James C. M. Ahern, Ivor Janković, and Ivor Karavanić. 2017. “The Assimilation Model of Modern Human Origins in Light of Current Genetic and Genomic Knowledge.” Quaternary International 450: 126–136.
Smith, Michael. 2009. “V. Gordon Childe and the Urban Revolution: A Historical Perspective on a Revolution in Urban Studies.” Town Planning Review 80 (1): 3–29.
Spence, M., & Jackson, L. J. (2014). The Bioarchaeology of Cannibalism at the Charity Site. Journal of The Ontario Archaeological Society , 94, 65–80. https://www.academia.edu/14371786/The_Bioarchaeology_of_Cannibalism_at_the_Charity_Site
Stock, Jay T. 2008. “Are Humans Still Evolving?” EMBO Reports 9 (Suppl 1): S51–S54.
Swisher, Mark E., Dennis L. Jenkins, Lionel E. Jackson Jr., and Fred M. Phillips. 2013. “A Reassessment of the Role of the Canadian Ice-Free Corridor in Light of New Geological Evidence.” Poster Symposium 5B: Geology, Geochronology and Paleoenvironments of the First Americans at the Paleoamerican Odyssey Conference, Santa Fe, New Mexico, October 16–19.
Thorne, A. G., and P. G. Macumber. 1972. “Discoveries of Late Pleistocene Man at Kow Swamp, Australia.” Nature 238 (5363): 316–319.
Trinkaus, Erik, Ştefan Milota, Ricardo Rodrigo, Gherase Mircea, and Oana Moldovan. 2003a. “Early Modern Human Cranial Remains from the Peştera Cu Oase, Romania.” Journal of Human Evolution 45 (3): 245–253.
Trinkaus, Erik, Oana Moldovan, Adrian Bîlgăr, Laurenţiu Sarcina, Sheela Athreya, Shara E Bailey, Ricardo Rodrigo, Gherase Mircea, Thomas Higham, and Christopher Bronk Ramsey. 2003b. “An Early Modern Human from the Peştera Cu Oase, Romania.” Proceedings of the National Academy of Sciences 100 (20): 11231–11236.
Ullrich, H. (2005). Cannibalistic rites within mortuary practices from the palaeolithic to middle aged in Europe. Anthropologie (1962-), 43(2/3), 249–261. http://www.jstor.org/stable/26292739
Velemínská, J., J. Brůzek, P. Velemínský, L. Bigoni, A. Sefcáková, and S. Katina. 2008. “Variability of the Upper-Palaeolithic Skulls from Predmostí Near Prerov (Czech Republic): Craniometric Comparison with Recent Human Standards.” Homo 59 (1): 1–26.
Vidal, Céline M., Christine S. Lane, Asfawossen Asrat, Dan N. Barfod, Darren F. Mark, Emma L. Tomlinson, Ambdemichael Zafu Tadesse, et al. (2022). “Age of the Oldest Known Homo sapiens from Eastern Africa. Nature 601 (7894): 579–583.
Villa, P., Bouville, C., Courtin, J., Helmer, D., Mahieu, E., Shipman, P., Belluomini, G. & Branca, M. (1986). Cannibalism in the Neolithic. Science, 233(4762), 431–437. doi:10.1126/science.233.4762.431
Villa, Paola, Sylvain Soriano, Tsenka Tsanova, Ilaria Degano, Thomas F. G. Higham, Francesco d’Errico, Lucinda Backwell, Jeannette J. Lucejko, Maria Perla Colombini, and Peter B. Beaumont. 2012. “Border Cave and the Beginning of the Later Stone Age in South Africa.” Proceedings of the National Academy of Sciences 109 (33): 13208–13213.
Wall, Jeffrey D., and Deborah Yoshihara Caldeira Brandt. 2016. “Archaic Admixture in Human History.” Current Opinion in Genetics & Development 41: 93–97.
White, Tim D., Berhane Asfaw, David DeGusta, Henry Gilbert, Gary D. Richards, Gen Suwa, and F. Clark Howell. 2003. “Pleistocene Homo sapiens from Middle Awash, Ethiopia.” Nature 423 (6941): 742–747.
Woo, Ju-Kang. 1959. “Human Fossils Found in Liukiang, Kwangsi, China.” Vertebrata PalAsiatica 3 (3): 109–118.
Wu, XiuJie, Wu Liu, Wei Dong, JieMin Que, and YanFang Wang. 2008. “The Brain Morphology of Homo Liujiang Cranium Fossil by Three-Dimensional Computed Tomography.” Chinese Science Bulletin 53 (16): 2513–2519.
Acknowledgments
I could not have undertaken this project without the help of many who got me to where I am today. I extend sincere thank yous to the many colleagues and former students who have inspired me to keep learning and talking about anthropology. Thank you also to all who are involved in this textbook project. The anonymous reviewers truly sparked improvements to the chapter. Lastly, the staff of Starbucks #5772 also contributed immensely to this text.
Keith Chan, Ph.D., Grossmont-Cuyamaca Community College District and MiraCosta College
This chapter is a revision from "Chapter 12: Modern Homo sapiens” by Keith Chan. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Identify the skeletal and behavioral traits that represent modern Homo sapiens.
- Critically evaluate different types of evidence for the origin of our species in Africa and our expansion around the world.
- Understand how the human lifestyle changed when people transitioned from foraging to agriculture.
- Hypothesize how human evolutionary trends may continue into the future.
The walls of a pink limestone cave in the hillside of Jebel Irhoud jutted out of the otherwise barren landscape of the Moroccan desert (Figure 13.1). Miners had excavated the cave in the 1960s, revealing some fossils. In 2007, a re-excavation of the site became a momentous occasion for science. A fossil cranium unearthed by a team of researchers was barely visible to the untrained eye. Just the fossil’s robust brows were peering out of the rock. This research team from the Max Planck Institute for Evolutionary Anthropology was the latest to explore the ancient human presence in this part of North Africa after a find by miners in 1960. Excavating near the first discovery, the researchers wanted to learn more about how Homo sapiens lived far from East Africa, where we thought our species originated.

The scientists were surprised when they analyzed the cranium, named Irhoud 10, and other fossils. Statistical comparisons with other human crania concluded that the Irhoud face shapes were typical of recent modern humans while the braincases matched ancient modern humans. Based on the findings of other scientists, the team expected these modern Homo sapiens fossils to be around 200,000 years old. Instead, dating revealed that the cranium had been buried for around 315,000 years.
Together, the modern-looking facial dimensions and the older date reshaped the interpretation of our species: modern Homo sapiens. Some key evolutionary changes from the archaic Homo sapiens (described in Chapter 11) to our species today happened 100,000 years earlier than we had thought and across the vast African continent rather than concentrated in its eastern region.
This revelation in the study of modern Homo sapiens is just one of the latest in this continually advancing area of biological anthropology. Researchers today are still discovering amazing fossils and ingenious ways to collect data and test hypotheses about our past. Through the collective work of many scientists, we are building an overall theory of modern human origins.
Defining Modernity
What defines modern Homo sapiens when compared to archaic Homo sapiens? Modern humans, like you and me, have a set of derived traits that are not seen in archaic humans or any other hominin. As with other transitions in hominin evolution, such as increasing brain size and bipedal ability, modern traits do not appear fully formed or all at once. In other words, the first modern Homo sapiens was not just born one day from archaic parents. The traits common to modern Homo sapiens appeared in a mosaic manner: gradually and out of sync with one another. There are two areas to consider when tracking the complex evolution of modern human traits. One is the physical change in the skeleton. The other is behavior inferred from the size and shape of the cranium and material culture evidence.
Skeletal Traits
The skeleton of modern Homo sapiens is less robust than that of archaic Homo sapiens. In other words, the modern skeleton is gracile, meaning that the structures are thinner and smoother. Differences related to gracility in the cranium are seen in the braincase, the face, and the mandible. There are also broad differences in the rest of the skeleton.
Cranial Traits

Several elements of the braincase differ between modern and archaic Homo sapiens. Overall, the shape is much rounder, or more globular, on a modern skull (Lieberman, McBratney, and Krovitz 2002; Neubauer, Hublin, and Gunz 2018; Pearson 2008; Figure 13.2). You can feel the globularity of your own modern human skull. Feel the height of your forehead with the palm of your hand. Viewed from the side, the tall vertical forehead of a modern Homo sapiens stands out when compared to the sloping archaic version. This is because the frontal lobe of the modern human brain is larger than the one in archaic humans, and the skull has to accommodate the expansion. The vertical forehead reduces a trait that is common to all other hominins: the brow ridge or supraorbital torus. The parietal lobes of the brain and the matching parietal bones on either side of the skull both bulge outward more in modern humans. At the back of the skull, the archaic occipital bun is no longer present. Instead, the occipital region of the modern human cranium has a derived tall and smooth curve, again reflecting the globular brain inside.
The trend of shrinking face size across hominins reaches its extreme with our species as well. The facial bones of a modern Homo sapiens are extremely gracile compared to all other hominins (Lieberman, McBratney, and Krovitz 2002). Continuing a trend in hominin evolution, technological innovations kept reducing the importance of teeth in reproductive success (Lucas 2007). As natural selection favored smaller and smaller teeth, the surrounding bone holding these teeth also shrank.
Related to smaller teeth, the mandible is also gracile in modern humans when compared to archaic humans and other hominins. Interestingly, our mandibles have pulled back so far from the prognathism of earlier hominins that we gained an extra structure at the most anterior point, called the mental eminence. You know this structure as the chin. At the skeletal level, it resembles an upside-down “T” at the centerline of the mandible (Pearson 2008). Looking back at archaic humans, you will see that they all lack a chin. Instead, their mandibles curve straight back without a forward point. What is the chin for and how did it develop? Flora Gröning and colleagues (2011) found evidence of the chin’s importance by simulating physical forces on computer models of different mandible shapes. Their results showed that the chin acts as structural support to withstand strain on the otherwise gracile mandible.
Postcranial Gracility

The rest of the modern human skeleton is also more gracile than its archaic counterpart. The differences are clear when comparing a modern Homo sapiens with a cold-adapted Neanderthal (Sawyer and Maley 2005), but the trends are still present when comparing modern and archaic humans within Africa (Pearson 2000). Overall, a modern Homo sapiens postcranial skeleton has thinner cortical bone, smoother features, and more slender shapes when compared to archaic Homo sapiens (Figure 13.3). Comparing whole skeletons, modern humans have longer limb proportions relative to the length and width of the torso, giving us lankier outlines.
Why is our skeleton so gracile compared to those of other hominins? Natural selection can drive the gracilization of skeletons in several ways (Lieberman 2015). A slender frame is believed to be adapted for the efficient long-distance running ability that started with Homo erectus. Furthermore, it is argued that slenderness is a genetic adaptation for cooling an active body in hotter climates, which aligns with the ample evidence that Africa was the home continent of our species.
Behavioral Modernity
Aside from physical differences in the skeleton, researchers have also uncovered evidence of behavioral changes associated with increased cultural complexity from archaic to modern humans. How did cultural complexity develop? Two investigations into this question are archaeology and the analysis of reconstructed brains.
Archaeology tells us much about the behavioral complexity of past humans by interpreting the significance of material culture. In terms of advanced culture, items created with an artistic flair, or as decoration, speak of abstract thought processes (Figure 13.4). The demonstration of difficult artistic techniques and technological complexity hints at social learning and cooperation as well. According to paleoanthropologist John Shea (2011), one way to track the complexity of past behavior through artifacts is by measuring the variety of tools found together. The more types of tools constructed with different techniques and for different purposes, the more modern the behavior. Researchers are still working on an archaeological way to measure cultural complexity that is useful across time and place.

The interpretation of brain anatomy is another promising approach to studying the evolution of human behavior. When looking at investigations on this topic in modern Homo sapiens brains, researchers found a weak association between brain size and test-measured intelligence (Pietschnig et al. 2015). Additionally, they found no association between intelligence and biological sex. These findings mean that there are more significant factors that affect tested intelligence than just brain size. Since the sheer size of the brain is not useful for weighing intelligence within a species, paleoanthropologists are instead investigating the differences in certain brain structures. The differences in organization between modern Homo sapiens brains and archaic Homo sapiens brains may reflect different cognitive priorities that account for modern human culture. As with the archaeological approach, new discoveries will refine what we know about the human brain and apply that knowledge to studying the distant past.
Taken together, the cognitive abilities in modern humans may have translated into an adept use of tools to enhance survival. Researchers Patrick Roberts and Brian A. Stewart (2018) call this concept the generalist-specialist niche: our species is an expert at living in a wide array of environments, with populations culturally specializing in their own particular surroundings. The next section tracks how far around the world these skeletal and behavioral traits have taken us.
First Africa, Then the World
What enabled modern Homo sapiens to expand its range further in 300,000 years than Homo erectus did in 1.5 million years? The key is the set of derived biological traits from the last section. It is theorized that the gracile frame and neurological anatomy allowed modern humans to survive and even flourish in the vastly different environments they encountered. Based on multiple types of evidence, the source of all of these modern humans was Africa. Instead of originating from just one location, evidence shows that modern Homo sapiens evolution occurred in a complex gene flow network across Africa, a concept called African multiregionalism (Scerri et al. 2018).
This section traces the origin of modern Homo sapiens and the massive expansion of our species across all of the continents (except Antarctica) by 12,000 years ago. While modern Homo sapiens first shared geography with archaic humans, modern humans eventually spread into lands where no human had gone before. Figure 13.5 shows the broad routes that our species took expanding around the world. I encourage you to make your own timeline with the dates in this part to see the overall trends.




Modern Homo sapiens Biology and Culture in Africa
We start with the ample fossil evidence supporting the theory that modern humans originated in Africa during the Middle Pleistocene, having evolved from African archaic Homo sapiens. The earliest dated fossils considered to be modern actually have a mosaic of archaic and modern traits, showing the complex changes from one type to the other. Experts have various names for these transitional fossils, such as Early Modern Homo sapiens or Early Anatomically Modern Humans. However they are labeled, the presence of some modern traits means that they illustrate the origin of the modern type. Three particularly informative sites with fossils of the earliest modern Homo sapiens are Jebel Irhoud, Omo, and Herto.

Recall from the start of the chapter that the most recent finds at Jebel Irhoud are now the oldest dated fossils that exhibit some facial traits of modern Homo sapiens. Besides Irhoud 10, the cranium that was dated to 315,000 years ago (Hublin et al. 2017; Richter et al. 2017), there were other fossils found in the same deposit that we now know are from the same time period. In total there are at least five individuals, representing life stages from childhood to adulthood. These fossils form an image of high variation in skeletal traits. For example, the skull named Irhoud 1 has a primitive brow ridge, while Irhoud 2 and Irhoud 10 do not (Figure 13.6). The braincases are lower than what is seen in the modern humans of today but higher than in archaic Homo sapiens. The teeth also have a mix of archaic and modern traits that defy clear categorization into either group.
Research separated by nearly four decades uncovered fossils and artifacts from the Kibish Formation in the Lower Omo Valley in Ethiopia. These Omo Kibish hominins were represented by braincases and fragmented postcranial bones of three individuals found kilometers apart, dating back to around 233,000 years ago (Day 1969; McDougall, Brown, and Fleagle 2005; Vidal et al. 2022). One interesting finding was the variation in braincase size between the two more-complete specimens: while the individual named Omo I had a more globular dome, Omo II had an archaic-style long and low cranium.
Also in Ethiopia, a team led by Tim White (2003) excavated numerous fossils at Herto. There were fossilized crania of two adults and a child, along with fragments of more individuals. The dates ranged between 160,000 and 154,000 years ago. The skeletal traits and stone-tool assemblage were both intermediate between the archaic and modern types. Features reminiscent of modern humans included a tall braincase and thinner zygomatic (cheek) bones than those of archaic humans (Figure 13.7). Still, some archaic traits persisted in the Herto fossils, such as the supraorbital tori. Statistical analysis by other research teams concluded that at least some cranial measurements fit just within the modern human range (McCarthy and Lucas 2014), favoring categorization with our own species.

The timeline of material culture suggests a long period of relying on similar tools before a noticeable diversification of artifacts types. Researchers label the time of stable technology shared with archaic types the Middle Stone Age, while the subsequent time of diversification in material culture is called the Later Stone Age.
In the Middle Stone Age, the sites of Jebel Irhoud, Omo, and Herto all bore tools of the same flaked style as archaic assemblages, even though they were separated by almost 150,000 years. The consistency in technology may be evidence that behavioral modernity was not so developed. No clear signs of art dating back this far have been found either. Other hypotheses not related to behavioral modernity could explain these observations. The tool set may have been suitable for thriving in Africa without further innovation. Maybe works of art from that time were made with media that deteriorated or perhaps such art was removed by later humans.
Evidence of what Homo sapiens did in Africa from the end of the Middle Stone Age to the Later Stone Age is concentrated in South African cave sites that reveal the complexity of human behavior at the time. For example, Blombos Cave, located along the present shore of the Cape of Africa facing the Indian Ocean, is notable for having a wide variety of artifacts. The material culture shows that toolmaking and artistry were more complex than previously thought for the Middle Stone Age. In a layer dated to 100,000 years ago, researchers found two intact ochre-processing kits made of abalone shells and grinding stones (Henshilwood et al. 2011). Marine snail shell beads from 75,000 years ago were also excavated (Figure 13.8; d’Errico et al. 2005). Together, the evidence shows that the Middle Stone Age occupation at Blombos Cave incorporated resources from a variety of local environments into their culture, from caves (ochre), open land (animal bones and fat), and the sea (abalone and snail shells). This complexity shows a deep knowledge of the region’s resources and their use—not just for survival but also for symbolic purposes.

On the eastern coast of South Africa, Border Cave shows new African cultural developments at the start of the Later Stone Age. Paola Villa and colleagues (2012) identified several changes in technology around 43,000 years ago. Stone-tool production transitioned from a slower process to one that was faster and made many microliths, small and precise stone tools. Changes in decorations were also found across the Later Stone Age transition. Beads were made from a new resource: fragments of ostrich eggs shaped into circular forms resembling present-day breakfast cereal O’s (d’Errico et al. 2012). These beads show a higher level of altering one’s own surroundings and a move from the natural to the abstract in terms of design.
Expansion into the Middle East and Asia
While modern Homo sapiens lived across Africa, some members eventually left the continent. These pioneers could have used two connections to the Middle East or West Asia. From North Africa, they could have crossed the Sinai Peninsula and moved north to the Levant, or eastern Mediterranean. Finds in that region show an early modern human presence. Other finds support the Southern Dispersal model, with a crossing from East Africa to the southern Arabian Peninsula through the Straits of Bab-el-Mandeb. It is tempting to think of one momentous event in which people stepped off Africa and into the Middle East, never to look back. In reality, there were likely multiple waves of movement producing gene flow back and forth across these regions as the overall range pushed east. The expanding modern human population could have thrived by using resources along the southern coast of the Arabian Peninsula to South Asia, with side routes moving north along rivers. The maximum range of the species then grew across Asia.
Modern Homo sapiens in the Middle East
Geographically, the Middle East is the ideal place for the African modern Homo sapiens population to inhabit upon expanding out of their home continent. In the Eastern Mediterranean coast of the Levant, there is a wealth of skeletal and material culture linked to modern Homo sapiens. Recent discoveries from Saudi Arabia further add to our view of human life just beyond Africa.
The Caves of Mount Carmel in present-day Israel have preserved skeletal remains and artifacts of modern Homo sapiens, the first-known group living outside Africa. The skeletal presence at Misliya Cave is represented by just part of the left upper jaw of one individual, but it is notable for being dated to a very early time, between 194,000 and 177,000 years ago (Hershkovitz et al. 2018). Later, from 120,000 to 90,000 years ago, fossils of multiple individuals across life stages were found in the caves of Es-Skhul and Qafzeh (Shea and Bar-Yosef 2005). The skeletons had many modern Homo sapiens traits, such as globular crania and more gracile postcranial bones when compared to Neanderthals. Still, there were some archaic traits. For example, the adult male Skhul V also possessed what researchers Daniel Lieberman, Osbjorn Pearson, and Kenneth Mowbray (2000) called marked or clear occipital bunning. Also, compared to later modern humans, the Mount Carmel people were more robust. Skhul V had a particularly impressive brow ridge that was short in height but sharply jutted forward above the eyes (Figure 13.9). The high level of preservation is due to the intentional burial of some of these people. Besides skeletal material, there are signs of artistic or symbolic behavior. For example, the adult male Skhul V had a boar’s jaw on his chest. Similarly, Qafzeh 11, a juvenile with healed cranial trauma, had an impressive deer antler rack placed over his torso (Figure 13.10; Coqueugniot et al. 2014). Perforated seashells colored with ochre, mineral-based pigment, were also found in Qafzeh (Bar-Yosef Mayer, Vandermeersch, and Bar-Yosef 2009).


One remaining question is, what happened to the modern humans of the Levant after 90,000 years ago? Another site attributed to our species did not appear in the region until 47,000 years ago. Competition with Neanderthals may have accounted for the disappearance of modern human occupation since the Neanderthal presence in the Levant lasted longer than the dates of the early modern Homo sapiens. John Shea and Ofer Bar-Yosef (2005) hypothesized that the Mount Carmel modern humans were an initial expansion from Africa that failed. Perhaps they could not succeed due to competition with the Neanderthals who had been there longer and had both cultural and biological adaptations to that environment.
Modern Homo sapiens of China
A long history of paleoanthropology in China has found ample evidence of modern human presence. Four notable sites are the caves at Fuyan, Liujiang, Tianyuan, and Zhoukoudian. In the distant past, these caves would have been at least seasonal shelters that unintentionally preserved evidence of human presence for modern researchers to discover.
At Fuyan Cave in Southern China, paleoanthropologists found 47 adult teeth associated with cave formations dated to between 120,000 and 80,000 years ago (Liu et al. 2015). It is currently the oldest-known modern human site in China, though other researchers question the validity of the date range (Michel et al. 2016). The teeth have the small size and gracile features of modern Homo sapiens dentition.
The fossil Liujiang (or Liukiang) hominin (67,000 years ago) has derived traits that classified it as a modern Homo sapiens, though primitive archaic traits were also present. In the skull, which was found nearly complete, the Liujiang hominin had a taller forehead than archaic Homo sapiens but also had an enlarged occipital region (Figure 13.11; Brown 1999; Wu et al. 2008). Other parts of the skeleton also had a mix of modern and archaic traits: for example, the femur fragments suggested a slender length but with thick bone walls (Woo 1959).

Another Chinese site to describe here is the one that has been studied the longest. In the Zhoukoudian Cave system (Figure 13.12), where Homo erectus and archaic Homo sapiens have also been found, there were three crania of modern Homo sapiens. These crania, which date to between 34,000 and 10,000 years ago, were all more globular than those of archaic humans but still lower and longer than those of later modern humans (Brown 1999; Harvati 2009). When compared to one another, the crania showed significant differences from one another. Comparison of cranial measurements to other populations past and present found no connection with modern East Asians, again showing that human variation was very different from what we see today.

Crossing to Australia
Expansion of the first modern human Asians, still following the coast, eventually entered an area that researchers call Sunda before continuing on to modern Australia. Sunda was a landmass made up of the modern-day Malay Peninsula, Sumatra, Java, and Borneo. Lowered sea levels connected these places with land bridges, making them easier to traverse. Proceeding past Sunda meant navigating Wallacea, the archipelago that includes the Indonesian islands east of Borneo. In the distant past, there were many megafauna, large animals that migrating humans would have used for food and materials (such as utilizing animals’ hides and bones). Further southeast was another landmass called Sahul, which included New Guinea and Australia as one contiguous continent. Based on fossil evidence, this land had never seen hominins or any other primates before modern Homo sapiens arrived. Sites along this path offer clues about how our species handled the new environment to live successfully as foragers.

The skeletal remains at Lake Mungo, land traditionally owned by Mutthi Mutthi, Ngiampaa, and Paakantji peoples, are the oldest known in the continent. The now-dry lake was one of a series located along the southern coast of Australia in New South Wales, far from where the first people entered from the north (Barbetti and Allen 1972; Bowler et al. 1970). Two individuals dating to around 40,000 years ago show signs of artistic and symbolic behavior, including intentional burial. The bones of Lake Mungo 1 (LM1), an adult female, were crushed repeatedly, colored with red ochre, and cremated (Bowler et al. 1970). Lake Mungo 3 (LM3), a tall, older male with a gracile cranium but robust postcranial bones, had his fingers interlocked over his pelvic region (Brown 2000).
Kow Swamp, within traditional Yorta Yorta land also in southern Australia, contained human crania that looked distinctly different from the ones at Lake Mungo (Durband 2014; Thorne and Macumber 1972). The crania, dated between 9,000 and 20,000 years ago, had extremely robust brow ridges and thick bone walls, but these were paired with globular features on the braincase (Figure 13.13).
While no fossil humans have been found at the Madjedbebe rock shelter in the North Territory of Australia, more than 10,000 artifacts found there show both behavioral modernity and variability (Clarkson et al. 2017). They include a diverse array of stone tools and different shades of ochre for rock art, including mica-based reflective pigment (similar to glitter). These impressive artifacts are as far back as 56,000 years old, providing the date for the earliest-known presence of humans in Australia.
From the Levant to Europe
The first modern human expansion into Europe occurred after other members of our species settled in East Asia and Australia. As the evidence from the Levant suggests, modern human movement to Europe may have been hampered by the presence of Neanderthals. It is suggested that another obstacle was the colder climate, which was incompatible with the biology of modern Homo sapiens from Africa, as they were adapted to high temperatures and ultraviolet radiation. Still, by 40,000 years ago, modern Homo sapiens had a detectable presence. This time was also the start of the Later Stone Age or Upper Paleolithic, when there was an expansion in cultural complexity. There is a wealth of evidence from this region due to a Western bias in research, the proximity of these findings to Western scientific institutions, and the desire of Western scientists to explore their own past.

In Romania, the site of Peștera cu Oase (Cave of Bones) had the oldest-known remains of modern Homo sapiens in Europe, dated to around 40,000 years ago (Trinkaus et al. 2003a). Among the bones and teeth of many animals were the fragmented cranium of one person and the mandible of another (the two bones did not fit each other). Both bones have modern human traits similar to the fossils from the Middle East, but they also had Neanderthal traits. Oase 1, the mandible, had a mental eminence but also extremely large molars (Trinkaus et al. 2003b). This mandible has yielded DNA that surprisingly is equally similar to DNA from present-day Europeans and Asians (Fu et al. 2015). This means that Oase 1 was not the direct ancestor of modern Europeans. The Oase 2 cranium has the derived traits of reduced brow ridges along with archaic wide zygomatic cheekbones and an occipital bun (Figure 13.14; Rougier et al. 2007).
Dating to around 26,000 years ago, Předmostí near Přerov in the Czech Republic was a site where people buried over 30 individuals along with many artifacts. Eighteen individuals were found in one mass burial area, a few covered by the scapulae of woolly mammoths (Germonpré, Lázničková-Galetová, and Sablin 2012). The Předmostí crania were more globular than those of archaic humans but tended to be longer and lower than in later modern humans (Figure 13.15; Velemínská et al. 2008). The height of the face was in line with modern residents of Central Europe. There was also skeletal evidence of dog domestication, such as the presence of dog skulls with shorter snouts than in wild wolves (Germonpré, Lázničková-Galetová, and Sablin et al. 2012). In total, Předmostí could have been a settlement dependent on mammoths for subsistence and the artificial selection of early domesticated dogs.

The sequence of modern Homo sapiens technological change in the Later Stone Age has been thoroughly dated and labeled by researchers working in Europe. Among them, the Gravettian tradition of 33,000 years to 21,000 years ago is associated with most of the known curvy female figurines, often assumed to be “Venus” figures. Hunting technology also advanced in this time with the first known boomerang, atlatl (spear thrower), and archery. The Magdalenian tradition spread from 17,000 to 12,000 years ago. This culture further expanded on fine bone tool work, including barbed spearheads and fishhooks (Figure 13.16).

Among the many European sites dating to the Later Stone Age, the famous cave art sites deserve mention. Chauvet-Pont-d'Arc Cave in southern France dates to separate Aurignacian occupations 31,000 years ago and 26,000 years ago. Over a hundred art pieces representing 13 animal species are preserved, from commonly depicted deer and horses to rarer rhinos and owls. Another French cave with art is Lascaux, which is several thousand years younger at 17,000 years ago in the Magdalenian period. At this site, there are over 6,000 painted figures on the walls and ceiling (Figure 13.17). Scaffolding and lighting must have been used to make the paintings on the walls and ceiling deep in the cave. Overall, visiting Lascaux as a contemporary must have been an awesome experience: trekking deeper in the cave lit only by torches giving glimpses of animals all around as mysterious sounds echoed through the galleries.

Peopling of the Americas
By 25,000 years ago, our species was the only member of Homo left on Earth. Gone were the Neanderthals, Denisovans, Homo naledi, and Homo floresiensis. The range of modern Homo sapiens kept expanding eastward into—using the name given to this area by Europeans much later—the Western Hemisphere. This section will address what we know about the peopling of the Americas, from the first entry to these continents to the rapid spread of Indigenous Americans across its varied environments.
While evidence points to an ancient land bridge called Beringia that allowed people to cross from what is now northeastern Siberia into modern-day Alaska, what people did to cross this land bridge is still being investigated. For most of the 20th century, the accepted theory was the Ice-Free Corridor model. It stated that northeast Asians (East Asians and Siberians) first expanded across Beringia inland through a passage between glaciers that opened into the western Great Plains of the United States, just east of the Rocky Mountains, around 13,000 years ago (Swisher et al. 2013). While life up north in the cold environment would have been harsh, migrating birds and an emerging forest might have provided sustenance as generations expanded through this land (Potter et al. 2018).
However, in recent decades, researchers have accumulated evidence against the Ice-Free Corridor model. Archaeologist K. R. Fladmark (1979) brought the alternate Coastal Route model into the archaeological spotlight; researcher Jon M. Erlandson has been at the forefront of compiling support for this theory (Erlandson et al. 2015). The new focus is the southern edge of the land bridge instead of its center: About 16,000 years ago, members of our species expanded along the coastline from northeast Asia, east through Beringia, and south down the Pacific Coast of North America while the inland was still sealed off by ice. The coast would have been free of ice at least part of the year, and many resources would have been found there, such as fish (e.g., salmon), mammals (e.g., whales, seals, and otters), and plants (e.g., seaweed).
South through the Americas
When the first modern Homo sapiens reached the Western Hemisphere, the spread through the Americas was rapid. Multiple migration waves crossed from North to South America (Posth et al. 2018). Our species took advantage of the lack of hominin competition and the bountiful resources both along the coasts and inland. The Americas had their own wide array of megafauna, which included woolly mammoths (Figure 13.18), mastodons, camels, horses, ground sloths, giant tortoises, and—a favorite of researchers—a two-meter-tall beaver. The reason we cannot see these amazing animals today may be that resources gained from these fauna were crucial to the survival for people over 12,000 years ago (Araujo et al. 2017). Several sites are notable for what they add to our understanding of the distant past in the Americas, including interactions with megafauna and other elements of the environment.

A 2019 discovery may allow researchers to improve theories about the peopling of the Americas. In White Sands National Park, New Mexico, 60 human footprints have been astonishingly dated to around 22,000 years ago (Bennett et al. 2021). This date and location do not match either the Ice-Free Corridor or Coastal Route models. Researchers are now working to verify the find and adjust previous models to account for the new evidence. This groundbreaking find is sparking new theories; it is another example of the fast pace of research performed on our past.
Monte Verde is a landmark site that shows that the human population had expanded down the whole vertical stretch of the Americas to Chile by 14,600 years ago. The site has been excavated by archaeologist Tom D. Dillehay and his team (2015). The remains of nine distinct edible species of seaweed at the site shows familiarity with coastal resources and relates to the Coastal Route model by showing a connection between the inland people and the sea.

Named after the town in New Mexico, the Clovis stone-tool style is the first example of a widespread culture across much of North America, between 13,400 and 12,700 years ago (Miller, Holliday, and Bright 2013). Clovis points were fluted with two small projections, one on each end of the base, facing away from the head (Figure 13.19). The stone points found at this site match those found as far as the Canadian border and northern Mexico, and from the west coast to the east coast of the United States. Fourteen Clovis sites also contained the remains of mammoths or mastodons, suggesting that hunting megafauna with these points was an important part of life for the Clovis people. After the spread of the Clovis style, it diversified into several regional styles, keeping some of the Clovis form but also developing their own unique touches.
The Big Picture: The Assimilation Hypothesis
How do researchers make sense of all of these modern Homo sapiens discoveries that cover over 300,000 years of time and stretch across every continent except Antarctica? How was modern Homo sapiens related to archaic Homo sapiens?
The Assimilation hypothesis proposes that modern Homo sapiens evolved in Africa first and expanded out but also interbred with the archaic Homo sapiens they encountered outside Africa (Figure 13.20). This hypothesis is powerful since it explains why Africa has the oldest modern human fossils, why early modern humans found in Europe and Asia bear a resemblance to the regional archaics, and why traces of archaic DNA can be found in our genomes today (Dannemann and Racimo 2018; Reich et al. 2010; Reich et al. 2011; Slatkin and Racimo 2016; Smith et al. 2017; Wall and Yoshihara Caldeira Brandt 2016).

While researchers have produced a model that satisfies the data, there are still a lot of questions for paleoanthropologists to answer regarding our origins. What were the patterns of migration in each part of the world? Why did the archaic humans go extinct? In what ways did archaic and modern humans interact? The definitive explanation of how our species started and what our ancestors did is still out there to be found. You are now in a great place to welcome the next discovery about our distant past—maybe you’ll even contribute to our understanding as well.
The Chain Reaction of Agriculture
While it may be hard to imagine today, for most of our species’ existence we were nomadic: moving through the landscape without a singular home. Instead of a refrigerator or pantry stocked with food, we procured nutrition and other resources as needed based on what was available in the environment. This section gives an overview of how the foraging lifestyle enabled the expansion of our species and how the invention of a new way of life caused a chain reaction of cultural change.
The Foraging Tradition
There are a variety of possible subsistence strategies, or methods of finding sustenance and resources. To understand our species is to understand the subsistence strategy of foraging, or the search for resources in the environment. While most (but not all) humans today live in cultures that practice agriculture (whereby we greatly shape the environment to mass produce what we need), we have spent far more time as nomadic foragers than as settled agriculturalists. As such, it has been suggested that our traits have evolved to be primarily geared toward foraging. For instance, our efficient bipedalism allows persistence-hunting across long distances as well as movement from resource to resource.
How does human foraging, also known as hunting and gathering, work? Anthropologists have used all four fields to answer this question (see Ember n.d.). Typically, people formed bands, or kin-based groups of around 50 people or less (rarely over 100). A band’s organization would be egalitarian, with a flexible hierarchy based on an individual’s age, level of experience, and relationship with others. Everyone would have a general knowledge of the skills assigned to their gender roles, rather than specializing in different occupations. A band would be able to move from place to place in the environment, using knowledge of the area to forage (Figure 13.21). In varied environments—from savannas to tropical forests, deserts, coasts, and the Arctic circle—people found sustenance needed for survival.

Humans made extensive use of the foraging subsistence strategy, but this lifestyle did have limitations. The ease of foraging depended on the richness of the environment. Due to the lack of storage, resources had to be dependably found when needed. While a bountiful environment would require just a few hours of foraging a day and could lead to a focus on one location, the level and duration of labor increased greatly in poor or unreliable environments. Labor was also needed to process the acquired resources, which contributed to the foragers’ daily schedule (Crittenden and Schnorr 2017).
The adaptations to foraging found in modern Homo sapiens may explain why our species became so successful both within Africa and in the rapid expansion around the world. Overcoming the limitations, each generation at the edge of our species’s range would have found it beneficial to expand a little further, keeping contact with other bands but moving into unexplored territory where resources were more plentiful. The cumulative effect would have been the spread of modern Homo sapiens across continents and hemispheres.
Why Agriculture?
After hundreds of thousands of years of foraging, some groups of people around 12,000 years ago started to practice agriculture. This transition, called the Neolithic Revolution, occurred at the start of the Holocene epoch. While the reasons for this global change are still being investigated, two likely co-occurring causes are a growing human population and natural global climate change.
Overcrowding could have affected the success of foraging in the environment, leading to the development of a more productive subsistence strategy (Cohen 1977). Foraging works best with low population densities since each band needs a lot of space to support itself. If too many people occupy the same environment, they deplete the area faster. The high population could exceed the carrying capacity, or number of people a location can reliably support. Reaching carrying capacity on a global level due to growing population and limited areas of expansion would have been an increasingly pressing issue after the expansion through the major continents by 14,600 years ago.
A changing global climate immediately preceded the transition to agriculture, so researchers have also explored a connection between the two events. Since the Last Glacial Maximum of 23,000 years ago, the Earth slowly warmed. Then, from 13,000 to 11,700 years ago, the temperature in most of the Northern Hemisphere dropped suddenly in a phenomenon called the Younger Dryas. Glaciers returned in Europe, Asia, and North America. In Mesopotamia, which includes the Levant, the climate changed from warm and humid to cool and dry. The change would have occurred over decades, disrupting the usual nomadic patterns and subsistence of foragers around the world. The disruption to foragers due to the temperature shift could have been a factor in spurring a transition to agriculture. Researchers Gregory K. Dow and colleagues (2009) believe that foraging bands would have clustered in the new resource-rich places where people started to direct their labor to farming the limited area. After the Younger Dryas ended, people expanded out of the clusters with their agricultural knowledge (Figure 13.22).

The double threat of the limitation of human continental expansion and the sudden global climate change may have placed bands in peril as more populations outpaced their environment’s carrying capacity. Not only had a growing population led to increased competition with other bands, but environments worldwide had shifted to create more uncertainty. As such, it has been proposed that as people in different areas around the world faced this unpredictable situation, they became the independent inventors of agriculture.
Agriculture around the World
Due to global changes to the human experience starting from 12,000 years ago, it has been suggested that cultures with no knowledge of each other turned toward intensely farming their local resources (see Figure 13.22). It is proposed that the first farmers engaged in artificial selection of their domesticates to enhance useful traits over generations. The switch to agriculture took time and effort with no guarantee of success and constant challenges (e.g. fires, droughts, diseases, and pests). The regions with the most widespread impact in the face of these obstacles became the primary centers of agriculture (Figure 13.23; Fuller 2010):
- Mesopotamia: The Fertile Crescent from the Tigris and Euphrates rivers through the Levant was where bands started to domesticate plants and animals around 12,000 years ago. The connection between the development of agriculture and the Younger Dryas was especially strong here. Farmed crops included wheat, barley, peas, and lentils. This was also where cattle, pigs, sheep, and goats were domesticated.
- South and East Asia: Multiple regions across this land had varieties of rice, millet, and soybeans by 10,000 years ago. Pigs were farmed with no connection to Mesopotamia. Chickens were also originally from this region, bred for fighting first and food second.
- New Guinea: Agriculture started here 10,000 years ago. Bananas, sugarcane, and taro were native to this island. Sweet potatoes were brought back from voyages to South America around the year C.E. 1000. No known animal farming occurred here.
- Mesoamerica: Agriculture from Central Mexico to northern South America also occurred from 10,000 years ago; it was also only plant based. Maize was a crop bred from teosinte grass, which has become one of the global staples. Beans, squash, and avocados were also grown in this region.
- The Andes: Starting around 8,000 years ago, local domesticated plants started with squash but later included potatoes, tomatoes, beans, and quinoa. Maize was brought down from Mesoamerica. The main farm animals were llamas, alpacas, and guinea pigs.
- Sub-Saharan Africa: This region went through a change 5,000 years ago called the Bantu expansion. The Bantu agriculturalists were established in West Central Africa and then expanded south and east. Native varieties of rice, yams, millet, and sorghum were grown across this area. Cattle were also domesticated here.
- Eastern North America: This region was the last major independent agriculture center, from 4,000 years ago. Squash and sunflower are the produce from this region that are most known today, though sumpweed and pitseed goosefoot were also farmed. Hunting was still the main source of animal products.

By 5,000 years ago, our species was well within the Neolithic Revolution. Agriculturalists spread to neighboring parts of the world with their domesticates, further expanding the use of this subsistence strategy. From this point, the human species changed from being primarily foragers to primarily agriculturalists with skilled control of their environments. The planet changed from mostly unaffected by human presence to being greatly transformed by humans. The revolution took millennia, but it was a true revolution as our species’ lifestyle was dramatically reshaped.
Cultural Effects of Agriculture
The worldwide adoption of agriculture altered the course of human culture and history forever. The core change in human culture due to agriculture is the move toward not moving: rather than live a nomadic lifestyle, farmers had to remain in one area to tend to their crops and livestock. The term for living bound to a certain location is sedentarism. This led to new aspects of life that were uncommon among foragers: the construction of permanent shelters and agricultural infrastructure, such as fields and irrigation, plus the development of storage technology, such as pottery, to preserve extra resources in case of future instability.

The high productivity of successful agriculture sparked further changes (Smith 2009). It is argued that since successful agriculture produced a much greater amount of food and other resources per unit of land compared to foraging, the population growth rate skyrocketed. The surplus of a bountiful harvest also provided insurance for harder times, reducing the risk of famine. Changes happened to society as well. With a few farming households producing enough food to feed many others, other people could focus on other tasks. So began specialization into different occupations such as craftspeople, traders, religious figures, and artists, spurring innovation in these areas as people could now devote time and effort toward specific skills. These interdependent people would settle an area together for convenience. The growth of these settlements led to urbanization, the founding of cities that became the foci of human interaction (Figure 13.24).
The formation of cities led to new issues that sparked the growth of further specializations, called institutions. These are cultural constructs that exist beyond the individual and have wide control over a population. Leadership of these cities became hierarchical with different levels of rank and control. The stratification of society increased social inequality between those with more or less power over others. Under leadership, people built impressive monumental architecture, such as pyramids and palaces, that embodied the wealth and power of these early cities. Alliances could unite cities, forming the earliest states. In several regions of the world, state organization expanded into empires, wide-ranging political entities that covered a variety of cultures.
Urbanization brought new challenges as well. The concentration of sedentary peoples was ideal for infectious diseases to thrive since they could jump from person to person and even from livestock to person (Armelagos, Brown, and Turner 2005). While successful agriculture provided a large surplus of food to thwart famine, the food produced offered less diverse food sources than foragers’ diets (Cohen and Armelagos 1984; Cohen and Crane-Kramer 2007). This shift in nutrition caused other diseases to flourish among those who adopted farming, such as dental cavities and malocclusion (the misalignment of teeth caused by soft, agricultural diets). The need to extract “wisdom teeth” or third molars seen in agricultural cultures today stems from this misalignment between the environment our ancestors adapted to and our lifestyles today.
As the new disease trends show, the adoption of agriculture and the ensuing cultural changes were not entirely positive. It is also important to note that this is not an absolutely linear progression of human culture from simple to complex. In many cases, empires have collapsed and, in some cases, cities dispersed to low-density bands that rejected institutions. However, a global trend has emerged since the adoption of agriculture, wherein population and social inequality have increased, leading to the massive and influential nation-states of today.
The rise of states in Europe has a direct impact on many of this book’s topics. Science started as a European cultural practice by the upper class that became a standardized way to study the world. Education became an institution to provide a standardized path toward producing and gaining knowledge. The scientific study of human diversity, embroiled in the race concept that still haunts us today, was connected to the European slave trade and colonialism.
Also starting in Europe, the Industrial Revolution of the 19th century turned cities into centers of mass manufacturing and spurred the rapid development of inventions (Figure 13.25). In the technologically interconnected world of today, human society has reached a new level of complexity with globalization. In this system, goods are mass-produced and consumed in different parts of the world, weakening the reliance on local farms and factories. The imbalanced relationship between consumers and producers of goods further increases economic inequality.

As states based on agriculture and industry keep exerting influence on humanity today, there are people, like the Hadzabe of Tanzania, who continue to live a lifestyle centered on foraging. Due to the overwhelming force that agricultural societies exert, foragers today have been marginalized to live in the least habitable parts of the world—the areas that are not conducive to farming, such as tropical rainforests, deserts, and the Arctic (Headland et al. 1989). Foragers can no longer live in the abundant environments that humans would have enjoyed before the Neolithic Revolution. Interactions with agriculturalists are typically imbalanced, with trade and other exchanges heavily favoring the larger group. One of anthropology’s important roles today is to intelligently and humanely manage equitable interactions between people of different backgrounds and levels of influence.
Special Topic: Indigenous Land Management
Insight into the lives of past modern humans has evolved as researchers revise previous theories and establish new connections with Indigenous knowledge holders.
The outdated view of foraging held that people lived off of the land without leaving an impact on the environment. Accompanying this idea was anthropologist Marshall Sahlins’s (1968) proposal that foragers were the “original affluent society” since they were meeting basic needs and achieving satisfaction with less work hours than agriculturalists and city-dwellers. This view countered an earlier idea that foragers were always on the brink of starvation. Sahlins’s theory took hold in the public eye as an attractive counterpoint to our busy contemporary lives in which we strive to meet our endless wants.
A fruitful type of study involving researchers collaborating with Indigenous experts has found that foragers did not just live off the land with minimal effort nor were they barely surviving in unchanging environments. Instead, they shaped the landscape to their needs using labor and strategies that were more subtle than what European colonizers and subsequent researchers were used to seeing. Research from two regions shows the latest developments in understanding Indigenous land management.
In British Columbia, Canada, the bridging of scientific and Indigenous perspectives has shown that the forests of the region are not untouched wilderness but, rather, have been crafted by Indigenous peoples thousands of years ago. Forest gardens adjacent to archaeological sites show higher plant diversity than unmanaged places even after 150 years (Armstrong et al. 2021). On the coast, 3,500-year-old archaeological sites are evidence of constructed clam gardens, according to Indigenous experts (Lepofsky et al. 2015). Another project, in consultation with Elders of the T’exelc (William Lakes First Nation) in British Columbia, introduced researchers to explanations of how forests were managed before the practice was disrupted by European colonialism (Copes-Gerbitz et al. 2021). Careful management of controlled fires reduced the density of the forest to favor plants such as raspberries and allow easier movement through the landscape.
Similarly, the study of landscapes in Australia, in consultation with Aboriginal Australians today, shows that areas previously considered wilderness by scientists were actually the result of controlling fauna and fires. The presence of grasslands with adjacent forests were purposely constructed to attract kangaroos for hunting (Gammage 2008). People also managed other animal and insect life, from emus to caterpillars. In Tasmania, a shift from productive grassland to wildfire-prone rainforest occurred after Aboriginal Australian land management was replaced by British colonial rule (Fletcher, Hall, and Alexander 2021). The site of Budj Bim of the Gunditjmara people has archaeological features of aquaculture, or the farming of fish, that date back 6,600 years (McNiven et al. 2012; McNiven et al. 2015). These examples show that Indigenous knowledge of how to manipulate the environment may be invaluable at the state level, such as by creating an Aboriginal ranger program to guide modern land management.
The Future of Humanity
A common question stemming from understanding human evolution is: What will the genetic and biological traits of our species be hundreds of thousands of years in the future? When faced with this question, people tend to think of directional selection. Maybe our braincases will be even larger, resembling the large-headed and small-bodied aliens of science fiction (Figure 13.26). Or, our hands could be specialized for interacting with our touch-based technology with less risk of repetitive injury. These ideas do not stand up to scrutiny. Since natural selection is based on adaptations that increase reproductive success, any directional change must be due to a higher rate of producing successful offspring compared to other alleles. Larger brains and more agile fingers would be convenient to possess, but they do not translate into an increase in the underlying allele frequencies.

Scientists are hesitant to professionally speculate on the unknowable, and we will never know what is in store for our species one thousand or one million years from now, but there are two trends in human evolution that may carry on into the future: increased genetic variation and a reduction in regional differences.
Rather than a directional change, genetic variation in our species could expand. Our technology can protect us from extreme environments and pathogens, even if our biological traits are not tuned to handle these stressors. The rapid pace of technological advancement means that biological adaptations will become less and less relevant to reproductive success, so nonbeneficial genetic traits will be more likely to remain in the gene pool. Biological anthropologist Jay T. Stock (2008) views environmental stress as needing to defeat two layers of protection before affecting our genetics. The first layer is our cultural adaptations. Our technology and knowledge can reduce pressure on one’s genotype to be “just right” to pass to the next generation. The second defense is our flexible physiology, such as our acclimatory responses. Only stressors not handled by these powerful responses would then cause natural selection on our alleles. These shields are already substantial, and cultural adaptations will only keep increasing in strength.
The increasing ability to travel far from one’s home region means that there will be a mixing of genetic variation on a global level in the future of our species. In recent centuries, gene flow of people around the world has increased, creating admixture in populations that had been separated for tens of thousands of years. For skin color, this means that populations all around the world could exhibit the whole range of skin colors, rather than the current pattern of decreasing melanin pigment farther from the equator. The same trend of intermixing would apply to all other traits, such as blood types. While our genetics will become more varied, the variation will be more intermixed instead of regionally isolated.
Our distant descendants will not likely be dextrous ultraintellectuals; more likely, they will be a highly variable and mobile species supported by novel cultural adaptations that make up for any inherited biological limitations. Technology may even enable the editing of DNA directly, changing these trends. With the uncertainty of our future, these are just the best-educated guesses for now. Our future is open and will be shaped little by little by the environment, our actions, and the actions of our descendants.
Summary
Modern Homo sapiens is the species that took the hominin lifestyle the furthest to become the only living member of that lineage. The largest factor that allowed us to persist while other hominins went extinct was likely our advanced ability to culturally adapt to a wide variety of environments. Our species, with its skeletal and behavioral traits, was well-suited to be generalist-specialists who successfully foraged across most of the world’s environments. The biological basis of this adaptation was our reorganized brain that facilitated innovation in cultural adaptations and intelligence for leveraging our social ties and finding ways to acquire resources from the environment. As the brain’s ability increased, it shaped the skull by reducing the evolutionary pressure to have large teeth and robust cranial bones to produce the modern Homo sapiens face.
Our ability to be generalist-specialists is seen in the geographical range that modern Homo sapiens covered in 300,000 years. In Africa, our species formed from multiregional gene flow that loosely connected archaic humans across the continent. People then expanded out to the rest of the continental Eurasia and even further to the Americas.
For most of our species’s existence, foraging was the general subsistence strategy within which people specialized to culturally adapt to their local environment. With omnivorousness and mobility, people found ways to extract and process resources, shaping the environment in return. When resource uncertainty hit the species, people around the world focused on agriculture to have a firmer control of sustenance. The new strategy shifted human history toward exponential growth and innovation, leading to our high dependence on cultural adaptations today.
While a cohesive image of our species has formed in recent years, there is still much to learn about our past. The work of many driven researchers shows that there are amazing new discoveries made all the time that refine our knowledge of human evolution. Technological innovations such as DNA analysis enable scientists to approach lingering questions from new angles. The answers we get allow us to ask even more insightful questions that will lead us to the next revelation. Like the pink limestone strata at Jebel Irhoud, previous effort has taken us so far and you are now ready to see what the next layer of discovery holds.
Hominin Species Summary
|
Hominin |
Modern Homo sapiens |
|
Dates |
315,000 years ago to present |
|
Region(s) |
Starting in Africa, then expanding around the world |
|
Famous discoveries |
Cro-Magnon individuals, discovered 1868 in Dordogne, France. Otzi the Ice Man, discovered 1991 in the Alps between Austria and Italy. Kennewick man, discovered 1996 in Washington state. |
|
Brain size |
1400 cc average |
|
Dentition |
Extremely small with short cusps. |
|
Cranial features |
An extremely globular brain case and gracile features throughout the cranium. The mandibular symphysis forms a chin at the anterior-most point. |
|
Postcranial features |
Gracile skeleton adapted for efficient bipedal locomotion at the expense of the muscular strength of most other large primates. |
|
Culture |
Extremely extensive and varied culture with many spoken and written languages. Art is ubiquitous. Technology is broad in complexity and impact on the environment. |
|
Other |
The only living hominin. Chimpanzees and bonobos are the closest living relatives. |
Review Questions
- What are the skeletal and behavioral traits that define modern Homo sapiens? What are the evolutionary explanations for its presence?
- What are some creative ways that researchers have learned about the past by studying fossils and artifacts?
- How do the discoveries mentioned in “First Africa, Then the World” fit the Assimilation model?
- What is foraging? What adaptations do we have for this subsistence strategy? Could you train to be a skilled forager?
- What are aspects of your life that come from dependence on agriculture and its cultural effects? Where did the ingredients of your favorite foods originate from?
Key Terms
African multiregionalism: The idea that modern Homo sapiens evolved as a complex web of small regional populations with sporadic gene flow among them.
Agriculture: The mass production of resources through farming and domestication.
Aquaculture: The farming of fish using techniques such as trapping, channels, and artificial ponds.
Assimilation hypothesis: Current theory of modern human origins stating that the species evolved first in Africa and interbred with archaic humans of Europe and Asia.
Atlatl: A handheld spear thrower that increased the force of thrown projectiles.
Band: A small group of people living together as foragers.
Beringia: Ancient landmass that connected Siberia and Alaska. The ancestors of Indigenous Americans would have crossed this area to reach the Americas.
Carrying capacity: The amount of organisms that an environment can reliably support.
Coastal Route model: Theory that the first Paleoindians crossed to the Americas by following the southern coast of Beringia.
Early Modern Homo sapiens, Early Anatomically Modern Human: Terms used to refer to transitional fossils between archaic and modern Homo sapiens that have a mosaic of traits. Humans like ourselves, who mostly lack archaic traits, are referred to as Late Modern Homo sapiens and simply Anatomically Modern Humans.
Egalitarian: Human organization without strict ranks. Foraging societies tend to be more egalitarian than those based on other subsistence strategies.
Foraging: Lifestyle consisting of frequent movement through the landscape and acquiring resources with minimal storage capacity.
Generalist-specialist niche: The ability to survive in a variety of environments by developing local expertise. Evolution toward this niche may have been what allowed modern Homo sapiens to expand past the geographical range of other human species.
Globalization: A recent increase in the interconnectedness and interdependence of people that is facilitated with long-distance networks.
Globular: Having a rounded appearance. Increased globularity of the braincase is a trait of modern Homo sapiens.
Gracile: Having a smooth and slender quality; the opposite of robust.
Holocene: The epoch of the Cenozoic Era starting around 12,000 years ago and lasting arguably through the present.
Ice-Free Corridor model: Theory that the first Native Americans crossed to the Americas through a passage between glaciers.
Institutions: Long-lasting and influential cultural constructs. Examples include government, organized religion, academia, and the economy.
Last Glacial Maximum: The time 23,000 years ago when the most recent ice age was the most intense.
Later Stone Age: Time period following the Middle Stone Age with a diversification in tool types, starting around 50,000 years ago.
Levant: The eastern coast of the Mediterranean. The site of early modern human expansion from Africa and later one of the centers of agriculture.
Megafauna: Large ancient animals that may have been hunted to extinction by people around the world.
Mental eminence: The chin on the mandible of modern H. sapiens. One of the defining traits of our species.
Microlith: Small stone tool found in the Later Stone Age; also called a bladelet.
Middle Stone Age: Time period known for Mousterian lithics that connects African archaic to modern Homo sapiens.
Monumental architecture: Large and labor-intensive constructions that signify the power of the elite in a sedentary society. A common type is the pyramid, a raised crafted structure topped with a point or platform.
Mosaic: Composed from a mix or composite of traits.
Neolithic Revolution: Time of rapid change to human cultures due to the invention of agriculture, starting around 12,000 years ago.
Ochre: Iron-based mineral pigment that can be a variety of yellows, reds, and browns. Used by modern human cultures worldwide since at least 80,000 years ago.
Sahul: Ancient landmass connecting New Guinea and Australia.
Sedentarism: Lifestyle based on having a stable home area; the opposite of nomadism.
Southern Dispersal model: Theory that modern H. sapiens expanded from East Africa by crossing the Red Sea and following the coast east across Asia.
Subsistence strategy: The method an organism uses to find nourishment and other resources.
Sunda: Ancient Asian landmass that incorporated modern Southeast Asia.
Supraorbital torus: The bony brow ridge across the top of the eye orbits on many hominin crania.
Upper Paleolithic: Time period considered synonymous with the Later Stone Age.
Urbanization: The increase of population density as people settled together in cities.
Wallacea: Archipelago southeast of Sunda with different biodiversity than Asia.
Younger Dryas: The rapid change in global climate—notably a cooling of the Northern Hemisphere—13,000 years ago.
For Further Exploration
Websites
First-person virtual tour of Lascaux cave with annotated cave art: Ministère de la Culture and Musée d’Archéologie Nationale. “Visit the cave” Lascaux website.
Online anthropology magazine articles related to paleoanthropology and human evolution: SAPIENS. “Evolution.” SAPIENS website.
Various presentations of information about hominin evolution: Smithsonian Institution. “What does it mean to be human?” Smithsonian National Museum of Natural History website.
Magazine-style articles on archaeology and paleoanthropology: ThoughtCo. “Archaeology.” ThoughtCo. Website.
Database of comparisons across hominins and primates: University of California, San Diego. “MOCA Domains.” Center for Academic Research & Training in Anthropogeny website.
Books
Engaging book that covers human-made changes to the environment with industrialization and globalization: Kolbert, Elizabeth. 2014. The Sixth Extinction: An Unnatural History. New York: Bloomsbury.
Overview of what human life was like among the environmental shifts of the Ice Age: Woodward, Jamie. 2014. The Ice Age: A Very Short Introduction. Oxford: OUP Press.
Articles
Recent review paper about the current state of paleoanthropology research: Stringer, C. 2016. “The Origin and Evolution of Homo sapiens.” Philosophical Transactions of the Royal Society B 371 (1698).
Overview of the history of American paleoanthropology and the many debates that have occurred over the years: Trinkaus, E. 2018. “One Hundred Years of Paleoanthropology: An American Perspective.” American Journal of Physical Anthropology 165 (4): 638–651.
Amazing magazine article that synthesizes hominin evolution and why it is important to study this subject: Wheelwright, Jeff. 2015. “Days of Dysevolution.” Discover 36 (4): 33–39.
Fascinating research on Ötzi, a mummy from 5,000 years ago: Wierer, Ursula, Simona Arrighi, Stefano Bertola, Günther Kaufmann, Benno Baumgarten, Annaluisa Pedrotti, Patrizia Pernter, and Jacques Pelegrin. 2018. “The Iceman’s Lithic Toolkit: Raw Material, Technology, Typology and Use.” PLOS One 13 (6): e0198292. https://doi.org/10.1371/journal.pone.0198292.
Documentaries
PBS NOVA series covering the expansion of modern Homo sapiens and interbreeding with archaic humans: Brown, Nicholas, dir. 2015. First Peoples. Edmonton: Wall to Wall Television. Amazon Prime Video.
PBS NOVA special featuring the footprints found in White Sands National Park: Falk, Bella, dir. 2016. Ice Age Footprints. Boston: Windfall Films. https://www.pbs.org/wgbh/nova/video/ice-age-footprints/.
PBS NOVA special about how modern humans evolved adaptations to different environments. Shows how present-day people live around the world: Thompson, Niobe, dir. 2016. Great Human Odyssey. Edmonton: Clearwater Documentary. https://www.pbs.org/wgbh/nova/evolution/great-human-odyssey.html.
References
Araujo, Bernardo B. A., Luiz Gustavo R. Oliveira-Santos, Matheus S. Lima-Ribeiro, José Alexandre F. Diniz-Filho, and Fernando A. S. Fernandez. 2017. “Bigger Kill Than Chill: The Uneven Roles of Humans and Climate on Late Quaternary Megafaunal Extinctions.” Quaternary International 431: 216–222.
Armelagos, George J., Peter J. Brown, and Bethany Turner. 2005. “Evolutionary, Historical, and Political Economic Perspectives on Health and Disease.” Social Science & Medicine 61 (4): 755–765.
Armstrong, C. G., J. E. D. Miller, A. C. McAlvay, P. M. Ritchie, and D. Lepofsky. 2021. “Historical Indigenous Land-Use Explains Plant Functional Trait Diversity. Ecology and Society 26 (2): 6.
Bar-Yosef Mayer, Daniella E., Bernard Vandermeersch, and Ofer Bar-Yosef. 2009. “Shells and Ochre in Middle Paleolithic Qafzeh Cave, Israel: Indications for Modern Behavior.” Journal of Human Evolution 56 (3): 307–314.
Barbetti, M., and H. Allen. 1972. “Prehistoric Man at Lake Mungo, Australia, by 32,000 Years Bp.” Nature 240 (5375): 46–48.
Bennett, M. R., D. Bustos, J. S. Pigati, K. B. Springer, T. M. Urban, V. T. Holliday, Sally C. Reynolds, et al. (2021). “Evidence of Humans in North America during the Last Glacial Maximum.” Science 373 (6562): 1528–1531.
Bowler, J. M., Rhys Jones, Harry Allen, and A. G. Thorne. 1970. “Pleistocene Human Remains from Australia: A Living Site and Human Cremation from Lake Mungo, Western New South Wales.” World Archaeology 2 (1): 39–60.
Brown, Peter. 1999. “The First Modern East Asians? Another Look at Upper Cave 101, Liujiang and Minatogawa 1.” In Interdisciplinary Perspectives on the Origins of the Japanese, edited by K. Omoto, 105–131. Kyoto: International Research Center for Japanese Studies.
Brown, Peter. 2000. “Australian Pleistocene Variation and the Sex of Lake Mungo 3.” Journal of Human Evolution 38 (5): 743–749.
Clarkson, Chris, Zenobia Jacobs, Ben Marwick, Richard Fullagar, Lynley Wallis, Mike Smith, Richard G. Roberts, et al. 2017. “Human Occupation of Northern Australia by 65,000 Years Ago.” Nature 547 (7663): 306–310.
Cohen, Mark Nathan. 1977. The Food Crisis in Prehistory: Overpopulation and the Origins of Agriculture. New Haven, CT: Yale University Press.
Cohen, Mark Nathan, and George J. Armelagos, eds. 1984. Paleopathology at the Origins of Agriculture. Orlando, FL: Academic Press.
Cohen, Mark Nathan, and Gillian M. M. Crane-Kramer, eds. 2007. Ancient Health: Skeletal Indicators of Agricultural and Economic Intensification. Gainesville, FL: University Press of Florida.
Copes-Gerbitz, K., S. Hagerman, and L. Daniels. 2021. “Situating Indigenous Knowledge for Resilience in Fire-Dependent Social-Ecological Systems.” Ecology and Society 26(4): 25. https://www.ecologyandsociety.org/vol26/iss4/art25/.
Coqueugniot, Hélène, Olivier Dutour, Baruch Arensburg, Henri Duday, Bernard Vandermeersch, and Anne-Marie Tillier. 2014. “Earliest Cranio-Encephalic Trauma from the Levantine Middle Palaeolithic: 3-D Reappraisal of the Qafzeh 11 Skull, Consequences of Pediatric Brain Damage on Individual Life Condition and Social Care.” PLOS ONE 9 (7): e102822.
Crittenden, Alyssa N., and Stephanie L. Schnorr. 2017. “Current Views on Hunter‐Gatherer Nutrition and the Evolution of the Human Diet.” American Journal of Physical Anthropology 162 (S63): 84–109.
d’Errico, Francesco, Lucinda Backwell, Paola Villa, Ilaria Degano, Jeannette J. Lucejko, Marion K. Bamford, Thomas F. G. Higham, Maria Perla Colombini, and Peter B. Beaumont. 2012. “Early Evidence of San Material Culture Represented by Organic Artifacts from Border Cave, South Africa.” Proceedings of the National Academy of Sciences 109 (33): 13214–13219.
d’Errico, Francesco, Christopher Henshilwood, Marian Vanhaeren, and Karen Van Niekerk. 2005. “Nassarius Kraussianus Shell Beads from Blombos Cave: Evidence for Symbolic Behaviour in the Middle Stone Age.” Journal of Human Evolution 48 (1): 3–24.
Dannemann, Michael, and Fernando Racimo. 2018. “Something Old, Something Borrowed: Admixture and Adaptation in Human Evolution.” Current Opinion in Genetics & Development 53: 1–8.
Day, M. H. 1969. “Omo Human Skeletal Remains.” Nature 222: 1135–1138.
Dillehay, Tom D., Carlos Ocampo, José Saavedra, Andre Oliveira Sawakuchi, Rodrigo M. Vega, Mario Pino, Michael B. Collins, et al. 2015. “New Archaeological Evidence for an Early Human Presence at Monte Verde, Chile.” PLOS ONE 10 (11): e0141923. doi:10.1371/journal.pone.0141923.
Dow, Gregory K., Clyde G. Reed, and Nancy Olewiler. 2009. “Climate Reversals and the Transition to Agriculture.” Journal of Economic Growth 14 (1): 27–53.
Durband, Arthur C. 2014. “Brief Communication: Artificial Cranial Modification in Kow Swamp and Cohuna.” American Journal of Physical Anthropology 155 (1): 173–178.
Ember, Carol R. N.d. “Hunter-Gatherers.” Explaining Human Culture. Human Relations Area Files. Accessed March 4, 2023. https://hraf.yale.edu/ehc/summaries/hunter-gatherers.
Erlandson, Jon M., Todd J. Braje, Kristina M. Gill, and Michael H. Graham. 2015. “Ecology of the Kelp Highway: Did Marine Resources Facilitate Human Dispersal from Northeast Asia to the Americas?” The Journal of Island and Coastal Archaeology 10 (3): 392–411.
Fladmark, K. R. 1979. “Routes: Alternate Migration Corridors for Early Man in North America.” American Antiquity 44 (1): 55–69.
Fletcher, M. S., T. Hall, and A. N. Alexandra. 2021. “The Loss of an Indigenous Constructed Landscape Following British Invasion of Australia: An Insight into the Deep Human Imprint on the Australian Landscape.” Ambio 50(1): 138–149.
Fu, Qiaomei, Mateja Hajdinjak, Oana Teodora Moldovan, Silviu Constantin, Swapan Mallick, Pontus Skoglund, Nick Patterson, et al. 2015. “An Early Modern Human from Romania with a Recent Neanderthal Ancestor.” Nature 524 (7564): 216–219.
Fuller, Dorian Q. 2010. “An Emerging Paradigm Shift in the Origins of Agriculture.” General Anthropology 17 (2): 1, 8–11.
Gammage, B. 2008. “Plain Facts: Tasmania under Aboriginal Management.” Landscape Research 33 (2): 241–254.
Germonpré, Mietje, Martina Lázničková-Galetová, and Mikhail V. Sablin. 2012. “Palaeolithic Dog Skulls at the Gravettian Předmostí Site, the Czech Republic.” Journal of Archaeological Science 39 (1): 184–202.
Gröning, Flora, Jia Liu, Michael J. Fagan, and Paul O’Higgins. 2011. “Why Do Humans Have Chins? Testing the Mechanical Significance of Modern Human Symphyseal Morphology with Finite Element Analysis.” American Journal of Physical Anthropology 144 (4): 593–606.
Harvati, Katerina. 2009. “Into Eurasia: A Geometric Morphometric Reassessment of the Upper Cave (Zhoukoudian) Specimens.” Journal of Human Evolution 57 (6): 751–762.
Headland, Thomas N., Lawrence A. Reid, M. G. Bicchieri, Charles A. Bishop, Robert Blust, Nicholas E. Flanders, Peter M. Gardner, Karl L. Hutterer, Arkadiusz Marciniak, and Robert F. Schroeder. 1989. “Hunter-Gatherers and Their Neighbors from Prehistory to the Present.” Current Anthropology 30 (1): 43–66.
Henshilwood, Christopher S., Francesco d’Errico, Karen L. van Niekerk, Yvan Coquinot, Zenobia Jacobs, Stein-Erik Lauritzen, Michel Menu, and Renata García-Moreno. 2011. “A 100,000-Year-Old Ochre-Processing Workshop at Blombos Cave, South Africa.” Science 334 (6053): 219–222.
Hershkovitz, Israel, Gerhard W. Weber, Rolf Quam, Mathieu Duval, Rainer Grün, Leslie Kinsley, Avner Ayalon, et al. 2018. “The Earliest Modern Humans Outside Africa.” Science 359 (6374): 456–459.
Hublin, Jean-Jacques, Abdelouahed Ben-Ncer, Shara E. Bailey, Sarah E. Freidline, Simon Neubauer, Matthew M. Skinner, Inga Bergmann, et al. 2017. “New Fossils from Jebel Irhoud, Morocco, and the Pan-African Origin of Homo sapiens.” Nature 546 (7657): 289–292.
Lepofsky, D., N. F. Smith, N. Cardinal, J. Harper, M. Morris, M., Gitla (Elroy White), Randy Bouchard, et al. 2015. “Ancient Shellfish Mariculture on the Northwest Coast of North America.” American Antiquity 80 (2): 236–259.
Lieberman, Daniel E. 2015. “Human Locomotion and Heat Loss: An Evolutionary Perspective.” Comprehensive Physiology 5 (1): 99–117.
Lieberman, Daniel E., Brandeis M. McBratney, and Gail Krovitz. 2002. “The Evolution and Development of Cranial Form in Homo sapiens.” Proceedings of the National Academy of Sciences 99 (3): 1134–1139.
Lieberman, Daniel E., Osbjorn M. Pearson, and Kenneth M. Mowbray. 2000. “Basicranial Influence on Overall Cranial Shape.” Journal of Human Evolution 38 (2): 291–315.
Liu, Wu, María Martinón-Torres, Yan-jun Cai, Song Xing, Hao-wen Tong, Shu-wen Pei, Mark Jan Sier, Xiao-hong Wu, R. Lawrence Edwards, and Hai Cheng. 2015. “The Earliest Unequivocally Modern Humans in Southern China.” Nature 526 (7575): 696-699.
Lucas, Peter W. 2007. “The Evolution of the Hominin Diet from a Dental Functional Perspective.” In Evolution of the Human Diet: The Known, the Unknown, and the Unknowable, edited by Peter S. Ungar, 31–38 Oxford, UK: Oxford University Press.
McCarthy, Robert C., and Lynn Lucas. 2014. “A Morphometric Reassessment of Bou-Vp-16/1 from Herto, Ethiopia.” Journal of Human Evolution 74: 114–117.
McDougall, Ian, Francis H. Brown, and John G. Fleagle. 2005. “Stratigraphic Placement and Age of Modern Humans from Kibish, Ethiopia.” Nature 433 (7027): 733–736.
McNiven, I. J., J. Crouch, T. Richards, N. Dolby, and G. Jacobsen. 2012. “Dating Aboriginal Stone-Walled Fishtraps at Lake Condah, Southeast Australia.” Journal of Archaeological Science 39 (2): 268–286.
McNiven, I., J. Crouch, T. Richards, K. Sniderman, N. Dolby, and G. Mirring. 2015. “Phased Redevelopment of an Ancient Gunditjmara Fish Trap over the Past 800 Years: Muldoons Trap Complex, Lake Condah, Southwestern Victoria.” Australian Archaeology 81 (1): 44–58.
Michel, Véronique, Hélène Valladas, Guanjun Shen, Wei Wang, Jian-xin Zhao, Chuan-Chou Shen, Patricia Valensi, and Christopher J. Bae. 2016. “The Earliest Modern Homo sapiens in China?” Journal of Human Evolution 101: 101–104.
Miller, D. Shane, Vance T. Holliday, and Jordon Bright. 2013. “Clovis across the Continent.” In Paleoamerican Odyssey, edited by Kelly E. Graf, Caroline V. Ketron, and Michael R. Waters, 207–220. College Station: Texas A&M University Press.
Neubauer, Simon, Jean-Jacques Hublin, and Philipp Gunz. 2018. “The Evolution of Modern Human Brain Shape.” Science Advances 4 (1): eaao5961. https://doi.org/10.1126/sciadv.aao5961.
Pearson, Osbjorn M. 2000. “Postcranial Remains and the Origin of Modern Humans.” Evolutionary Anthropology 9: 229–247.
Pearson, Osbjorn M. 2008. “Statistical and Biological Definitions of ‘Anatomically Modern’ Humans: Suggestions for a Unified Approach to Modern Morphology.” Evolutionary Anthropology: Issues, News, and Reviews 17 (1): 38–48.
Pietschnig, Jakob, Lars Penke, Jelte M. Wicherts, Michael Zeiler, and Martin Voracek. 2015. “Meta-Analysis of Associations between Human Brain Volume and Intelligence Differences: How Strong Are They and What Do They Mean?” Neuroscience & Biobehavioral Reviews 57: 411–432.
Posth, Cosimo, Nathan Nakatsuka, Iosif Lazaridis, Pontus Skoglund, Swapan Mallick, Thiseas C. Lamnidis, Nadin Rohland, et al. 2018. “Reconstructing the Deep Population History of Central and South America.” Cell 175 (5): 1185–1197.
Potter, Ben A., James F. Baichtal, Alwynne B. Beaudoin, Lars Fehren-Schmitz, C. Vance Haynes, Vance T. Holliday, Charles E. Holmes, et al. 2018. “Current Evidence Allows Multiple Models for the Peopling of the Americas.” Science Advances 4 (8): eaat5473. https://doi.org/10.1126/sciadv.aat5473.
Reich, David, Richard E. Green, Martin Kircher, Johannes Krause, Nick Patterson, Eric Y. Durand, Bence Viola, et al. 2010. “Genetic History of an Archaic Hominin Group from Denisova Cave in Siberia.” Nature 468 (7327): 1053–1060.
Reich, David, Nick Patterson, Martin Kircher, Frederick Delfin, Madhusudan R. Nandineni, Irina Pugach, Albert Min-Shan Ko, et al. 2011. “Denisova Admixture and the First Modern Human Dispersals into Southeast Asia and Oceania.” American Journal of Human Genetics 89 (4): 516–528.
Richter, Daniel, Rainer Grün, Renaud Joannes-Boyau, Teresa E. Steele, Fethi Amani, Mathieu Rué, Paul Fernandes, et al. 2017. “The Age of the Hominin Fossils from Jebel Irhoud, Morocco, and the Origins of the Middle Stone Age.” Nature 546 (7657): 293–296.
Roberts, Patrick, and Brian A. Stewart. 2018. “Defining the ‘Generalist-Specialist’ Niche for Pleistocene Homo sapiens.” Nature Human Behaviour 2: 542–550.
Rougier, Helene, Ştefan Milota, Ricardo Rodrigo, Mircea Gherase, Laurenţiu Sarcinǎ, Oana Moldovan, João Zilhão, et al. 2007. “Peştera Cu Oase 2 and the Cranial Morphology of Early Modern Europeans.” Proceedings of the National Academy of Sciences 104 (4): 1165–1170.
Sahlins, Marshall. 1968. “Notes on the Original Affluent Society.” In Man the Hunter, edited by R. B. Lee and I. DeVore, 85–89. New York: Aldine Publishing Company.
Sawyer, G. J., and Blaine Maley. 2005. “Neanderthal Reconstructed.” The Anatomical Record (Part B: New Anat.) 283 (1): 23–31.
Scerri, Eleanor M. L., Mark G. Thomas, Andrea Manica, Philipp Gunz, Jay T. Stock, Chris Stringer, Matt Grove, et al. 2018. “Did Our Species Evolve in Subdivided Populations Across Africa, and Why Does It Matter?” Trends in Ecology & Evolution 33 (8): 582–594.
Shea, John J. 2011. “Refuting a Myth about Human Origins.” American Scientist 99 (2): 128–135.
Shea, John J., and Ofer Bar-Yosef. 2005. “Who Were the Skhul/Qafzeh People? An Archaeological Perspective on Eurasia’s Oldest Modern Humans.” Journal of the Israel Prehistoric Society 35: 451–468.
Slatkin, Montgomery, and Fernando Racimo. 2016. “Ancient DNA and Human History.” Proceedings of the National Academy of Sciences 113 (23): 6380–6387.
Smith, Fred H., James C. M. Ahern, Ivor Janković, and Ivor Karavanić. 2017. “The Assimilation Model of Modern Human Origins in Light of Current Genetic and Genomic Knowledge.” Quaternary International 450: 126–136.
Smith, Michael. 2009. “V. Gordon Childe and the Urban Revolution: A Historical Perspective on a Revolution in Urban Studies.” Town Planning Review 80 (1): 3–29.
Stock, Jay T. 2008. “Are Humans Still Evolving?” EMBO Reports 9 (Suppl 1): S51–S54.
Swisher, Mark E., Dennis L. Jenkins, Lionel E. Jackson Jr., and Fred M. Phillips. 2013. “A Reassessment of the Role of the Canadian Ice-Free Corridor in Light of New Geological Evidence.” Poster Symposium 5B: Geology, Geochronology and Paleoenvironments of the First Americans at the Paleoamerican Odyssey Conference, Santa Fe, New Mexico, October 16–19.
Thorne, A. G., and P. G. Macumber. 1972. “Discoveries of Late Pleistocene Man at Kow Swamp, Australia.” Nature 238 (5363): 316–319.
Trinkaus, Erik, Ştefan Milota, Ricardo Rodrigo, Gherase Mircea, and Oana Moldovan. 2003a. “Early Modern Human Cranial Remains from the Peştera Cu Oase, Romania.” Journal of Human Evolution 45 (3): 245–253.
Trinkaus, Erik, Oana Moldovan, Adrian Bîlgăr, Laurenţiu Sarcina, Sheela Athreya, Shara E Bailey, Ricardo Rodrigo, Gherase Mircea, Thomas Higham, and Christopher Bronk Ramsey. 2003b. “An Early Modern Human from the Peştera Cu Oase, Romania.” Proceedings of the National Academy of Sciences 100 (20): 11231–11236.
Velemínská, J., J. Brůzek, P. Velemínský, L. Bigoni, A. Sefcáková, and S. Katina. 2008. “Variability of the Upper-Palaeolithic Skulls from Predmostí Near Prerov (Czech Republic): Craniometric Comparison with Recent Human Standards.” Homo 59 (1): 1–26.
Vidal, Céline M., Christine S. Lane, Asfawossen Asrat, Dan N. Barfod, Darren F. Mark, Emma L. Tomlinson, Ambdemichael Zafu Tadesse, et al. (2022). “Age of the Oldest Known Homo sapiens from Eastern Africa. Nature 601 (7894): 579–583.
Villa, Paola, Sylvain Soriano, Tsenka Tsanova, Ilaria Degano, Thomas F. G. Higham, Francesco d’Errico, Lucinda Backwell, Jeannette J. Lucejko, Maria Perla Colombini, and Peter B. Beaumont. 2012. “Border Cave and the Beginning of the Later Stone Age in South Africa.” Proceedings of the National Academy of Sciences 109 (33): 13208–13213.
Wall, Jeffrey D., and Deborah Yoshihara Caldeira Brandt. 2016. “Archaic Admixture in Human History.” Current Opinion in Genetics & Development 41: 93–97.
White, Tim D., Berhane Asfaw, David DeGusta, Henry Gilbert, Gary D. Richards, Gen Suwa, and F. Clark Howell. 2003. “Pleistocene Homo sapiens from Middle Awash, Ethiopia.” Nature 423 (6941): 742–747.
Woo, Ju-Kang. 1959. “Human Fossils Found in Liukiang, Kwangsi, China.” Vertebrata PalAsiatica 3 (3): 109–118.
Wu, XiuJie, Wu Liu, Wei Dong, JieMin Que, and YanFang Wang. 2008. “The Brain Morphology of Homo Liujiang Cranium Fossil by Three-Dimensional Computed Tomography.” Chinese Science Bulletin 53 (16): 2513–2519.
Acknowledgments
I could not have undertaken this project without the help of many who got me to where I am today. I extend sincere thank yous to the many colleagues and former students who have inspired me to keep learning and talking about anthropology. Thank you also to all who are involved in this textbook project. The anonymous reviewers truly sparked improvements to the chapter. Lastly, the staff of Starbucks #5772 also contributed immensely to this text.
Hayley Mann, M.A., Binghamton University
Student contributors for this chapter: Emma Costa, Shima Gahima, Will Lefebvre, Audrey Chékinaël
This chapter is a revision from "Chapter 3: Molecular Biology and Genetics" by Hayley Mann, Xazmin Lowman, and Malaina Gaddis. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Explain and identify the purpose of both DNA replication and the cell cycle.
- Identify key differences between mitosis and meiosis.
- Outline the process of protein synthesis, including transcription and translation.
- Use principles of Mendelian inheritance to predict genotypes and phenotypes of future generations.
- Explain complexities surrounding patterns of genetic inheritance and polygenic traits.
- Discuss challenges to and bioethical concerns of genetic testing.
I [Hayley Mann] started my Bachelor’s degree in 2003, which was the same year the Human Genome Project released its first draft sequence. I initially declared a genetics major because I thought it sounded cool. However, upon taking an actual class, I discovered that genetics was challenging. In addition to my genetics major, I signed up for biological anthropology classes and soon learned that anthropology could bring all those molecular lessons to life. For instance, we are composed of cells, proteins, nucleic acids, carbohydrates, and lipids. Anthropologists often include these molecules in their studies to identify how humans vary; if there are meaningful differences, they propose theories to explain them. Anthropologists study biomolecules in both living and ancient individuals. Ancient biomolecules can also be found on artifacts such as stone tools and cooking vessels. Over the years, scientific techniques for studying organic molecules have improved, which has unlocked new insights into the deep human past.
Cells and Molecules
Molecules of Life
All organisms are composed of four basic types of molecules that are essential for cell structure and function: proteins, lipids, carbohydrates, and nucleic acids (Figure 4.1). Proteins are crucial for cell shape and nearly all cellular tasks, including receiving signals from outside the cell and mobilizing intra-cellular responses. Lipids are a class of organic compounds that include fats, oils, and hormones. Carbohydrates are sugar molecules and serve as energy to cells in the form of glucose. Lastly, nucleic acids, including deoxyribonucleic acid (DNA), carry genetic information about a living organism.
|
Molecule |
Definition |
Example |
|
Proteins |
Composed of one or more long chains of amino acids (i.e., basic units of protein) Often folded into complex 3D shapes that relate to function Proteins interact with other types of proteins and molecules |
Proteins come in different categories including structural (e.g., collagen, keratin, lactase, hemoglobin, cell membrane proteins), defense proteins (e.g, antibodies), enzymes (e.g., lactase), hormones (e.g., insulin), and motor proteins (e.g., actin) |
|
Lipids |
Insoluble in water due to hydrophilic (water-loving) head and a hydrophobic (water-repelling) tail |
Fats, such as triglycerides, store energy for your body Steroid hormones (e.g., estrogen and testosterone) act as chemical messengers to communicate between cells and tissues, as well as biochemical pathways inside of the cell |
|
Carbohydrates |
Large group of organic molecules that are composed of carbon and hydrogen atoms |
Starches and sugars, including blood glucose, provide cells with energy |
|
Nucleic Acids |
Carries the genetic information of an organism |
DNA RNA |
Cells
In 1665, Robert Hooke observed slices of plant cork using a microscope. Hooke noted that the microscopic plant structures he saw resembled cella, meaning “a small room” in Latin. Approximately two centuries later, biologists recognized the cell as being the most fundamental unit of life and that all life is composed of cells. Cellular organisms can be characterized as two main cell types: prokaryotes and eukaryotes (Figure 4.2).
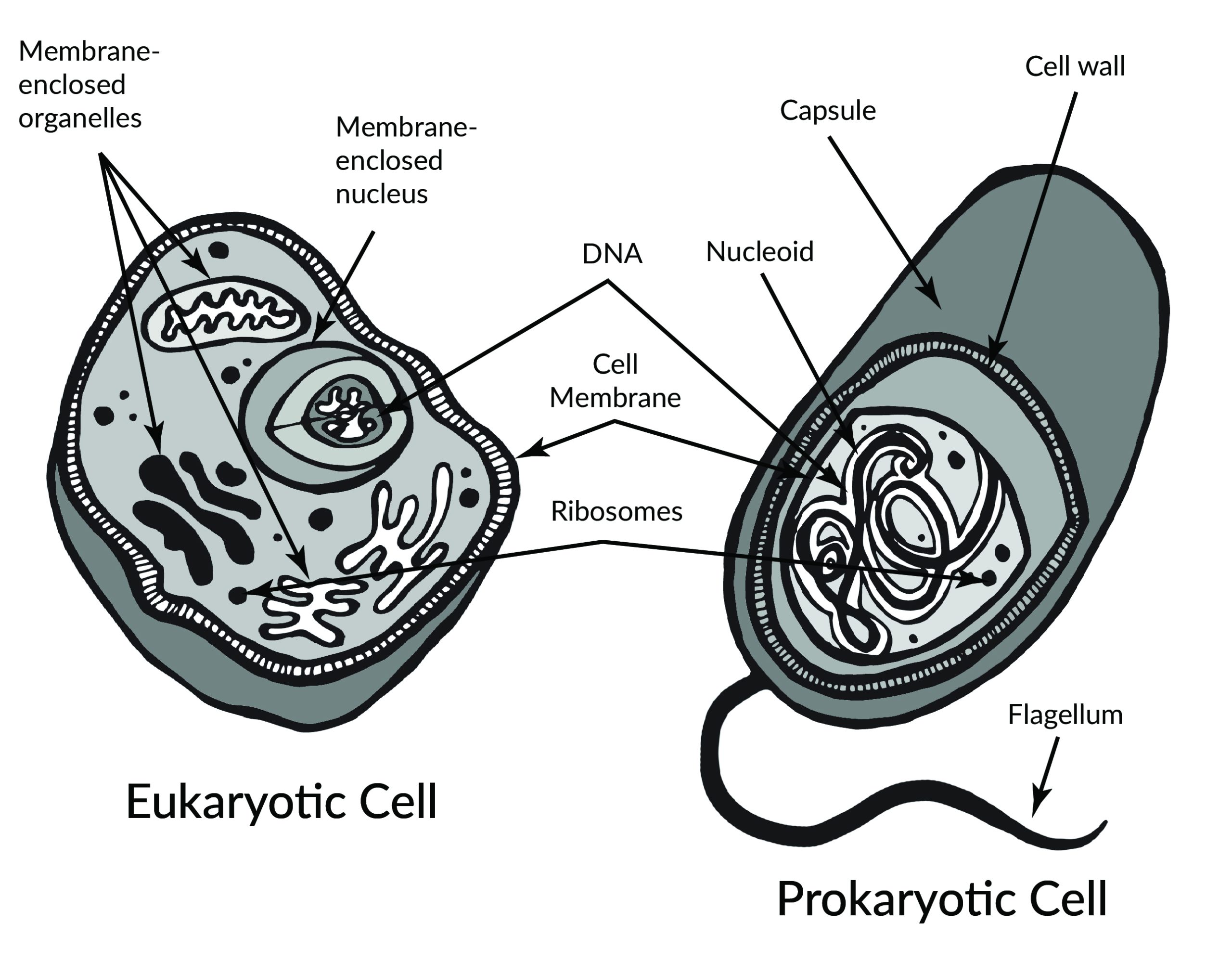
Prokaryotes include bacteria and archaea, and they are composed of a single cell. Additionally, their DNA and organelles are not surrounded by individual membranes. Thus, no compartments separate their DNA from the rest of the cell (see Figure 4.2). It is well known that some bacteria can cause illness in humans. For instance, Escherichia coli (E. coli) and Salmonella contamination can result in food poisoning symptoms. Pneumonia and strep throat are caused by Streptococcal bacteria. Neisseria gonorrhoeae is a sexually transmitted bacterial disease. Although bacteria are commonly associated with illness, not all bacteria are harmful. For example, researchers are studying the relationship between the microbiome and human health. The bacteria that are part of the healthy human microbiome perform beneficial roles, such as digesting food, boosting the immune system, and even making vitamins (e.g., B12 and K).
Eukaryotes can be single-celled or multi-celled in their body composition. In contrast to prokaryotes, eukaryotes possess membranes that surround their DNA and organelles. An example of a single-celled eukaryote is the microscopic algae found in ponds (phytoplankton), which can produce oxygen from the sun. Yeasts are also single-celled, and fungi can be single- or multicellular. Plants and animals are all multicellular.
Although plant and animal cells have a surprising number of similarities, there are some key differences (Figure 4.3). For example, plant cells possess a thick outer cell membrane made of a fibrous carbohydrate called cellulose. Animal and plant cells also have different tissues. For most plants, the outermost layer of cells forms a waxy cuticle that helps to protect the cells and to prevent water loss. Humans have skin, which is the outermost cell layer that is predominantly composed of a tough protein called keratin. Overall, humans have a diversity of tissue types (e.g., cartilage, brain, and heart).
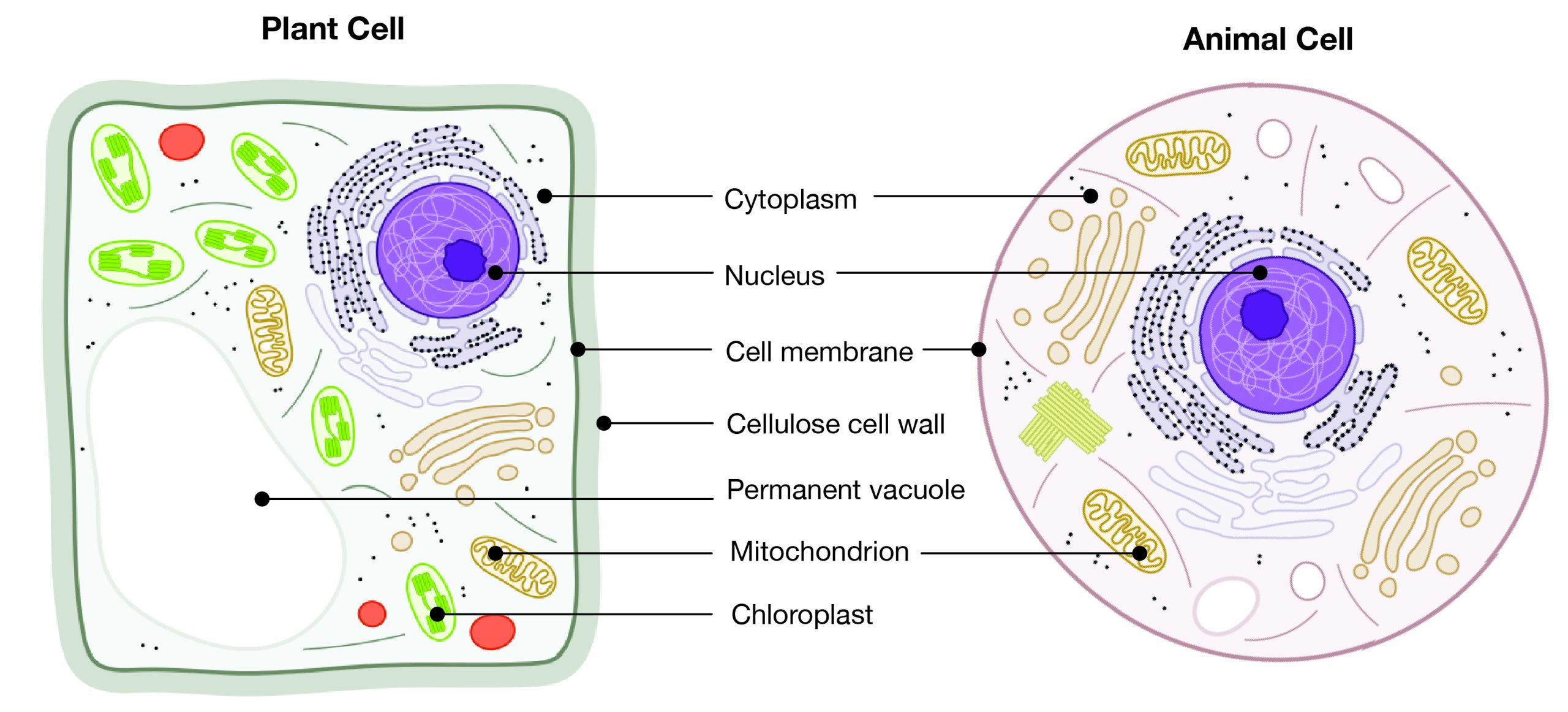
Animal Cell Organelles
An animal cell is surrounded by a double membrane called the phospholipid bilayer (Figure 4.4). A closer look reveals that this protective barrier is made of lipids and proteins that provide structure and function for cellular activities, such as regulating the passage of molecules and ions (e.g., H2O and sodium) into and out of the cell. Cytoplasm is the jelly-like matrix inside of the cell membrane. Part of the cytoplasm comprises organelles, which perform different specialized tasks for the cell (Figure 4.5). An example of an organelle is the nucleus, where the cell’s DNA is located.
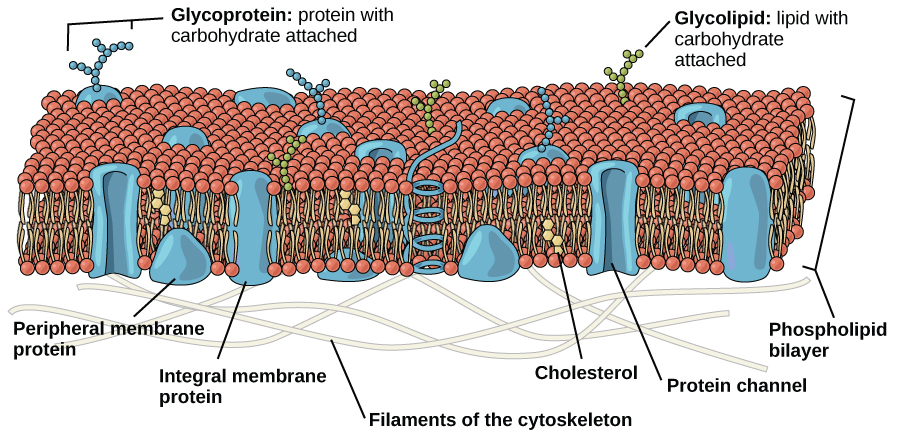
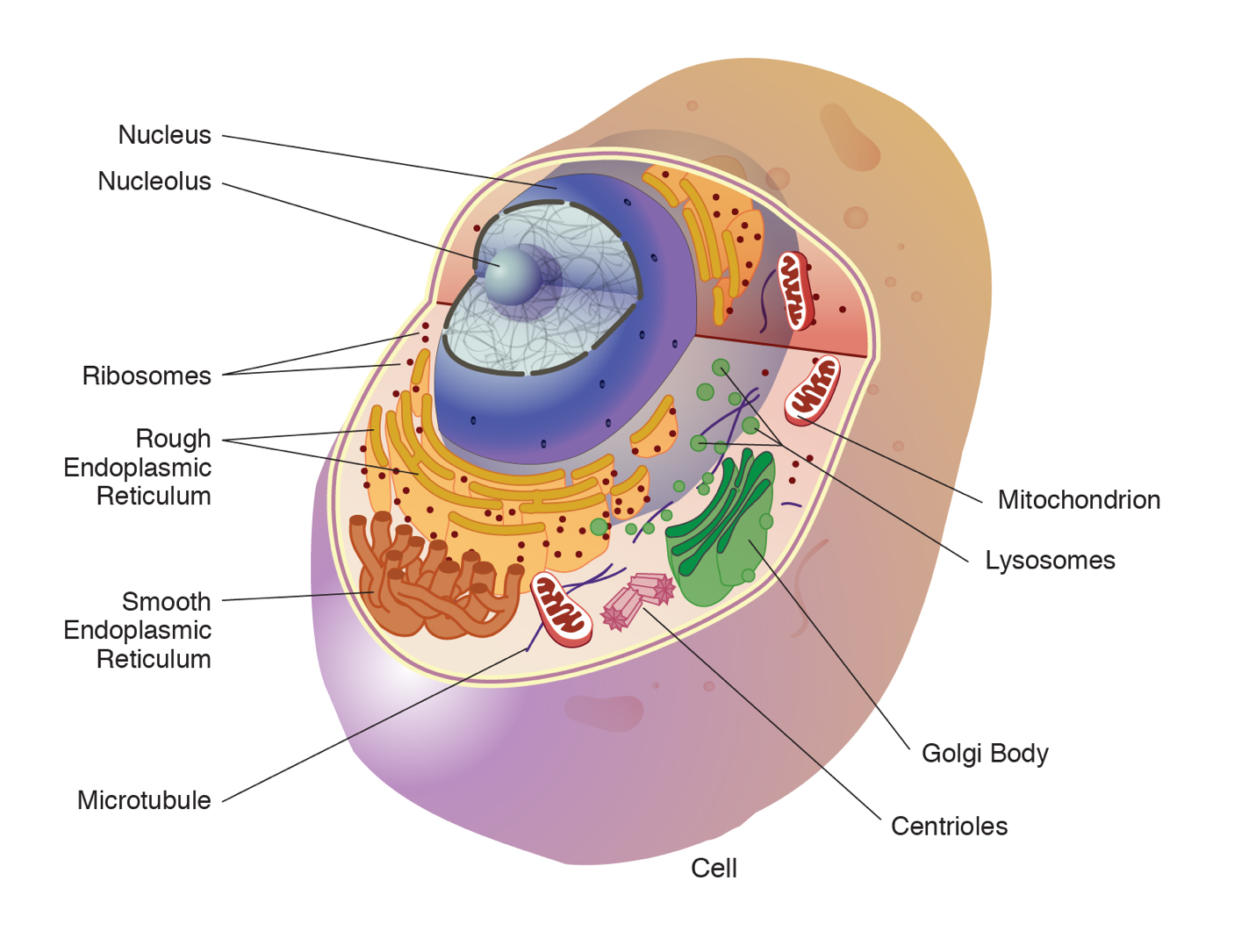
Another organelle is the mitochondrion. Mitochondria are often referred to as “powerhouse centers” because they produce energy for the cell in the form of adenosine triphosphate (ATP). Depending on the species and tissue type, multicellular eukaryotes can have hundreds to thousands of mitochondria in each of their cells. Scientists have determined that mitochondria were once symbiotic prokaryotic organisms (i.e., helpful bacteria) that transformed into cellular organelles over time. This evolutionary explanation helps explain why mitochondria also have their own DNA, called mitochondrial DNA (mtDNA). All organelles have important physiological functions and disease can occur when organelles do not perform their role optimally. Figure 4.6 lists other organelles found in the cell and their specialized cellular roles.
|
Cell structure |
Description |
|
Centrioles |
Assist with the organization of mitotic spindles, which extend and contract for the purpose of cellular movement during mitosis and meiosis. |
|
Cytoplasm |
Gelatinous fluid located inside of cell membrane that contains organelles. |
|
Endoplasmic reticulum (ER) |
Continuous membrane with the nucleus that helps transport, synthesize, modify, and fold proteins. Rough ER has embedded ribosomes, whereas smooth ER lacks ribosomes. |
|
Golgi body |
Layers of flattened sacs that receive and transmit messages from the ER to secrete and transport proteins within the cell. |
|
Lysosome |
Located in the cytoplasm; contains enzymes to degrade cellular components. |
|
Microtubule |
Involved with cellular movement including intracellular transport and cell division. |
|
Mitochondrion |
Responsible for cellular respiration, where energy is produced by converting nutrients into ATP. |
|
Nucleolus |
Resides inside of the nucleus and is the site of ribosomal RNA (rRNA) transcription, processing, and assembly. |
|
Nucleopore |
Pores in the nuclear envelope that are selectively permeable. |
|
Nucleus |
Contains the cell’s DNA and is surrounded by the nuclear envelope. |
|
Ribosome |
Located in the cytoplasm and also the membrane of the rough endoplasmic reticulum. Messenger RNA (mRNA) binds to ribosomes and proteins are synthesized. |
Introduction to Genetics
Genetics is the study of heredity. Biological parents pass down their genetic traits to their offspring. Although children resemble their parents, genetic traits often vary in appearance or molecular function. For example, two parents with normal color vision can sometimes produce a son with red-green colorblindness. Molecular geneticists study the biological mechanisms responsible for creating variation between individuals, such as DNA mutations (see Chapter 4), cell division, and genetic regulation.
Molecular anthropologists use genetic data to test anthropological questions. Some of these anthropologists utilize ancient DNA (aDNA), which is DNA that is extracted from anything once living, including human, animal, and plant remains. Over time, DNA becomes degraded (i.e., less intact), but specialized laboratory techniques can make copies of short degraded aDNA segments, which can then be reassembled to provide more complete DNA information.
DNA Structure
The discovery, in 1953, of the molecular structure of deoxyribonucleic acid (DNA) was one of the greatest scientific achievements of all time. Using X-ray crystallography, Rosalind Franklin (Figure 4.7) provided an image that clearly showed the double helix shape of DNA. Due to controversy, Franklin’s colleagues received more recognition for the DNA discovery. In 1962, Watson, Crick, and Wilkins won the Nobel Prize, while Franklin, who had died in 1958, was not honoured. Today, her vital contributions and scientific skill are widely recognized.
The double helix shape of DNA can be described as a twisted ladder (Figure 4.8). More specifically, DNA is a double-stranded molecule with its two strands oriented in opposite directions (i.e., antiparallel). Each strand is composed of nucleotides with a sugar phosphate backbone. There are four different types of DNA nucleotides: adenine (A), thymine (T), cytosine (C), and guanine (G). The two DNA strands are held together by nucleotide base pairs, which have chemical bonding rules. The complementary base-pairing rules are as follows: A and T bond with each other, while C and G form a bond. The chemical bonds between A-T and C-G are formed by “weak” hydrogen atom interactions, which means the two strands can be easily separated. A DNA sequence is the order of nucleotide bases (A, T, G, C) along only one DNA strand. If one DNA strand has the sequence CATGCT, then the other strand will have a complementary sequence GTACGA. This is an example of a short DNA sequence. In reality, there are approximately three billion DNA base pairs in human cells.
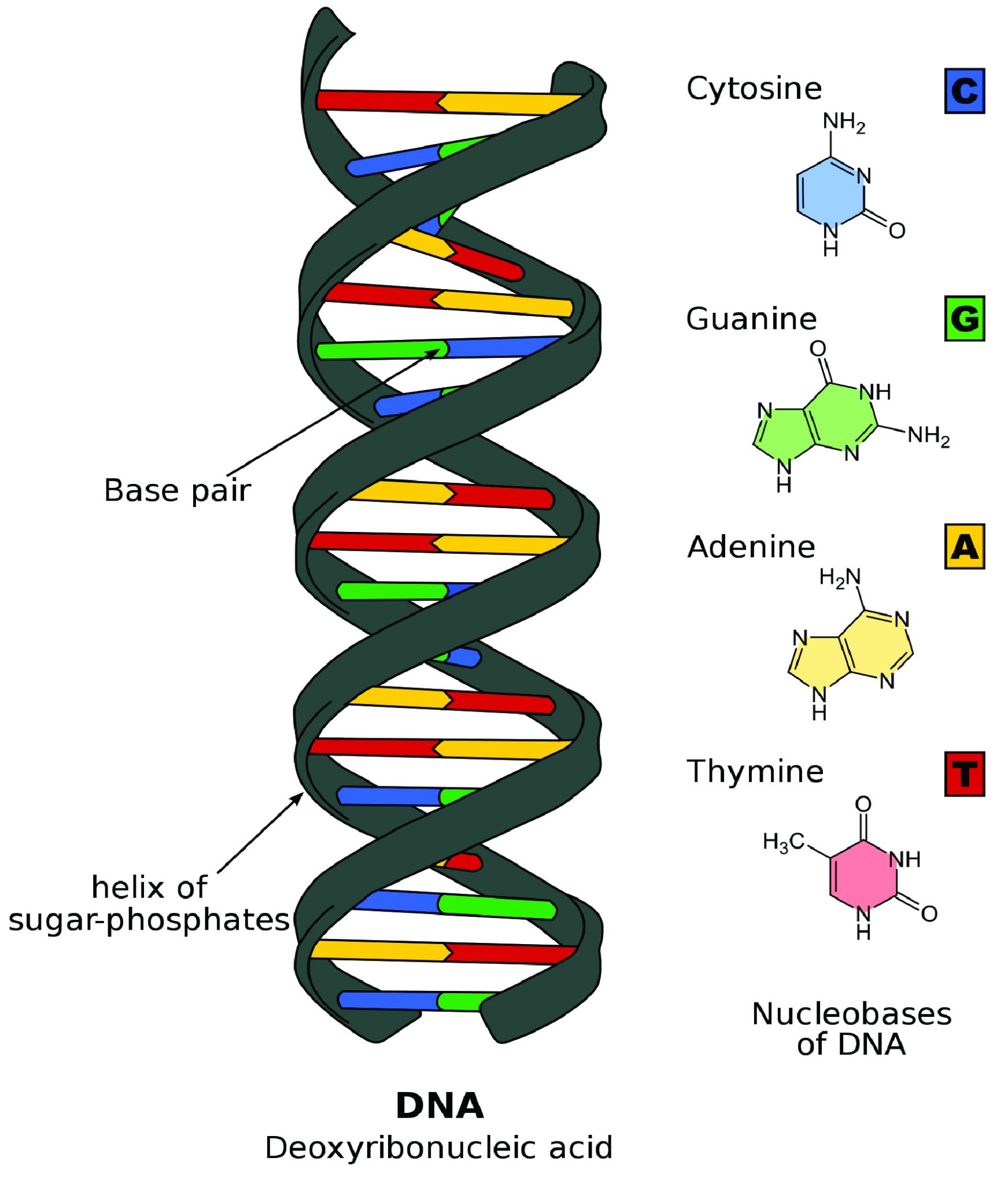
DNA Is Highly Organized within the Nucleus
If you removed the DNA from a single human cell and stretched it out completely, it would measure approximately two meters (about 6.5 feet). Therefore, DNA molecules must be compactly organized in the nucleus. To achieve this, the double helix configuration of DNA undergoes coiling. An analogy would be twisting a string until coils are formed and then continuing to twist so that secondary coils are formed, and so on. To assist with coiling, DNA is first wrapped around proteins called histones. This creates a complex called chromatin, which resembles “beads on a string” (Figure 4.9). Next, chromatin is further coiled into a chromosome. Another important feature of DNA is that chromosomes can be altered from tightly coiled (chromatin) to loosely coiled (euchromatin). Most of the time, chromosomes in the nucleus remain in a euchromatin state so that DNA sequences are accessible for regulatory processes to occur.
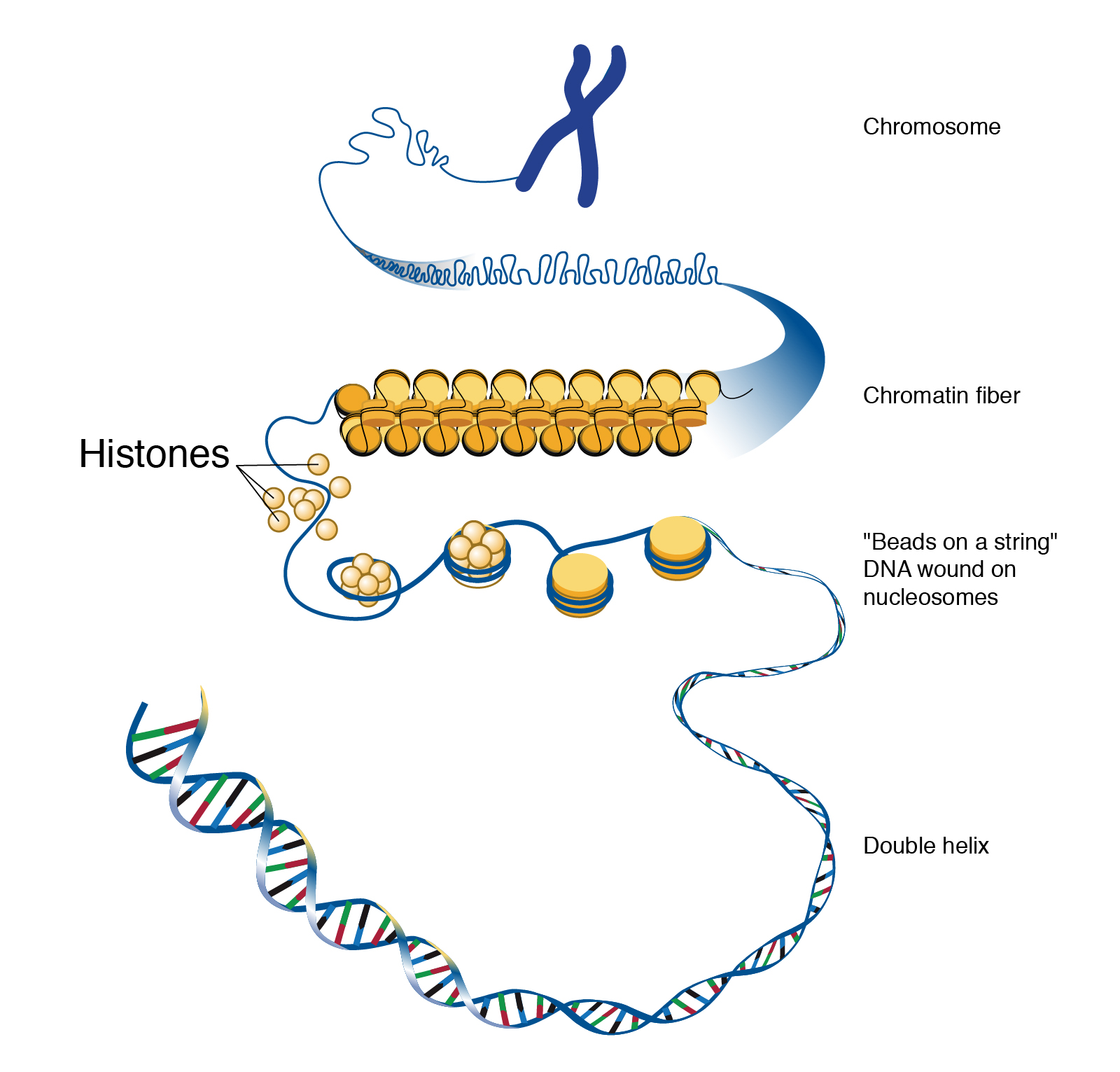
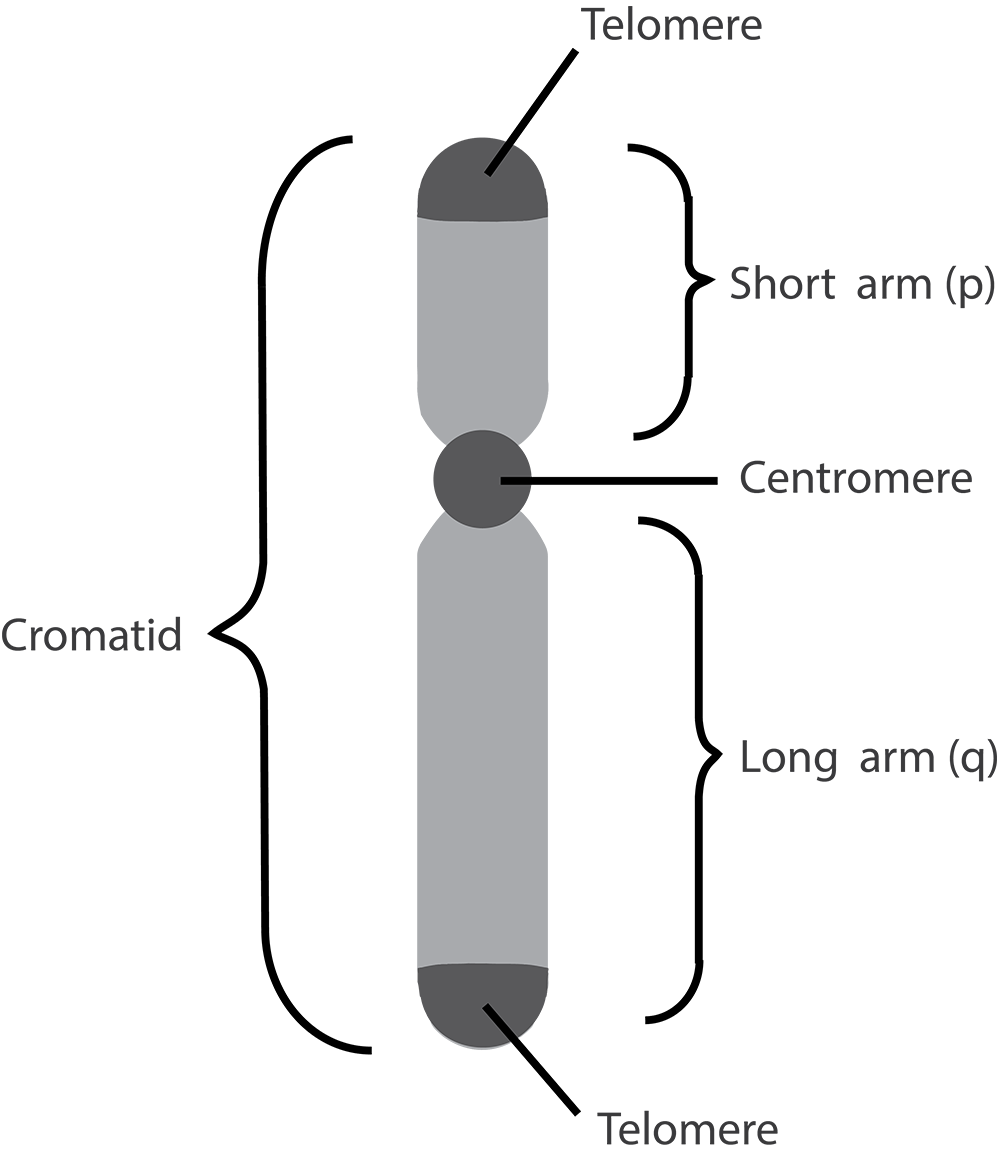
Human body cells typically have 23 pairs of chromosomes, for a total of 46 chromosomes in each cell’s nucleus. An interesting fact is that the number of chromosomes an organism possesses varies by species, and this figure is not dependent upon the size or complexity of the organism. For instance, chimpanzees have a total of 48 chromosomes, while hermit crabs have 254. Chromosomes also have a distinct physical structure, including centromeres (the “center”) and telomeres (the ends) (Figure 4.10). Because of the centromeric region, chromosomes are described as having two different “arms,” where one arm is long and the other is shorter. Centromeres play an important role during cell division, which will be discussed in the next section. Telomeres are located at the ends of chromosomes; they help protect the chromosomes from degradation after every round of cell division.
Special Topic: First Nation Immunity and European Diseases—A Study of Ancient DNA

Beginning in the early fifteenth century, First Nations progressively suffered from high mortality rates as the result of colonization from foreign powers. European-borne diseases such as measles, tuberculosis, influenza, and smallpox are largely responsible for the population collapse of Indigenous peoples in the Americas. Many Europeans who immigrated to the Americas had lived in large sedentary populations, which also included coexisting with domestic animals and pests. Although a few prehistoric Indigenous populations can be characterized as large agricultural societies (especially in Mesoamerica), their overall culture, community lifestyle, and subsistence practices were markedly different from that of Europeans. Therefore, because they did not share the same urban living environments as Europeans, it is believed that Indigenous peoples were susceptible to many European diseases.
In 2016, a Nature article published by John Lindo and colleagues was the first to investigate whether pre-contact Indigenous peoples possessed a genetic susceptibility to European diseases. Their study included Tsimshians, a First Nation community from British Columbia (Figure 4.11a-b). DNA from both present-day and ancient individuals (who lived between 500 and 6,000 years ago) was analyzed. The research team discovered that a change occurred in the HLA-DQA1 gene, which is a member of the major histocompatibility complex (MHC) immune system molecules. MHC molecules are responsible for detecting and triggering an immune response against pathogens. Lindo and colleagues (2016) concluded that HLA-DQA1 gene helped Indigenous peoples adapt to their local environmental ecology. However, when European-borne epidemics occurred in the Northwest during the 1800s, a certain HLA-DQA1 DNA sequence variant (allele) associated with ancient Tsimshian immunity was no longer adaptive. As the result of past selective pressures from European diseases, present-day Tsimshians have different HLA-DQA1 allele frequencies. The precise role that HLA-DQA1 plays in immune adaptation requires further investigation. But overall, this study serves as an example of how studying ancient DNA from the remains of deceased individuals can help provide insight into living human populations and historical events.
DNA Replication and Cell Division
For life to continue and flourish, cells must be able to divide. Tissue growth and cellular damage repair are also necessary to maintain an organism throughout its life. All these rely on the dynamic processes of DNA replication and the cell cycle. The mechanisms highlighted in this section are tightly regulated and represent only part of the life cycle of a cell.
DNA Replication
DNA replication is the process by which new DNA is copied from an original DNA template. It is one phase of the highly coordinated cell cycle, and it requires a variety of enzymes with special functions. The creation of a complementary DNA strand from a template strand is described as semi-conservative replication. The result of semi-conservative replication is two separate double-stranded DNA molecules, each of which is composed of an original “parent” template strand and a newly synthesized “daughter” DNA strand.
DNA replication progresses in three steps referred to as initiation, elongation, and termination. During initiation, enzymes are recruited to specific sites along the DNA sequence (Figure 4.12). For example, an initiator enzyme, called helicase, “unwinds” DNA by breaking the hydrogen bonds between the two parent strands. The unraveling of the helix into two separated strands exposes the strands and creates a fork, which is the active site of DNA replication.
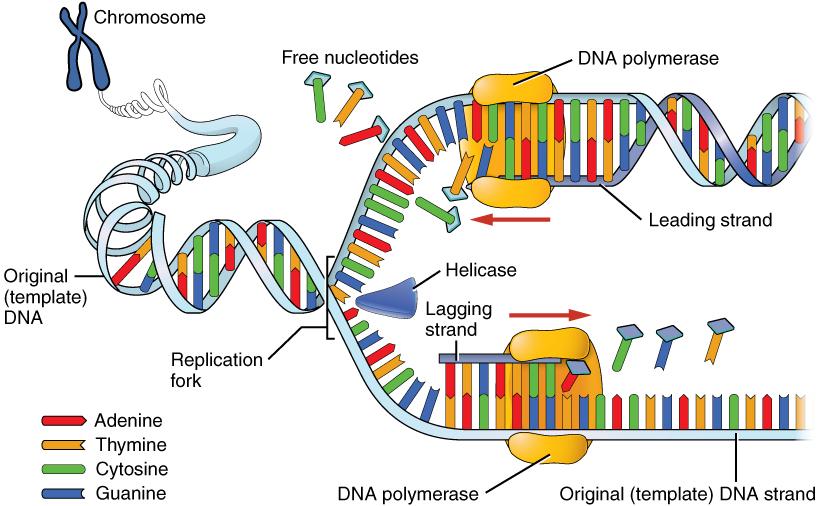
Elongation is the assembly of new DNA daughter strands from the exposed original parent strands. The two parent strands can further be classified as leading strand or lagging strand and are distinguished by the direction of replication. Enzymes called DNA polymerases read parent template strands in a specific direction. Complementary nucleotides are added, and the newly formed daughter strands will grow. On the leading parent strand, a DNA polymerase will create one continuous strand. The lagging parent strand is created in several disconnected sections and other enzymes fill in the missing nucleotide gaps between these sections.
Finally, termination refers to the end of DNA replication activity. It is signaled by a stop sequence in the DNA that is recognized by machinery at the replication fork. The end result of DNA replication is that the number of chromosomes are doubled so that the cell can divide into two.
DNA Mutations
DNA replication should result in the creation of two identical DNA nucleotide sequences. However, although DNA polymerases are quite precise during DNA replication, copying mistakes are estimated to occur every 107 DNA nucleotides. Variation from the original DNA sequence is known as a mutation (Refer to Chapter 4). Briefly, mutations can result in single-nucleotide changes, as well as the insertion or deletion of nucleotides and repeated sequences. Depending on where they occur in the genome, mutations can be deleterious (harmful). For example, mutations may occur in regions that control cell cycle regulation, which can result in cancer (see Special Topic: The Cell Cycle and Immortality of Cancer Cells). Many other types of mutations, however, are not harmful to an organism.
Regardless of their effect, the cell attempts to reduce the frequency of mutations that occur during DNA replication. To accomplish this, there are polymerases with proofreading capacities that can identify and correct mismatched nucleotides. These safeguards reduce the frequency of DNA mutations so that they only occur every 109 nucleotides.
Mitotic Cell Division
There are two types of cells in the body: germ cells (sperm and egg) and somatic cells. The body and its various tissues comprises somatic cells. Organisms that contain two sets of chromosomes in their somatic cells are called diploid organisms. Humans have 46 chromosomes and they are diploid because they inherit one set of chromosomes (n = 23) from each parent. As a result, they have 23 matching pairs of chromosomes, which are known as homologous chromosomes. As seen in Figure 4.13, homologous chromosome pairs vary in size and are generally numbered from largest (chromosome 1) to smallest (chromosome 22) with the exception of the 23rd pair, which is made up of the sex chromosomes (X and Y). Typically, the female sex is XX and the male sex is XY. Individuals inherit an X chromosome from their chromosomal mother and an X or Y from their chromosomal father.
To grow and repair tissues, somatic cells must divide. As discussed previously, for cell division to occur, a cell must first replicate its genetic material. During DNA replication, each chromosome produces double the amount of genetic information. The duplicated arms of chromosomes are known as sister chromatids, and they are attached at the centromeric region. To elaborate, the number of chromosomes stays the same (n = 46); however, the amount of genetic material is doubled in the cell as the result of replication.
Mitosis is the process of somatic cell division that gives rise to two diploid daughter cells (Figure 4.14). Once DNA and other organelles in the cell have finished replication, mitotic spindle fibers physically align each chromosome at the center of the cell. Next, the spindle fibers divide the sister chromatids and move each one to opposite sides of the cell. At this phase, there are 46 chromosomes on each side of a human cell. The cell can now divide into two fully separated daughter cells.
Meiotic Cell Division
Gametogenesis is the production of gametes (sperm and egg cells); it involves two rounds of cell division called meiosis. Similar to mitosis, the parent cell in meiosis is diploid. However, meiosis has a few key differences, including the number of daughter cells produced (four cells, which require two rounds of cell division to produce) and the number of chromosomes each daughter cell has (see Figure 4.14).
During the first round of division (known as meiosis I), each chromosome (n = 46) replicates its DNA so that sister chromatids are formed. Next, with the help of spindle fibers, homologous chromosomes align near the center of the cell and sister chromatids physically swap genetic material. In other words, the sister chromatids of matching chromosomes cross over with each other at matching DNA nucleotide positions. The occurrence of homologous chromosomes crossing over, swapping DNA, and then rejoining segments is called genetic recombination. The “genetic shuffling” that occurs in gametes increases organismal genetic diversity by creating new combinations of genes on chromosomes that are different from the parent cell. Genetic mutations can also arise during recombination. For example, there may be an unequal swapping of genetic material that occurs between the two sister chromatids, which can result in deletions or duplications of DNA nucleotides. Once genetic recombination is complete, homologous chromosomes are separated and two daughter cells are formed.
The daughter cells after the first round of meiosis are haploid, meaning they only have one set of chromosomes (n = 23). During the second round of cell division (known as meiosis II), sister chromatids are separated and two additional haploid daughter cells are formed. Therefore, the four resulting daughter cells have one set of chromosomes (n = 23), and they also have a genetic composition that is not identical to the parent cells nor to each other.
Although both sperm and egg gamete production undergo meiosis, they differ in the final number of viable daughter cells. In the case of spermatogenesis, four mature sperm cells are produced. Although four egg cells are also produced in oogenesis, only one of these egg cells will result in an ovum (mature egg). During fertilization, an egg cell and sperm cell fuse, which creates a diploid cell that develops into an embryo. The ovum also provides the cellular organelles necessary for embryonic cell division. This includes mitochondria, which is why humans, and most other multicellular eukaryotes, have the same mtDNA sequence as their mothers.
Chromosomal Disorders: Aneuploidies
During mitosis or meiosis, entire deletions or duplications of chromosomes can occur due to error. For example, homologous chromosomes may fail to separate properly, so one daughter cell may end up with an extra chromosome while the other daughter cell has one less. Cells with an unexpected (or abnormal) number of chromosomes are known as aneuploid. Adult or embryonic cells can be tested for chromosome number (karyotyping). Aneuploid cells are typically detrimental to a dividing cell or developing embryo, which can lead to a loss of pregnancy. However, the occurrence of individuals being born with three copies of the 21st chromosome is relatively common; this genetic condition is known as Down Syndrome. Moreover, individuals can also be born with aneuploid sex chromosome conditions such as XXY, XXX, and XO (referring to only one X chromosome).
Special Topic: The Cell Cycle and Immortality of Cancer Cells
DNA replication is part of a series of preparatory phases that a cell undergoes prior to cell division, collectively known as interphase (Figure 4.15). During interphase, the cell not only doubles its chromosomes through DNA replication, but it also increases its metabolic capacity to provide energy for growth and division. Transition into each phase of the cell cycle is tightly controlled by proteins that serve as checkpoints. If a cell fails to pass a checkpoint, then DNA replication and/or cell division will not continue. Some of the reasons why a cell may fail at a checkpoint is DNA damage, lack of nutrients to continue the process, or insufficient size. In turn, a cell may undergo apoptosis, which is a mechanism for cell death.
Unchecked cellular growth is a distinguishing hallmark of cancer. In other words, as cancer cells grow and proliferate, they acquire the capacity to avoid death and replicate indefinitely. This uncontrolled and continuous cell division is also known as “immortality.” As previously mentioned, most cells lose the ability to divide due to shortening of telomeres on the ends of chromosomes over time. One way in which cancer cells retain replicative immortality is that the length of their telomeres is continuously protected. Chemotherapy, often used to treat cancer, targets the cell cycle (especially cell division) to halt the propagation of genetically abnormal cells. Another therapeutic approach that continues to be investigated is targeting telomere activity to stop the division of cancer cells.
Researchers have exploited the immortality of cancer cells for molecular research. The oldest immortal cell line is HeLa cells (Figure 4.16), which were harvested from Henrietta Lacks, an African American woman diagnosed with cervical cancer in 1955. At that time, extracted cells frequently died during experiments, but surprisingly HeLa cells continued to replicate. Propagation of Lacks’s cell line has significantly contributed to medical research, including contributing to ongoing cancer research and helping to test the polio vaccine in the 1950s. However, Lacks had not given her consent for her tumor biopsy to be used in cell culture research. Moreover, her family was unaware of the extraction and remarkable application of her cells for two decades. The history of HeLa cell origin was first revealed in 1976. The controversy voiced by the Lacks family was included in an extensive account of HeLa cells published in Rebecca Skloot’s 2010 book, The Immortal Life of Henrietta Lacks. A film based on the book was also released in 2017 (Wolfe 2017).
Protein Synthesis
At the beginning of the chapter, we defined proteins as strings of amino acids that fold into complex 3-D shapes. There are 20 standard amino acids that can be strung together in different combinations in humans, and the result is that proteins can perform an impressive amount of different functions. For instance, muscle fibers are proteins that help facilitate movement. A special class of proteins (immunoglobulins) help protect the organism by detecting disease-causing pathogens in the body. Protein hormones, such as insulin, help regulate physiological activity. Blood hemoglobin is a protein that transports oxygen throughout the body. Enzymes are also proteins, and they are catalysts for biochemical reactions that occur in the cell (e.g., metabolism). Larger-scale protein structures can be visibly seen as physical features of an organism (e.g., hair and nails).
Transcription and Translation
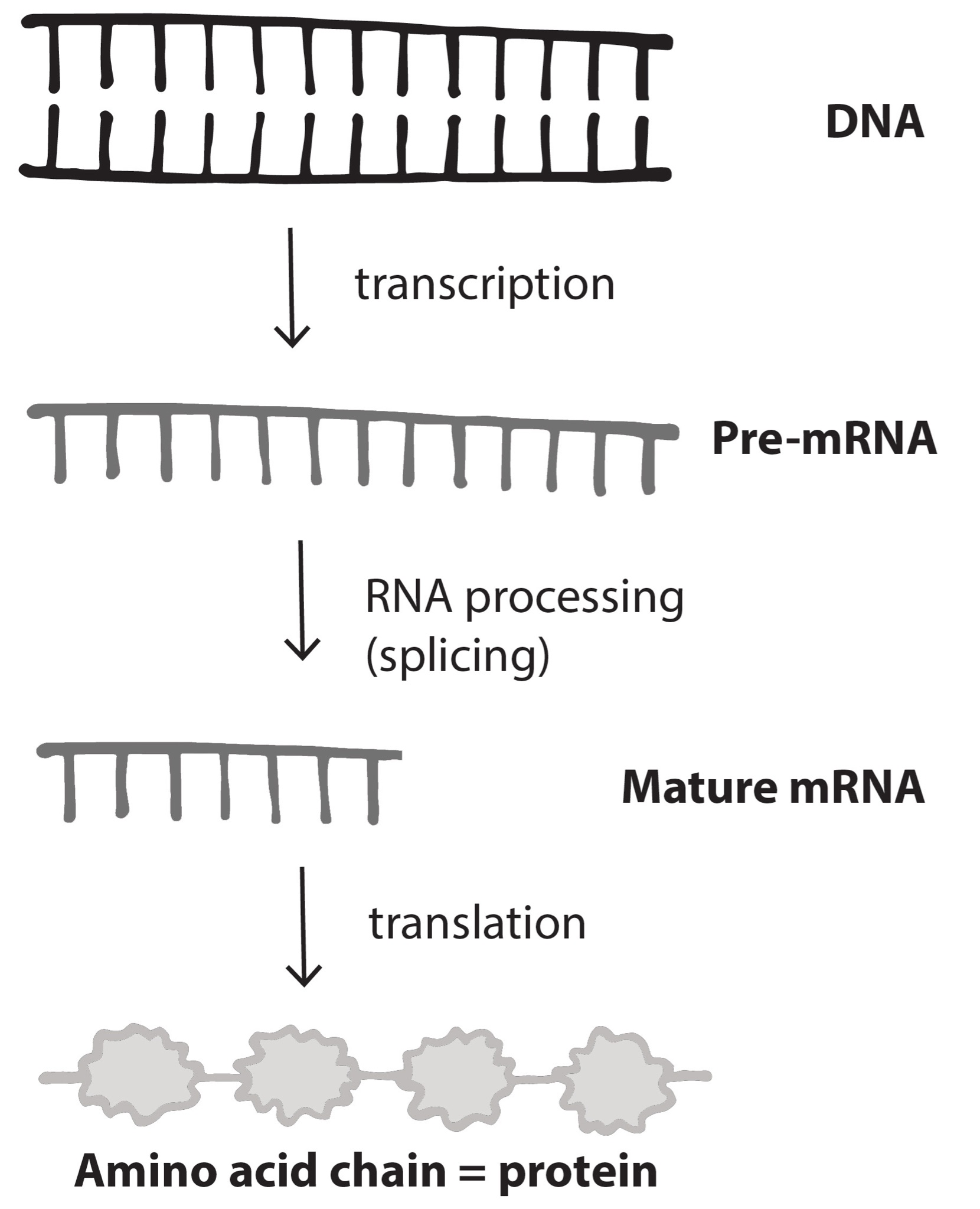
Nucleotides in our DNA provide the coding instructions on how to make proteins. Making proteins, also known as protein synthesis, can be broken down into two main steps referred to as transcription and translation. The purpose of transcription, the first step, is to make an ribonucleic acid (RNA) copy of our genetic code. Although there are many different types of RNA molecules that have a variety of functions within the cell, we will mainly focus on messenger RNA (mRNA). Transcription concludes with the processing (splicing) of the mRNA. The second step, translation, uses mRNA as the instructions for chaining together amino acids into a new protein molecule (Figure 4.17).
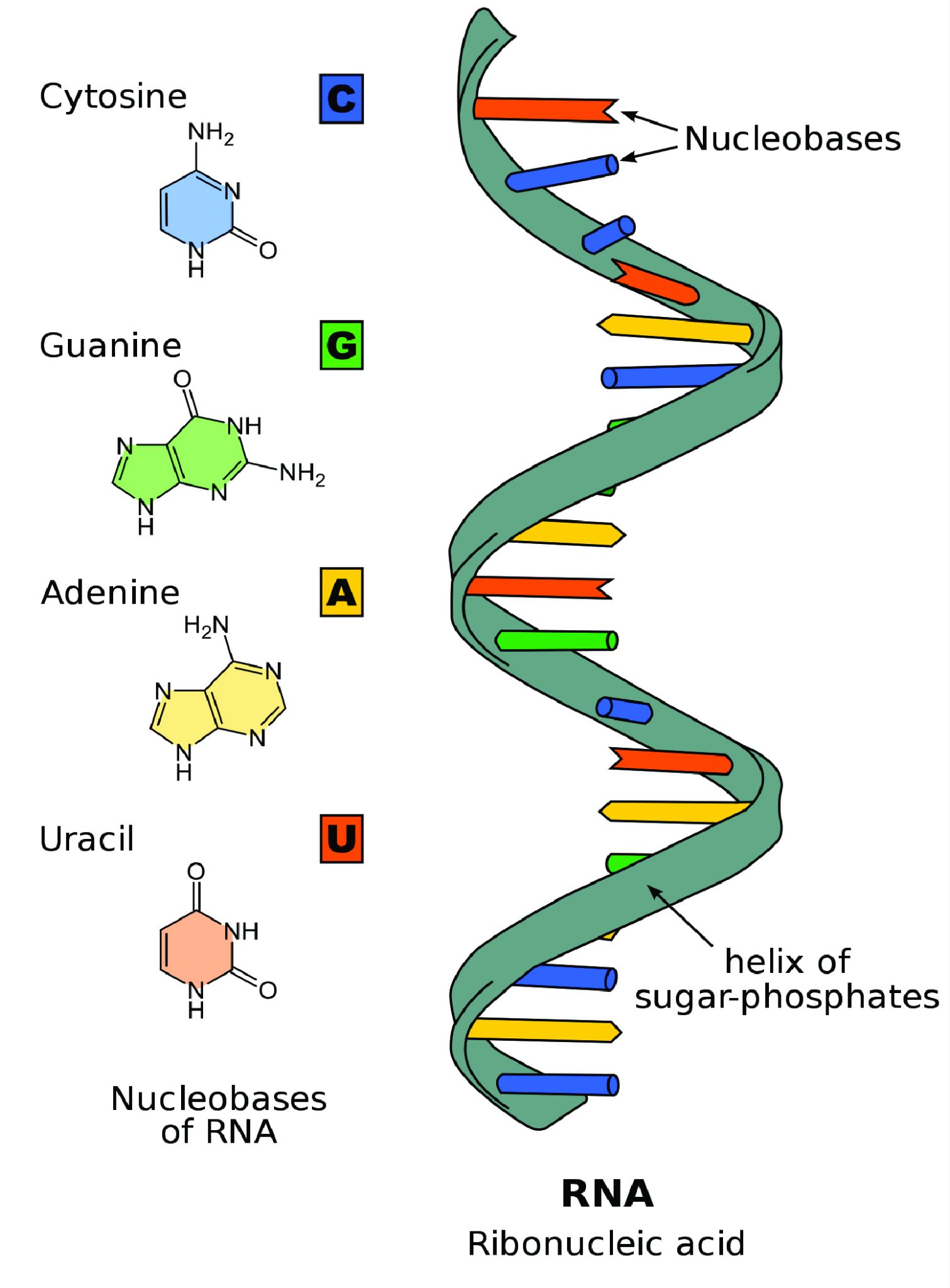
Unlike double-stranded DNA, RNA molecules are single-stranded nucleotide sequences (Figure 4.18). Additionally, while DNA contains the nucleotide thymine (T), RNA does not—instead its fourth nucleotide is uracil (U). Uracil is complementary to (or can pair with) adenine (A), while cytosine (C) and guanine (G) continue to be complementary to each other.
For transcription to proceed, a gene must first be turned “on” by the cell. A gene is a segment of DNA that codes for RNA, and genes can vary in length from a few hundred to as many as two million base pairs in length. The double-stranded DNA is then separated, and one side of the DNA is used as a coding template that is read by RNA polymerase. Next, complementary free-floating RNA nucleotides are linked together (Figure 4.19) to form a single-stranded mRNA. For example, if a DNA template is TACGGATGC, then the newly constructed mRNA sequence will be AUGCCUACG.
Genes contain segments called introns and exons. Exons are considered “coding” while introns are considered “noncoding”—meaning the information they contain will not be needed to construct proteins. When a gene is first transcribed into pre-mRNA, introns and exons are both included (Figure 4.20). However, once transcription is finished, introns are removed in a process called splicing. During splicing, a protein/RNA complex attaches itself to the pre-mRNA. Next, introns are removed and the remaining exons are connected, thus creating a shorter mature mRNA that serves as a template for building proteins.
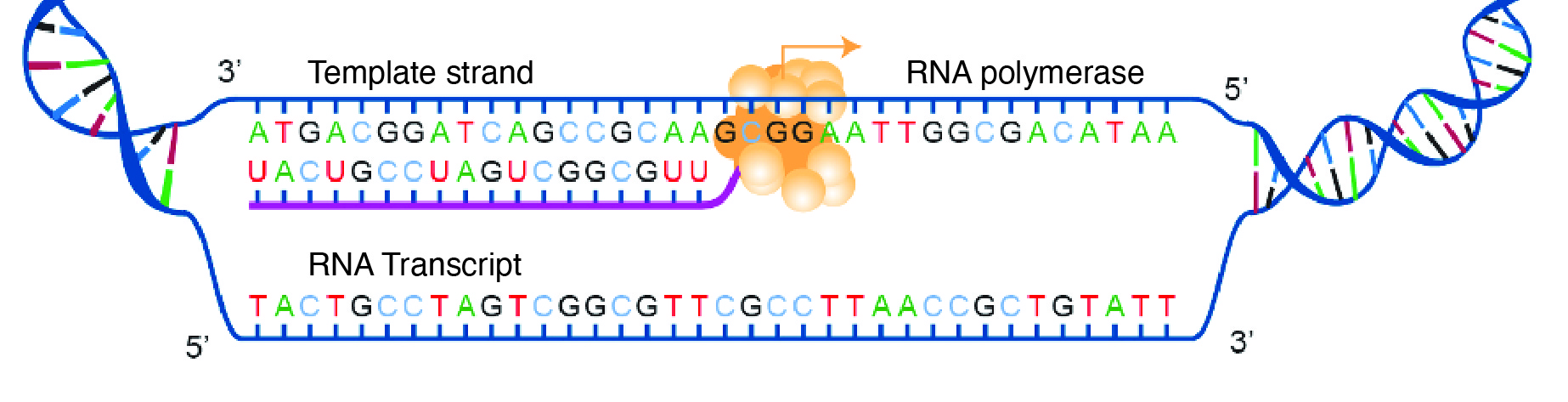
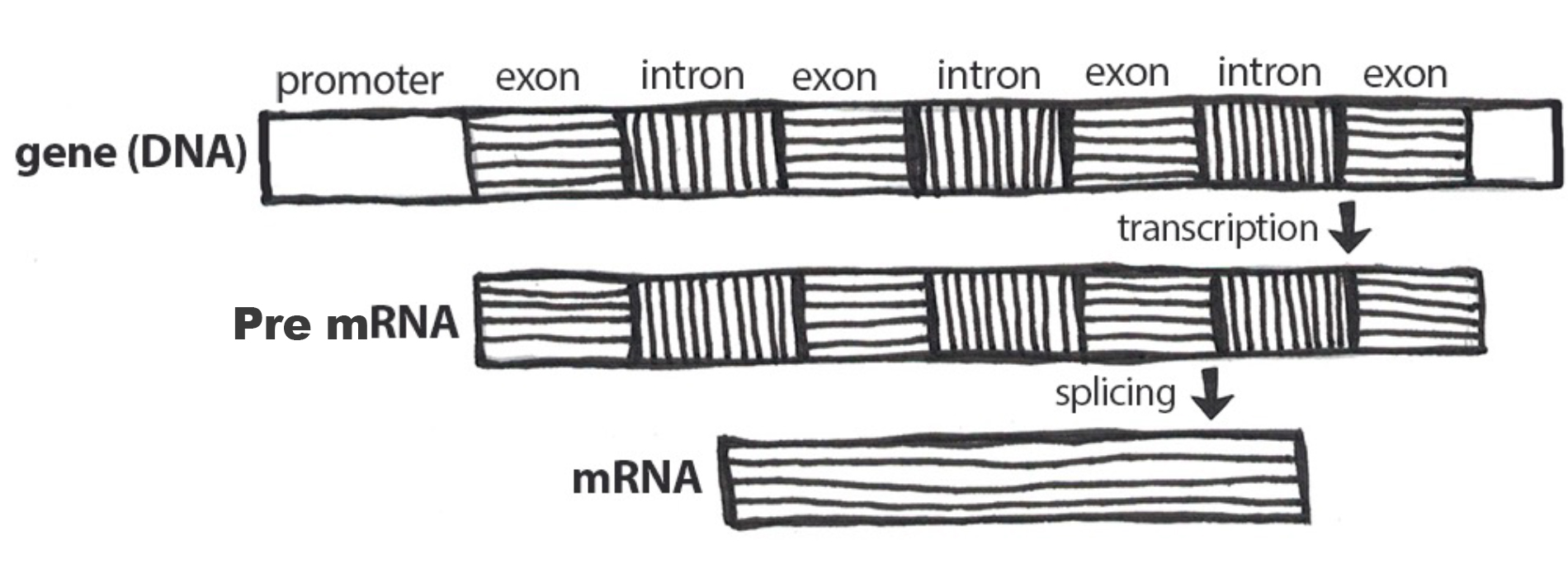
As described above, the result of transcription is a single-stranded mRNA copy of a gene. Translation is the process by which amino acids are chained together to form a new protein. During translation, the mature mRNA is transported outside of the nucleus, where it is bound to a ribosome (Figure 4.21). The nucleotides in the mRNA are read in triplets, which are called codons. Each mRNA codon corresponds to an amino acid, which is carried to the ribosome by a transfer RNA (tRNA). Thus, tRNAs is the link between the mRNA molecule and the growing amino acid chain.
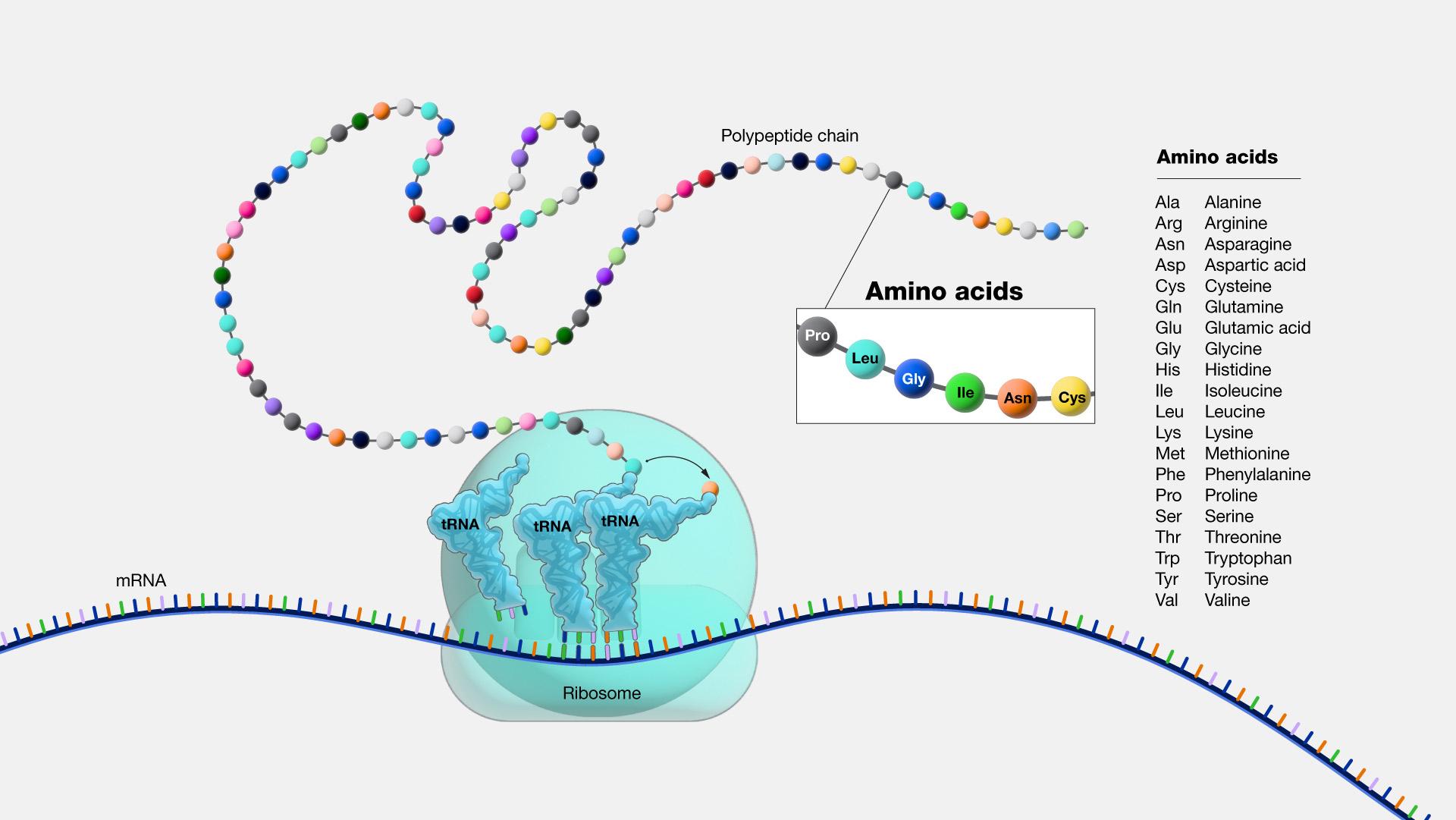
Continuing with our mRNA sequence example from above, the mRNA sequence AUG-CCU-ACG codes for three amino acids. Using a codon table (Figure 4.22), AUG is a codon for methionine (Met), CCU is proline (Pro), and ACG is threonine (Thr). Therefore, the protein sequence is Met-Pro-Thr. Methionine is the most common “start codon” (AUG) for the initiation of protein translation in eukaryotes. As the ribosome moves along the mRNA, the growing amino acid chain exits the ribosome and folds into a protein. When the ribosome reaches a “stop” codon (UAA, UAG, or UGA), the ribosome stops adding any new amino acids, detaches from the mRNA, and the protein is released. Depending upon the amino acid sequence, a linear protein may undergo additional “folding.” The final three-dimensional protein shape is integral to completing a specific structural or functional task.
Dig Deeper: Protein Synthesis
To see protein synthesis in animation, please check out the From DNA to Protein video on YourGenome.org.
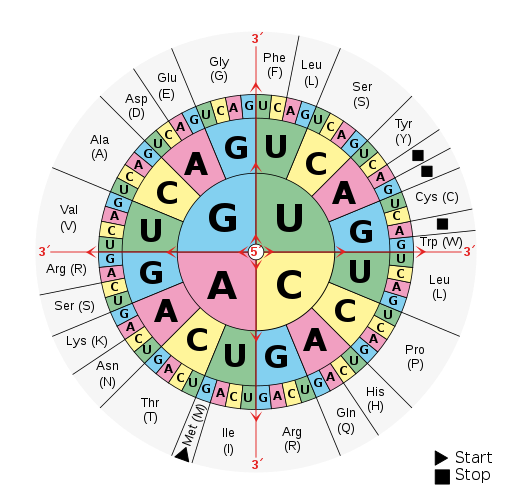
Mendelian Genetics

Gregor Johann Mendel (1822–1884) is often described as the “Father of Genetics.” Mendel was a monk who conducted pea plant breeding experiments in a monastery located in the present-day Czech Republic (Figure 4.23). After several years of experiments, Mendel presented his work to a local scientific community in 1865 and published his findings the following year. Although his meticulous effort was notable, the importance of his work was not recognized for another 35 years. One reason for this delay in recognition is that his findings did not agree with the predominant scientific viewpoints on inheritance at the time. For example, it was believed that parental physical traits “blended” together and offspring inherited an intermediate form of that trait. In contrast, Mendel showed that certain pea plant physical traits (e.g., flower color) were passed down separately to the next generation in a statistically predictable manner. Mendel also observed that some parental traits disappeared in offspring but then reappeared in later generations. He explained this occurrence by introducing the concept of “dominant” and “recessive” traits. Mendel established a few fundamental laws of inheritance, and this section reviews some of these concepts. Moreover, the study of traits and diseases that are controlled by a single gene is commonly referred to as Mendelian genetics.
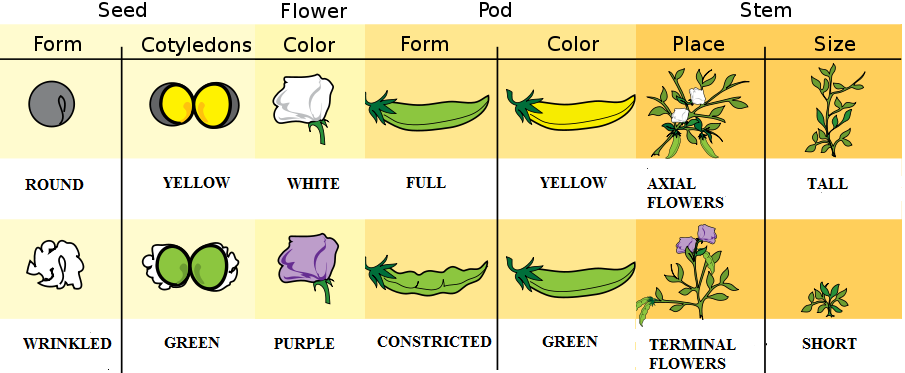
The physical appearance of a trait is called an organism’s phenotype. Figure 4.24 shows pea plant (Pisum sativum) phenotypes that were studied by Mendel, and in each of these cases the physical traits are controlled by a single gene. In the case of Mendelian genetics, a phenotype is determined by an organism’s genotype. A genotype consists of two gene copies, wherein one copy was inherited from each parent. Gene copies are also known as alleles (Figure 4.25), which means they are found in the same gene location on homologous chromosomes. Alleles have a nonidentical DNA sequence, which means their phenotypic effect can be different. In other words, although alleles code for the same trait, different phenotypes can be produced depending on which two alleles (i.e., genotypes) an organism possesses. For example, Mendel’s pea plants all have flowers, but their flower color can be purple or white. Flower color is therefore dependent upon which two color alleles are present in a genotype.
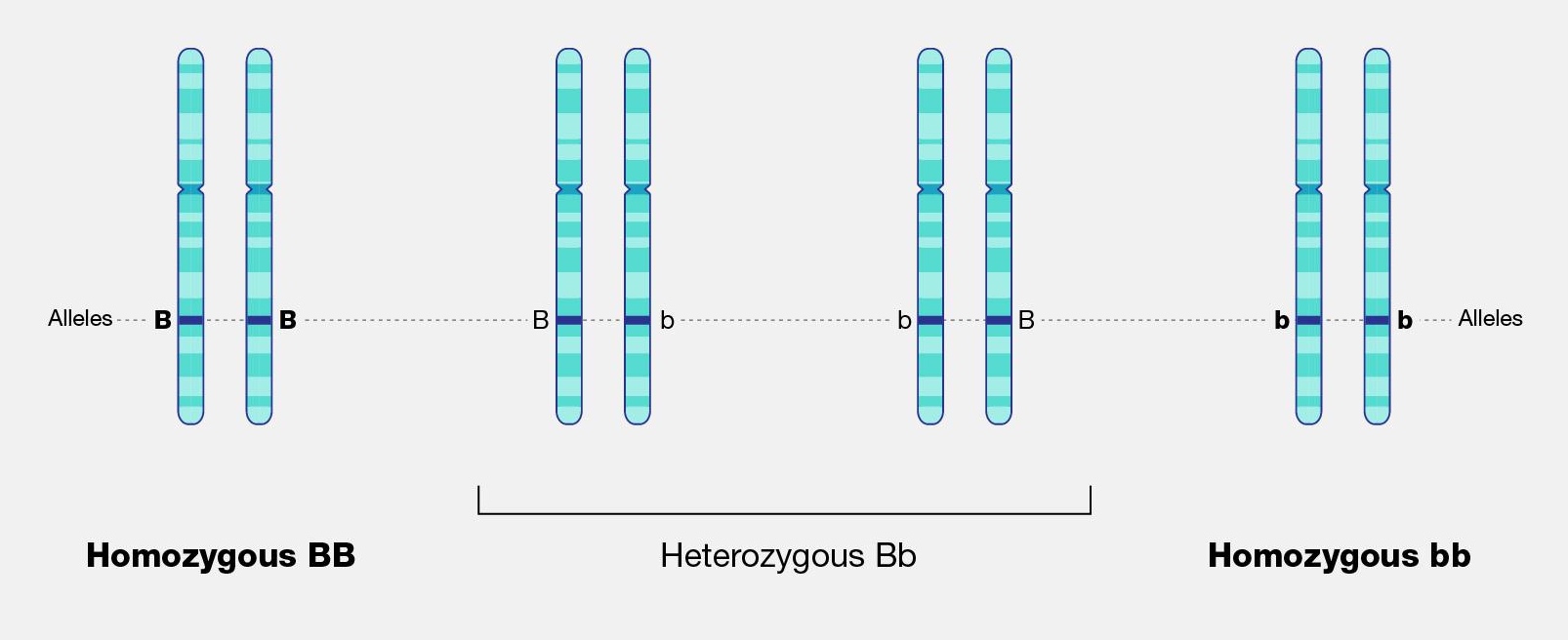
A Punnett square is a diagram that can help visualize Mendelian inheritance patterns. For instance, when parents of known genotypes mate, a Punnett square can help predict the ratio of Mendelian genotypes and phenotypes that their offspring would possess. When discussing genotype, biologists use upper and lower case letters to denote the different allele copies. Figure 4.26 is a Punnett square that includes two heterozygous parents for flower color (Bb). A heterozygous genotype means there are two different alleles for the same gene. Therefore, a pea plant that is heterozygous for flower color has one purple allele and one white allele. When an organism is homozygous for a specific trait, it means their genotype consists of two copies of the same allele. Using the Punnett square example, the two heterozygous pea plant parents can produce offspring with two different homozygous genotypes (BB or bb) or offspring that are heterozygous (Bb).
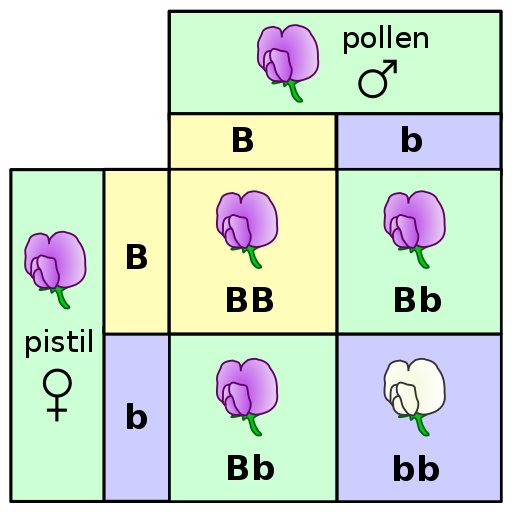
A pea plant with purple flowers could be heterozygous (Bb) or homozygous (BB). This is because the purple color allele (B) is dominant to the white color allele (b), and therefore it only needs one copy of that allele to phenotypically express purple flowers. Because the white flower allele is recessive, a pea plant must be homozygous for the recessive allele in order to have a white color phenotype (bb). As seen by the Punnett square example (Figure 4.26), three of four offspring will have purple flowers and the other one will have white flowers.
The Law of Segregation was introduced by Mendel to explain why we can predict the ratio of genotypes and phenotypes in offspring. As discussed previously, a parent will have two alleles for a certain gene (with each copy on a different homologous chromosome). The Law of Segregation states that the two copies will be segregated from each other and will each be distributed to their own gamete. We now know that the process where that occurs is meiosis.
Offspring are the products of two gametes combining, which means the offspring inherits one allele from each gamete for most genes. When multiple offspring are produced (like with pea plant breeding), the predicted phenotype ratios are more clearly observed. The pea plants Mendel studied provide a simplistic model to understand single-gene genetics. While many traits anthropologists are interested in have a more complicated inheritance (e.g., are informed by many genes), there are a few known Mendelian traits in humans. Additionally, some human diseases also follow a Mendelian pattern of inheritance (Figure 4.27). Because humans do not have as many offspring as other organisms, we may not recognize Mendelian patterns as easily. However, understanding these principles and being able to calculate the probability that an offspring will have a Mendelian phenotype is still important.
| Mendelian disorder | Gene |
| Alpha Thalassemia | HBA1 |
| Cystic Fibrosis | CFTR |
| Fragile X Syndrome | FMR1 |
| Glucose-6-Phosphate Dehydrogenase Deficiency | G6PD |
| Hemophilia A | F8 |
| Huntington disease | HTT |
| Mitochondrial DNA Depletion Syndrome | TYMP |
| Oculocutaneous Albinism: Type 1 | TYR |
| Polycystic Kidney Disease | PKHD1 |
| Sickle-cell anemia | HBB |
| Spinal Muscular Atrophy: SMN1 Linked | SMN1 |
| Tay-Sachs Disease | HEXA |
| Wilson Disease | ATP7B |
Example of Mendelian Inheritance: The ABO Blood Group System
In 1901, Karl Landsteiner at the University of Vienna published his discovery of ABO blood groups. While conducting blood immunology experiments in which he combined the blood of individuals who possess different blood cell types, he observed an agglutination (clotting) reaction. The presence of agglutination implies there is an incompatible immunological reaction; no agglutination will occur in individuals with the same blood type. This work was clearly important because it resulted in a higher survival rate of patients who received blood transfusions. Blood transfusions from someone with a different type of blood causes agglutinations, and the resulting coagulated blood can not easily pass through blood vessels, resulting in death. Landsteiner received the Nobel Prize (1930) for his discovery and explaination of the ABO blood group system.
Blood cell surface antigens are proteins that coat the surface of red blood cells, and antibodies are specifically “against” or “anti” to the antigens from other blood types. Thus, antibodies are responsible for causing agglutination between incompatible blood types. Understanding the interaction of antigens and antibodies helps to determine ABO compatibility amongst blood donors and recipients. To better comprehend blood phenotypes and ABO compatibility, blood cell antigens and plasma antibodies are presented in Figure 4.28. Individuals that are blood type A have A antigens on the red blood cell surface, and anti-B antibodies, which will bind to B antigens should they come in contact. Alternatively, individuals with blood type B have B antigens and anti-A antibodies. Individuals with blood type AB have both A and B antigens but do not produce antibodies for the ABO system. This does not mean type AB does not have any antibodies present, just that specifically anti-A and anti-B antibodies are not produced. Individuals who are blood type O have nonspecific antigens and produce both anti-A and anti-B antibodies.
Figure 4.29 shows a table of the ABO allele system, which has a Mendelian pattern of inheritance. Both the A and B alleles function as dominant alleles, so the A allele always codes for the A antigen, and the B allele codes for the B antigen. The O allele differs from A and B, because it codes for a nonfunctional antigen protein, which means there is no antigen present on the cell surface of O blood cells. To have blood type O, two copies of the O allele must be inherited, one from each parent, thus the O allele is considered recessive. Therefore, someone who is a heterozygous AO genotype is phenotypically blood type A, and a genotype of BO is blood type B. The ABO blood system also provides an example of codominance, which is when both alleles are observed in the phenotype. This is true for blood type AB: when an individual inherits both the A and B alleles, then both A and B antigens will be present on the cell surface.
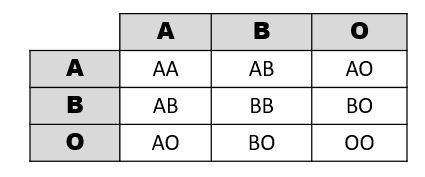
Also found on the surface of red blood cells is the rhesus group antigen, known as “Rh factor.” In reality, there are several antigens on red blood cells independent from the ABO blood system, however, the Rh factor is the second most important antigen to consider when determining blood donor and recipient compatibility. Rh antigens must also be considered when a pregnant mother and her baby have incompatible Rh factors. In such cases, a doctor can administer necessary treatment steps to prevent pregnancy complications and hemolytic disease, which is when the mother’s antibodies break down the newborn’s red blood cells.
An individual can possess the Rh antigen (be Rh positive) or lack the Rh antigen (be Rh negative). The Rh factor is controlled by a single gene and is inherited independently of the ABO alleles. Therefore, all blood types can either be positive (O+, A+, B+, AB+) or negative (O-, A-, B-, AB-).
Individuals with O+ red blood cells can donate blood to A+, B+, AB+, and O+ blood type recipients. Because O- individuals do not have AB or Rh antigens, they are compatible with all blood cell types and are referred to as “universal donors.” Individuals that are AB+ are considered to be “universal recipients” because they do not possess antibodies against other blood types.
Mendelian Patterns of Inheritance and Pedigrees
A pedigree can be used to investigate a family’s medical history by determining if a health issue is inheritable and will possibly require medical intervention. A pedigree can also help determine if it is a Mendelian recessive or dominant genetic condition. Figure 4.30 is a pedigree example of a family with Huntington’s disease, which has a Mendelian dominant pattern of inheritance. In a standard pedigree, males are represented by a square and females are represented by a circle. Biological family members are connected to a horizontal line, with biological parents above and offspring below. When an individual is affected with a certain condition, the square or circle is filled in as a solid color. With a dominant condition, at least one of the parents will have the disease and an offspring will have a 50% chance of inheriting the affected chromosome. Therefore, dominant genetic conditions tend to be present in every generation. In the case of Huntington’s, some individuals may not be diagnosed until later in adulthood, so parents may unknowingly pass this dominantly inherited disease to their children.
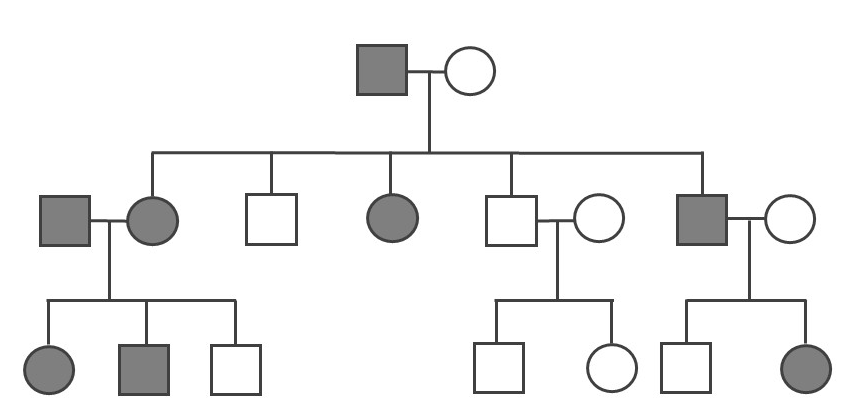
Because the probability of inheriting a disease-causing recessive allele is more rare, recessive medical conditions can skip generations. Figure 4.31 is an example of a family that carries a recessive cystic fibrosis mutation. A parent that is heterozygous for the cystic fibrosis allele has a 50% chance of passing down their affected chromosome to the next generation. If a child has a recessive disease, then it means both of their parents are carriers (heterozygous) for that condition. In most cases, carriers for recessive conditions show no serious medical symptoms. Individuals whose family have a known medical history for certain conditions sometimes seek family planning services (see the Genetic Testing section).
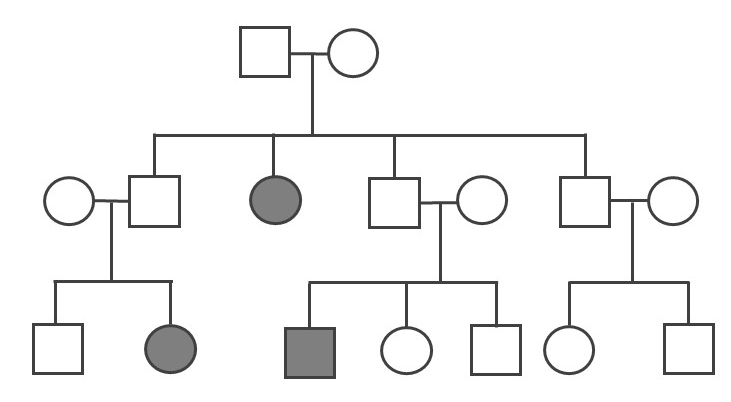
Pedigrees can also help distinguish if a health issue has either an autosomal or X-linked pattern of inheritance. As previously discussed, there are 23 pairs of chromosomes and 22 of these pairs are known as autosomes. The provided pedigree examples (Figure 4.30–31) are autosomally linked genetic diseases. This means the genes that cause the disease are on one of the chromosomes numbered 1 to 22. The conditions caused by genes located on the X chromosome are referred to as X-linked diseases.
Figure 4.32 depicts a family in which the mother is a carrier for the X-linked recessive disease Duchenne Muscular Dystrophy (DMD). The mother is a carrier for DMD, so daughters and sons will have a 50% chance of inheriting the pathogenic DMD allele. Because females have two X chromosomes, females who inherit only one copy will not have the disease (although in rare cases, female carriers may show some symptoms of the disease). On the other hand, males who inherit a copy of an X-linked pathogenic DMD allele will typically be affected with the condition. Thus, males are more susceptible to X-linked conditions because they only have one X chromosome. Therefore, when evaluating a pedigree, if a higher proportion of males are affected with the disease, this could suggest the disease is X-linked recessive.
Compared to the X chromosome, the Y chromosome is smaller with only a few genes. Y-linked traits are therefore rare and can only be passed from a chromosomal father to a biological XY child.
Other Patterns of Inheritance
Complexity Surrounding Mendelian Inheritance
Pea plant trait genetics are relatively simple compared to what we know about genetic inheritance today. The vast majority of genetically controlled traits are not strictly dominant or recessive, so the relationship among alleles and predicting phenotype is often more complicated. For example, traits that exhibit incomplete dominance occur when a heterozygote exhibits a phenotype that is an intermediate phenotype of both alleles. In snapdragon flowers, the red flower color (R) is dominant and white is recessive (r). Therefore, the homozygous dominant RR is red and homozygous recessive rr is white. However, because the R allele is not completely dominant, the heterozygote Rr is a blend of red and white, which results in a pink flower (Figure 4.33).

An example of incomplete dominance in humans is the enzyme β-hexosaminidase A (Hex A), which is encoded by the gene HEXA. Patients with two dysfunctional HEXA alleles are unable to metabolize a specific lipid-sugar molecule (GM2 ganglioside); because of this, the molecule builds up and causes damage to nerve cells in the brain and spinal cord. This condition is known as Tay-Sachs disease, and it usually appears in infants who are three to six months old. Most children with Tay-Sachs do not live past early childhood. Individuals who are heterozygous for the functional type HEXA allele and one dysfunctional allele have reduced Hex A activity. However, the amount of enzyme activity is still sufficient, so carriers do not exhibit any neurological phenotypes and appear healthy.
Some genes and alleles can also have higher penetrance than others. Penetrance can be defined as the proportion of individuals who have a certain allele and also express an expected phenotype. If a genotype always produces an expected phenotype, then those alleles are said to be fully penetrant. However, in the case of incomplete (or reduced) penetrance, an expected phenotype may not occur even if an individual possesses the alleles that are known to control a trait or cause a disease.
A well-studied example of genetic penetrance is the cancer-related genes BRCA1 and BRCA2. Mutations in these genes can affect crucial processes such as DNA repair, which can lead to breast and ovarian cancers. Although BRCA1 and BRCA2 mutations have an autosomal dominant pattern of inheritance, it does not mean an individual will develop cancer if they inherit a pathogenic allele. Several lifestyle and environmental factors can also influence the risk for developing cancer. Regardless, if a family has a history of certain types of cancers, then it is often recommended that genetic testing be performed for individuals who are at risk. Moreover, publically available genetic testing companies are now offering health reports that include BRCA1 and BRCA2 allele testing (see the Genetic Testing section).
Polygenic Traits
While Mendelian traits tend to be influenced by a single gene, the vast majority of human phenotypes are polygenic traits. The term polygenic means “many genes.” Therefore, a polygenic trait is influenced by many genes that work together to produce the phenotype. Human phenotypes such as hair color, eye color, height, and weight are examples of polygenic traits. Hair color, for example, is largely determined by the type and quantity of a pigment called melanin, which is produced by a specialized cell type within the skin called melanocytes. The quantity and ratio of melanin pigments determine black, brown, blond, and red hair colors. MC1R is a well-studied gene that encodes a protein expressed on the surface of melanocytes that is involved in the production of eumelanin pigment. Typically, people with two functional copies of MC1R have brown hair. People with reduced functioning MC1R allele copies tend to produce pheomelanin, which results in blond or red hair. However, MC1R alleles have variable penetrance, and studies are continually identifying new genes (e.g., TYR, TYRP1, SLC24A5, and KITLG) that also influence hair color. Individuals with two nonfunctioning copies of the gene TYR have a condition called oculocutaneous albinism—their melanocytes are unable to produce melanin so these individuals have white hair, light eyes, and pale skin.
In comparison to Mendelian diseases, complex diseases (e.g., Type II diabetes, coronary heart disease, Alzheimer's, and schizophrenia) are more prevalent in humans. Complex diseases are polygenic, but their development is also influenced by physical, environmental, sociocultural, and individual lifestyle factors. Families can be more predisposed to certain diseases; however, complex diseases often do not have a clear pattern of inheritance.
Although research of complex traits and diseases continue, geneticists may not know all of the genes involved with a given complex disease. Additionally, how much genetic versus nongenetic determinants contribute to a disease phenotype can be difficult to decipher. Therefore, predicting individual medical risk and risk across different human populations is often a significant challenge. For instance, cardiovascular diseases (CVDs) continue to be one of the leading causes of death around the world. Development of CVDs has been linked to nutrient exposure during fetal development, high fat and sedentary lifestyles, drug usage, adverse socioeconomic conditions, and various genes. Human environments are diverse, and public health research including the field of Human Biology can help identify risk factors and behaviors associated with chronic diseases. Large-scale clinical genetic studies with powerful bioinformatic approaches can also help elucidate some of these complex relationships.
Genomics and Epigenetics
A genome is all of the genetic material of an organism. In the case of humans, this includes 46 chromosomes and mtDNA. The human genome contains approximately three billion base pairs of DNA and has regions that are both noncoding and coding. Scientists now estimate that the human genome contains 20,000–25,000 protein-coding genes, with each chromosome containing a few hundred to a few thousand genes. As our knowledge of heredity increases, researchers have begun to realize the importance of epigenetics, or changes in gene expression that do not result in a change of the underlying DNA sequence. Epigenetics research is also crucial for unraveling gene regulation, which involves complex interactions between DNA, RNA, proteins, and the environment.
Genomics
The vast majority of the human genome is noncoding, meaning there are no instructions to make a protein or RNA product in these regions. Historically, noncoding DNA was referred to as “junk DNA” because these vast segments of the genome were thought to be irrelevant and nonfunctional. However, continual improvement of DNA sequencing technology along with worldwide scientific collaborations and consortia have contributed to our increased understanding of how the genome functions. Through these technological advances and collaborations, we have since discovered that many of these noncoding DNA regions are involved in dynamic genetic regulatory processes.
Genomics is a diverse field of molecular biology that focuses on genomic evolution, structure, and function; gene mapping; and genotyping (determining the alleles present). Evolutionary genomics determined that humans share about 98.8% percent of their DNA with chimpanzees. Given the phenotypic differences between humans and chimpanzees, having a DNA sequence difference of 1.2% seems surprising. However, a lot of genomics research is also focused on understanding how noncoding genomic regions influence how individual genes are turned “on” and “off” (i.e., regulated). Therefore, although DNA sequences are identical, regulatory differences in noncoding genetic regions (e.g., promoters) are believed to be largely responsible for the physical differences between humans and chimpanzees.
Further understanding of genomic regulatory elements can lead to new therapies and personalized treatments for a broad range of diseases. For example, targeting the regulatory region of a pathogenic gene to “turn off” its expression can prevent its otherwise harmful effects. Such molecular targeting approaches can be personalized based on an individual’s genetic makeup. Genome-wide association studies (GWAS), which seek to determine genes that are linked to complex traits and diseases, typically require significant computational efforts. This is because millions of DNA sequences must be analyzed and GWAS sometimes include thousands of participants. During the beginning of the genomics field, most of the large-scale genomics studies only included North American, European, and East Asian participants and patients. Researchers are now focusing on increasing ethnic diversity in genomic studies and databases. In turn, accuracy of individual disease risk across all human populations will be improved and more rare disease–causing alleles will be identified.
Epigenetics
All cells within your body have the same copy of DNA. For example, a brain neuron has the same DNA blueprint as does a skin cell on your arm. Although these cells have the same genetic information, they are considered specialized. The reason all cells within the body have the same DNA but different morphologies and functions is that different subsets of genes are turned “on” and “off” within the different cell types. A more precise explanation is that there is differential expression of genes among different cell types. In the case of neuronal cells, a unique subset of genes are active that allow them to grow axons to send and receive messages. This subset of genes will be inactive in non-neuronal cell types such as skin cells. Epigenetics is a branch of genetics that studies how these genes are regulated through mechanisms that do not change the underlying DNA sequence.
The prefix epi- means “on, above, or near,” and epigenetic mechanisms such as DNA methylation and histone modifications occur on, above, or near DNA. The addition of a methyl group (— CH₃) to DNA is known as DNA methylation (Figure 4.34). DNA methylation and other modifications made to the histones around which DNA are wrapped are thought to make chromatin more compact. This DNA is inaccessible to transcription factors and RNA polymerases, thus preventing genes from being turned on (i.e., transcribed). Other histone modifications have the opposite effect by loosening chromatin, which makes genes accessible to transcription factors.
It is important to note that environmental factors can alter DNA methylation and histone modifications and also that these changes can be passed from generation to generation. For example, someone’s epigenetic profile can be altered during a stressful time (e.g., natural disasters, famine, etc.), and those regulatory changes can be inherited by the next generation. Moreover, our epigenetic expression profile changes as we age. For example, certain places in our genome become “hyper” or “hypo” methylated over time. Identical twins also have epigenetic profiles that become more different as they age. Researchers are only beginning to understand the significance of these genome-wide epigenetic changes. Scientists have also discovered that changes in epigenetic modifications can alter gene expression in ways that contribute to diseases. It is also important to note that, unlike DNA mutations (which permanently change the nucleotide sequence), epigenetic changes can be easily reversed. A lot of research now focuses on how drugs can alter or modulate changes in DNA methylation and histone modifications to treat diseases such as cancer.
Environmental Disruptors and Their Impact on Human Reproductive Systems
The National Institute of Environmental Health Sciences (NIEHS) defines endocrine-disrupting chemicals (EDCs) as synthetic or natural compounds that interfere with the body’s hormonal systems. Found in pesticides, plastics, industrial chemicals, and pollutants, EDCs can mimic, block, or alter the natural action of hormones (NIEHS, 2024). Their effects on reproductive health are profound, particularly during critical developmental windows while also affecting subsequent generations through epigenetic changes.
NIEHS declared EDC’s:
| Atrazine | one of the most commonly applied herbicides in the world, often used to control weeds in corn, sorghum, and sugarcane crops. |
| Bisphenol A (BPA) | used to make polycarbonate plastics and epoxy resins. It is used in manufacturing, food packaging, toys, and other applications. BPA resins may be found in the lining of some canned foods and beverages. |
| Dioxins | a byproduct of certain manufacturing processes, such as herbicide production and paper bleaching. They can be released into the air from waste burning and wildfires. |
| Perchlorate | a colorless salt manufactured and used as an industrial chemical to make rockets, explosives, and fireworks, which can be found in some groundwater. |
| Polyfluoroalkyl Substances (PFAS) | a large group of chemicals used widely in industrial applications, such as firefighting foam, nonstick pans, paper, and textile coatings. |
| Phthalates | a large group of compounds used as liquid plasticizers. They are found in hundreds of products including some food packaging, cosmetics, fragrances, children’s toys, and medical device tubing. Cosmetics that may contain phthalates include nail polish, hair spray, aftershave lotion, cleanser, and shampoo. |
| Phytoestorgens | naturally occurring substances with hormone-like activity found in some plants; they may have a similar effect to estrogen produced by the body. Soy foods, for example, contain phytoestrogens. |
| Polybrominated diphenyl ethers (PBDE) | used to make flame retardants for products such as furniture foam and carpet. |
| Polychlorinated biphenyls (PCBs) | used to make electrical equipment, such as transformers, and are in hydraulic fluids, heat transfer fluids, lubricants, and plasticizers. PCBs were mass-produced globally until they were banned in 1979. |
| Triclosan | an ingredient that was previously added to some antimicrobial and personal care products, like liquid body wash and soaps. |
The Male Reproductive System: Vulnerabilities, Epigenetics, and Disruptions
The male reproductive system is highly sensitive to hormonal interference, especially during prenatal and early postnatal development. Over the past 50 years, epidemiological data gathered by the NIEHS has revealed alarming changes: increased cases of prostate and testicular cancers, male-descended testes, and anatomical malformations of male genitalia (Sweeney et al., 2015). These changes are accompanied by a global decline in sperm quality, underscoring the widespread vulnerability of male reproductive health to environmental factors. The testes, as the site of sperm production and testosterone synthesis, are particularly susceptible to EDC interference. Proper testicular development depends on tightly regulated hormonal signalling, which EDCs can disrupt by mimicking or blocking hormones like testosterone and estrogen, leading to improper testicular formation and increased risk of testicular cancer. Prostate development is also a target for EDC interference. African American men, for example, exhibit twice the risk of developing prostate cancer than Caucasian men. This disparity has been attributed to hereditary, lifestyle, and environmental factors, often causing elevated maternal estrogen levels during gestation. This prenatal exposure to EDCs can mimic estrogen and predispose developing prostate tissues to cancerous changes in adulthood (2015).
The Female Reproductive System: Epigenetics and Fertility Challenges
Female fertility relies on a delicate hormonal balance to regulate processes such as ovulation, implantation, and pregnancy. EDCs can disrupt this balance by mimicking, antagonizing, or altering the action of hormones. Their interference contributes to a wide range of reproductive disorders, including early puberty, premature ovarian failure, anovulation, and infertility. Epigenetics plays a central role in female reproductive health. DNA methylation, histone modifications, and ncRNA generation are crucial for regulating ovarian and uterine function; However, EDCs can affect these regulatory mechanisms. An example of this is primordial germ cells (PGCs) in female embryos, which need to undergo extensive epigenetic reprogramming during development (Biswas et al., 2021). This process erases genomic imprinting and reactivates the inactive X chromosome, creating a "blank slate" for the next generation; however, EDCs can disrupt this critical period of epigenetic resetting, leading to long-term consequences for reproductive health.
The ovarian follicle–the functional unit of female reproduction–is particularly vulnerable to these chemicals. Being exposed to EDCs can deplete the pool of these follicles, leading to temporary or permanent infertility (2021). Additionally, EDCs interfere with estrogen receptor function, a crucial regulator of female reproductive processes. These chemicals bind to these receptors, altering the recruitment of enzymes involved in histone modification and chromatin remodelling; this disrupts gene expression patterns critical for ovarian and uterine health. One striking example is diethylstilbestrol (DES), a synthetic estrogen once prescribed to pregnant women (2021). DES exposure has been linked to ovarian cancer in subsequent generations, highlighting the transgenerational effects of EDCs on the female reproductive system. In severe cases, EDCs induce multigenerational reproductive disorders, as observed in studies linking DES to ovarian cancer in the grandchildren of exposed individuals.
Epigenetic Therapy
Heritable Changes and Some Related Drugs
As has been said, epigenetics involves heritable changes in gene expression, without involving DNA alteration. These changes, being heritable and often involving abnormal DNA methylation patterns within the four DNA methyltransferases (DNMTs) or histone modifications in chromatin, can lead to disease development. DNMTs (DNMT1, DNMT2, DNMT3A, and DNMT3B) have functions specific to themselves and are at the core of the DNA methylation process. Regarding the histone modifications mentioned, histones have been recognized to mutate under various mechanisms, such as acetylation, methylation and phosphorylation. The acetylation of histones involves histone acetyltransferases (HATs), which are associated with the activation of gene transcription. This process is reversed by the deacetylation of histones, which is associated with the silencing of gene transcription under histone deacetylases (HDACs). (Peedicayil, 2006)
Epigenetic therapy, with the use of specialized drug developments, aims to correct epigenetic defects, which are reversible under pharmacological intervention, by targeting enzymes such as HATs, HDACs and DNMTs, as well as histone methyltransferases. For instance, certain drugs are being developed as DNMT inhibitors, stopping the methylation of DNA associated with inappropriate transcriptional silencing of genes, and potentially increasing haemoglobin F to help patients affected by sickle cell anemia. These DNMT inhibitor drugs have been classified under three categories based on their structures: nucleoside analogue DNMT inhibitors, non-nucleoside analogue DNMT inhibitors, and antisense oligonucleotides (2006). Nucleoside analogue DNMT inhibitors are analogues of cytosine, the nucleotide affected by methylation from DNMTs, and are incorporated into replicating DNA, replacing cytosine, thus being S-phase-specific drugs. Non-nucleoside analogue DNMT inhibitors are researched to reduce the myelotoxic effects of drugs directly incorporated into the DNA, and are brought into the patient differently. Antisense oligonucleotides are drugs made up of sequences of nucleotides complementary to mRNAs, made to block translation, by acting on the DNMT1 for instance. Additionally, drugs such as HDAC inhibitors help maintain the acetylation of histones, leading to apoptosis, growth arrest or differentiation of tumour cells, giving this drug an anticancer effect, suppressing tumour growth. (2006)
Implications with Cancers
Research published in The Indian Journal for Medical Research has shown that these drugs show promising results in cancer treatment trials involving solid tumours and hematological malignancies. However, they have limitations, for instance, the fact that DNMT and HDAC inhibitors could activate oncogenes due to limited specificity, leading to further tumor progression; or their high myelotoxicity levels, a side effect thought to be due to their incorporation into DNA, and nucleotide analogue inhibitors (2006). Though that is the case, it is important to know that epigenetic drugs alone or in combination with conventional anticancer drugs, may prove to be a significant advance over the use of conventional anticancer drugs, and may also be a way to prevent diseases. Additionally, combination therapy strategies targeting various epigenetic markers, such as DNMTs for cancer-related genes and non-selective HDAC inhibitors, have been shown to yield promising results, simultaneously inducing the expression of tumor suppressor genes and inhibiting the expression of key oncogenes. As recently explored by researchers in Cell Death Discovery, this specific case of combination therapy would synergistically induce gene expression while maintaining the selectivity required to increase targeting of particular tumor types based on gene expression profiles. (Yu et al., 2024)
To date, the majority of cases in which epigenetic defects have led to disease pathogenesis are cancers (Peedicayil, 2006), cancer cells often developing due to uncontrolled cell growth and resistance to cell death mechanisms, made possible with abnormal DNA methylation patterns as well as histone modifications (Yu et al., 2024). Epigenetic alterations have therefore been identified within the core of tumor progression mechanisms in cancer cells, including tumorigenesis, promotion, progression, and recurrence, suggesting epigenetic heterogeneity at the cellular level (2024). Certain drugs have been developed, showing specifically good results for cancer treatments, by inhibiting enzymes such as KMTs and KDMs. These can be added to the growing list of drugs fitting into epigenetic therapy, including DNMT and HDAC inhibitors, as well as combination therapy treatments, for cancer and other diseases.
Purpose of Study and Future Developments
Studying the link between epigenetics and diseases is crucial for multiple reasons, one of which is enabling scientists and researchers to better understand disease mechanisms, detect abnormal epigenetic changes, and, in turn, develop more effective treatments or possibly even prevent diseases from developing in the first place. As previously mentioned, epigenetic therapy has been shown to bring promising results in drug trials surrounding cancer treatments. Still, the range of diseases to be treated with this new pharmacology approach is vast, molecules other than DNMTs and HDACs being related to epigenetic mechanisms within gene expression, such as BET proteins and KDMs, potentially being a source of new medications or treatments (Yu et al., 2024; Peedicayil, 2006). Additionally, by understanding someone's epigenetic profile, a form of personalized “precision medicine” (Yu et al., 2024, p. 8) is developed, offering less toxic and more effective treatments with fewer undesired side effects. Researchers expanding this field of knowledge would be able to understand, in more concrete terms, how external factors are linked to epigenetic changes and, consequently, disease risk, potentially halting disease progression and developing new prevention mechanisms. Personalized medicine combines both genetic and epigenetic data, including gene expression profiles, DNA methylation patterns, histone modification profiles, and identified biomarkers, to create precise disease management and prediction.
It is crucial to keep in mind that diseases like cancer are linked to major causes of morbidity and mortality worldwide, which could be reduced with therapeutic medicine such as epigenetic therapy, aiming to detect cancer biomarkers to improve risk assessment, diagnosis, and targeted treatment interventions, limiting the burden of chronic and life-threatening diseases. With the advancement of epigenetic therapies, new sequencing techniques, as well as AI (2024), have opened avenues to establish precision diagnostics and therapeutics for patients.
With this said, epigenetics is a relatively new area of scientific research. This field has exploded in the last few decades, especially with the advancement of technologies that allow researchers to examine DNA methylation patterns, histone modifications, and non-coding RNA molecules across the genome. While the potential of epigenetics in explaining complex diseases, including those linked to environmental factors such as endocrine-disrupting chemicals (EDCs), is immense, we’ve identified two key challenges. One major limitation is the complexity and variability of epigenetic marks. These modifications can differ significantly across cell types, tissues, and even individuals, making it difficult to generalize findings.
Additionally, epigenetic changes are dynamic and can fluctuate over time, which complicates the task of linking them to specific environmental exposures or health outcomes. Another challenge lies in the transgenerational aspect of epigenetics. While it's clear that epigenetic changes can be passed from one generation to the next, the mechanisms behind this inheritance are not fully understood. It's also difficult to pinpoint exactly when and how these modifications occur in development, especially since environmental exposures may affect individuals at different stages of their life, with varying effects depending on the timing and dose.
Special Topic: Epigenetics and X Chromosome Inactivation

Mary Lyon was a British geneticist who presented a hypothesis for X chromosome inactivation (called the Lyon hypothesis) based on her work and other studies of the day. Females inherit two X chromosomes, one from each parent. Males have one functional X chromosome; however, this does not mean females have more active genes than males. During the genetic embryonic development of many female mammals, one of the X chromosomes is inactivated at random, so females have one functional X chromosome. The process of X chromosome inactivation in females occurs through epigenetic mechanisms, such as DNA methylation and histone modifications. Recent studies have analyzed the role of a long noncoding RNA called X-inactive specific transcript (XIST), which is largely responsible for the random silencing of one of the X chromosomes. The presence of two X chromosomes is the signal for XIST RNA to be expressed so that one X chromosome can be inactivated. However, some cells may have an active paternal X chromosome while other cells may have an active maternal X chromosome. This phenomenon is easily seen in calico and tortoiseshell cats (Figure 4.35). In cats, the gene that controls coat color is found on the X chromosome. During early embryo development, random inactivation of X chromosomes gives rise to populations of cells that express black or orange, which results in the unique coat patterning. Therefore, calico cats are typically always female.
Genetic Testing
To assist with public health efforts, newborn screening for genetic diseases has been available in the United States for over 50 years. One of the first available genetic tests was to confirm a phenylketonuria (PKU) diagnosis in infants, which is easily treatable with a dietary change. Currently, each state decides what genes are included on newborn screening panels and some states even have programs to help with infant medical follow-ups. There are now hundreds of laboratories that provide testing for a few thousand different genes that can inform medical decisions for infants and adults. Moreover, genetic testing has been made available publicly to anyone without the assistance of medical professionals.
Clinical Testing
Clinical genetics tests assist patients with making medically informed decisions about family planning and health. Applications of this technology include assistance with in vitro fertilization (IVF) procedures, embryo genetic screening, and personalized medicine such as matching patients to cancer therapies. To ensure accuracy of patient genetic screening, it is important that all clinical laboratories are regulated. The Clinical Laboratory Improvement Amendments (CLIA) are United States federal standards that all human laboratory testing clinics must follow. A major benefit provided by some clinical genetic testing companies is access to genetic counselors, who have specialized education and training in medical genetics and counseling. For individuals with a family history of genetic disease, a physician may recommend genetic carrier screening to see if there is a risk for passing on a disease to a child. Genetic counselors provide expertise with interpretation of genetic testing results, as well as help guide and support patients when making impactful medical decisions.
Review Questions
- What is the purpose of DNA replication? Explain in a few sentences what happens during DNA replication. When do DNA mutations happen? And how does this create phenotypic variation (i.e., different phenotypes of the same physical trait)?
- Using your own words, what are homologous chromosomes and sister chromatids? What are the key differences between mitosis and meiosis?
- Determine if the pedigree diagram below (Figure 4.41) represents an autosomal dominant, autosomal recessive, or X-linked recessive pattern of inheritance. You should write the genotype (i.e., AA, Aa, or aa) above each square to help you (note: there may sometimes be two possible answers for a square’s genotype). Please also explain why you concluded a particular pattern of inheritance.
- Use base pairing rules to transcribe the following DNA template sequence into mRNA: GTAAAGGTGCTGGCCATC. Next, use the protein codon table (see Figure 4.21) to translate the sequence. In regard to transcription, explain what the significance is of the first and last codon/protein in the sequence.
- In your opinion, what do you think the benefits are of direct-to-consumer (DTC) genetic testing? What are the drawbacks and/or greater ethical concerns? Do you think benefits outweigh concerns?
- Imagine that you submit your DNA sample to a genetic testing company and among the various diseases for which they test, there is an allele that is associated with late-onset Alzheimer’s disease. You have the option to view your Alzheimer’s result or to not view your result. What do you do and why?
Key Terms
Adenosine triphosphate (ATP): A high-energy compound produced by mitochondria that powers cellular processes.
Allele: A nonidentical DNA sequence found in the same gene location on a homologous chromosome, or gene copy, that codes for the same trait but produces a different phenotype.
Amino acids: Organic molecules that are the building blocks of protein. Each of the 20 different amino acids have their own unique chemical property. Amino acids are chained together to form proteins.
Ancient DNA (aDNA): DNA that is extracted from organic remains and that often dates from hundreds to thousands of years ago. Also, aDNA is typically degraded (i.e., damaged) due to exposure to the elements such as heat, acidity, and humidity.
Aneuploid: A cell with an unexpected amount of chromosomes. The loss or gain of chromosomes can occur during mitotic or meiotic division.
Antibodies: Immune-related proteins that can detect and bind to foreign substances in the blood such as pathogens.
Apoptosis: A series of molecular steps that is activated leading to cell death. Apoptosis can be activated when a cell fails checkpoints during the cell cycle; however, cancer cells have the ability to avoid apoptosis.
Autosomal: Refers to a pattern of inheritance in which an allele is located on an autosome (non sex chromosome).
Base pairs: Chemical bonding between nucleotides. In DNA, adenine (A) pairs with thymine (T) and cytosine (C) pairs with guanine (G); in RNA, adenine (A) always pairs with uracil (U).
Carbohydrate: Molecules composed of carbon and hydrogen atoms that can be broken down to supply energy.
Carrier: An individual who has a heterozygous genotype that is typically associated with a disease.
Cell cycle: A cycle the cell undergoes with checkpoints between phases to ensure that DNA replication and cell division occur properly.
Cell surface antigen: A protein that is found on a red blood cell’s surface.
Centromere: A structural feature that is defined as the “center” of a chromosome and that creates two different arm lengths. This term also refers to the region of attachment for microtubules during mitosis and meiosis.
Chromatin: DNA wrapped around histone complexes. During cell division, chromatin becomes a condensed chromosome.
Chromosome: DNA molecule that is wrapped around protein complexes, including histones.
Codominance: The effects of both alleles in a genotype can be seen in the phenotype.
Codons: A sequence that comprises three DNA nucleotides that together code for a protein.
Complex diseases: A category of diseases that are polygenic and are also influenced by environment and lifestyle factors.
Cytoplasm: The “jelly-like” matrix inside of the cell that contains many organelles and other cellular molecules.
Deleterious: A mutation that increases an organism’s susceptibility to disease.
Deoxyribonucleic acid (DNA): A molecule that carries the hereditary information passed down from parents to offspring. DNA can be described as a “double helix”’ shape. It includes two chains of nucleotides held together by hydrogen bonds with a sugar phosphate backbone.
Diploid: Refers to an organism or cell with two sets of chromosomes.
DNA methylation: Methyl groups bind DNA, which modifies the transcriptional activity of a gene by turning it “on” or “off.”
DNA polymerase: Enzyme that adds nucleotides to existing nucleic acid strands during DNA replication. These enzymes can be distinguished by their processivity (e.g., DNA replication).
DNA replication: Cellular process in which DNA is copied and doubled.
DNA sequence: The order of nucleotide bases. A DNA sequence can be short, long, or representative of entire chromosomes or organismal genomes.
Dominant: Refers to an allele for which one copy is sufficient to be visible in the phenotype.
Elongation: The assembly of new DNA from template strands with the help of DNA polymerases.
Enzymes: Proteins responsible for catalyzing (accelerating) various biochemical reactions in cells.
Epigenetic profile: The methylation pattern throughout a genome—that is, which genes (and other genomic sites) are methylated and unmethylated.
Epigenetics: Changes in gene expression that do not result in a change of the underlying DNA sequence. These changes typically involve DNA methylation and histone modifications. These changes are reversible and can also be inherited by the next generation.
Euchromatin: Loosely coiled chromosomes found within the nucleus that are accessible for regulatory processing of DNA.
Eukaryote: Single-celled or multicelled organism characterized by a distinct nucleus, with each organelle surrounded by its own membrane.
Exon: Protein-coding segment of a gene.
Gametes: Haploid cells referred to as an egg and sperm that will fuse together during sexual reproduction to form a diploid organism.
Gene: Segment of DNA that contains protein-coding information and various regulatory (e.g., promoter) and noncoding (e.g., introns) regions.
Genetic recombination: A cellular process that occurs during meiosis I in which homologous chromosomes pair up and sister chromatids on different chromosomes physically swap genetic information.
Genome: All the genetic information of an organism.
Genotype: The combination of two alleles that code for or are associated with the same gene.
Genotyping: A molecular procedure that is performed to test for the presence of certain alleles or to discover new ones.
Germ cells: Specialized cells that form gametes (egg and sperm cells).
Haploid: Cell or organism with one set of chromosomes (n = 23).
Helicase: A protein that breaks the hydrogen bonds that hold double-stranded DNA together.
Heterozygous: Genotype that consists of two different alleles.
Histones: Proteins that DNA wraps around to assist with DNA organization within the nucleus.
Homologous chromosomes: A matching pair of chromosomes wherein one chromosome is maternally inherited and the other is paternally inherited.
Homozygous: Genotype that consists of two identical alleles.
Incomplete dominance: Heterozygous genotype that produces a phenotype that is a blend of both alleles.
Initiation: The recruitment of proteins to separate DNA strands and begin DNA replication.
Interphase: Preparatory period of the cell cycle when increased metabolic demand allows for DNA replication and doubling of the cell prior to cell division.
Introns: Segment of DNA that does not code for proteins.
Karyotyping: The microscopic procedure wherein the number of chromosomes in a cell is determined.
Lagging strand: DNA template strand that is opposite to the leading strand during DNA replication. This strand is created in several disconnected sections and other enzymes fill in the missing nucleotide gaps between these sections.
Leading strand: DNA template strand in which replication proceeds continuously.
Lipids: Fatty acid molecules that serve various purposes in the cell, including energy storage, cell signaling, and structure.
Meiosis: The process that gametes undergo to divide. The end of meiosis results in four haploid daughter cells.
Mendelian genetics: A classification given to phenotypic traits that are controlled by a single gene.
Messenger RNA (mRNA): RNA molecule that is transcribed from DNA. Its tri-nucleotide codons are “read” by a ribosome to build a protein.
Microarray technology: A genotyping procedure that utilizes a microarray chip, which is a collection of thousands of short nucleotide sequences attached to a solid surface that can probe genomic DNA.
Microbiome: The collective genomes of the community of microorganisms that humans have living inside of their bodies.
Mitochondrial DNA (mtDNA): Circular DNA segment found in mitochondria that is inherited maternally.
Mitochondrion: Specialized cellular organelle that is the site for energy production. It also has its own genome (mtDNA).
Mitosis: The process that somatic cells undergo to divide. The end of mitosis results in two diploid daughter cells.
Molecular anthropologists: Individuals who use molecular techniques (primarily genetics) to compare ancient and modern populations and to study living populations of humans and nonhuman primates.
Molecular geneticists: Biologists that study the structure and function of genes.
Mutation: A nucleotide sequence variation from the template DNA strand that can occur during replication. Mutations can also happen during recombination.
Next-generation sequencing: A genotyping technology that involves producing millions of nucleotide sequences (from a single DNA sample) that are then read with a sequencing machine. It can be used for analyzing entire genomes or specific regions and requires extensive program-based applications.
Nuclear envelope: A double-layered membrane that encircles the nucleus.
Nucleic acid: A complex structure (like DNA or RNA) that carries genetic information about a living organism.
Nucleotide: The basic structural component of nucleic acids, which includes DNA (A, T, C, and G) and RNA (A, U, C, and G).
Nucleus: Double-membrane cellular organelle that helps protect DNA and also regulates nuclear activities.
Organelle: A structure within a cell that performs specialized tasks that are essential for the cell. There are different types of organelles, each with its own function.
Pathogenic: A genetic mutation (i.e., allele) that has a harmful phenotypic disease-causing effect.
Pedigree: A diagram of family relationships that indicates which members may have or carry certain genetic and/or phenotypic traits.
Penetrance: The proportion of how often the possession of an allele results in an expected phenotype. Some alleles are more penetrant than others.
Phenotype: The physical appearance of a given trait.
Phospholipid bilayer: Two layers of lipids that form a barrier due to the properties of a hydrophilic (water-loving) head and a hydrophobic (water-repelling) tail.
Polygenic trait: A phenotype that is controlled by two or more genes.
Polymerase chain reaction (PCR): A molecular biology procedure that can make copies of genomic DNA segments. A small amount of DNA is used as a starting template and is then used to make millions of copies.
Prokaryote: A single-celled organism characterized by the lack of a nucleus and membrane-enclosed organelles.
Promoter: The region of a gene that initiates transcription. Transcription factors can bind and DNA methylation may occur at a promoter site, which can modify the transcriptional activities of a gene.
Protein: Chain of amino acids that folds into a three-dimensional structure that allows a cell to function in a variety of ways.
Protein synthesis: A multi-step process by which amino acids are strung together by RNA machinery read from a DNA template.
Recessive: Refers to an allele whose effect is not normally seen unless two copies are present in an individual’s genotype.
Ribonucleic acid (RNA): Single-stranded nucleic acid molecule.There are different RNAs found within cells and they perform a variety of functions, such as cell signaling and involvement in protein synthesis.
Ribosomal RNA (rRNA): A ribosome-bound molecule that is used to correctly assemble amino acids into proteins.
Ribosome: An organelle in the cell found in the cytoplasm or endoplasmic reticulum. It is responsible for reading mRNA and protein assemblage.
RNA polymerase: An enzyme that catalyzes the process of making RNA from a DNA template.
Sanger-sequencing: A process that involves the usage of fluorescently labeled nucleotides to visualize DNA (PCR fragments) at the nucleotide level.
Semi-conservative replication: DNA replication in which new DNA is replicated from an existing DNA template strand.
Sequencing: A molecular laboratory procedure that produces the order of nucleotide bases (i.e., sequences).
Sister chromatids: During DNA replication, sister chromatids are produced on the chromosome. In cell division, sister chromatids are pulled apart so that two cells can be formed. In meiosis, sister chromatids are also the sites of genetic recombination.
Somatic cells: Diploid cells that comprise body tissues and undergo mitosis for maintenance and repair of tissues.
Splicing: The process by which mature mRNAs are produced. Introns are removed (spliced) and exons are joined together.
Sugar phosphate backbone: A biochemical structural component of DNA. The “backbone” consists of deoxyribose sugars and phosphate molecules.
Telomere: A compound structure located at the ends of chromosomes to help protect the chromosomes from degradation after every round of cell division.
Termination: The halt of DNA replication activity that occurs when a DNA sequence “stop” codon is encountered.
Tissue: A cluster of cells that are morphologically similar and perform the same task.
Transcription: The process by which DNA nucleotides (within a gene) are copied, which results in a messenger RNA molecule.
Transcription factors: Proteins that bind to regulatory regions of genes (e.g., promoter) and increase or decrease the amount of transcriptional activity of a gene, including turning them “on” or “off.”
Transfer RNA (tRNA): RNA molecule involved in translation. Transfer RNA transports amino acids from the cell’s cytoplasm to a ribosome.
Translation: The process by which messenger RNA codons are read and amino acids are “chained together” to form proteins.
X-linked: Refers to a pattern of inheritance where the allele is located on the X or Y chromosome.
For Further Exploration
National Human Genome Research Institute
NOVA. 2018. Gene Sequencing Speeds Diagnosis of Deadly Newborn Diseases. NOVA, March 7, 2018. Accessed January 31, 2023. https://www.pbs.org/wgbh/nova/next/body/newborn-gene-sequencing/.
Zimmer, Carl. N.d. “Carl Zimmer’s Game of Genomes.” STATnews. Accessed January 31, 2023. https://www.statnews.com/feature/game-of-genomes/season-one/.
Illumina. 2016. “Illumina Sequencing by Synthesis.” YouTube.com, October 5, 2016. Accessed January 31, 2023. https://www.youtube.com/watch?v=fCd6B5HRaZ8.
References
Aartsma-Rus, Annemieke, Ieke B. Ginjaar, and Kate Bushby. 2016. “The Importance of Genetic Diagnosis for Duchenne Muscular Dystrophy.” Journal of Medical Genetics 53 (3): 145–151.
Acuna-Hidalgo, Rocio, Joris A. Veltman, and Alexander Hoischen. 2016. “New Insights into the Generation and Role of De Novo Mutations in Health and Disease.” Genome Biology 17 (241): 1–19.
Albert, Benjamin, Susanna Tomassetti, Yvonne Gloor, Daniel Dilg, Stefano Mattarocci, Slawomir Kubik, Lukas Hafner, and David Shore. 2019. "Sfp1 Regulates Transcriptional Networks Driving Cell Growth and Division through Multiple Promoter-Binding Modes." Genes & Development 33 (5–6): 288–293.
Almathen, Faisal, Haitham Elbir, Hussain Bahbahani, Joram Mwacharo, and Olivier Hanotte. 2018. “Polymorphisms in Mc1r and Asip Genes Are Associated with Coat Color Variation in the Arabian Camel.” Journal of Heredity 109 (6): 700–706.
Ballester, Leomar Y., Rajyalakshmi Luthra, Rashmi Kanagal-Shamanna, and Rajesh R. Singh. 2016. “Advances in Clinical Next-Generation Sequencing: Target Enrichment and Sequencing Technologies.” Expert Review of Molecular Diagnostics 16 (3): 357–372.
Baranovskiy, Andrey G., Vincent N. Duong, Nigar D. Babayeva, Yinbo Zhang, Youri I. Pavlov, Karen S. Anderson, and Tahir H. Tahirov. 2018. “Activity and Fidelity of Human DNA Polymerase Alpha Depend on Primer Structure.” Journal of Biological Chemistry 293 (18): 6824–6843.
Biswas, S., Ghosh, S., Das, S., & Maitra, S. (2021). Female Reproduction: At the Crossroads of Endocrine Disruptors and Epigenetics. Proceedings of the Zoological Society, 74(4), 532–545. https://doi.org/10.1007/s12595-021-00403-4
Brezina, Paulina R., Raymond Anchan, and William G. Kearns. 2016. “Preimplantation Genetic Testing for Aneuploidy: What Technology Should You Use and What Are the Differences?” Journal of Assisted Reproduction and Genetics 33 (7): 823–832.
Bultman, Scott J. 2017. “Interplay Between Diet, Gut Microbiota, Epigenetic Events, and Colorectal Cancer." Molecular Nutrition & Food Research 61 (1):1–12.
Cutting, Garry R. 2015. “Cystic Fibrosis Genetics: From Molecular Understanding to Clinical Application.” Nature Reviews Genetics 16 (1): 45–56.
D'Alessandro, Giuseppina., and Fabrizio d'Adda di Fagagna. 2017. “Transcription and DNA Damage: Holding Hands or Crossing Swords?” Journal of Molecular Biology 429 (21): 3215–3229.
De Craene, Johan-Owen, Dimitri L. Bertazzi, Séverine Bar, and Sylvie Friant. 2017. “Phosphoinositides, Major Actors in Membrane Trafficking and Lipid Signaling Pathways.” International Journal of Molecular Sciences 18 (3): 1–20.
Deng, Lian, and Shuhua Xu. 2018. “Adaptation of Human Skin Color in Various Populations.” Hereditas 155 (1): 1–12.
Dever, Thomas E., Terri G. Kinzy, and Graham D. Pavitt. 2016. “Mechanism and Regulation of Protein Synthesis in Saccharomyces Cerevisiae.” Genetics 203 (1): 65–107.
Eme, Laura, Anja Spang, Jonathan Lombard, Courtney W. Stairs, and Thijs J. G. Ettema. 2017. “Archaea and the Origin of Eukaryotes.” Nature Reviews Microbiology 15 (12): 711–723.
Gomez-Carballa, Alberto, Jacobo Pardo-Seco, Stefania Brandini, Alessandro Achilli, Ugo A. Perego, Michael D. Coble, Toni M. Diegoli, et al. 2018. “The Peopling of South America and the Trans-Andean Gene Flow of the First Settlers.” Genome Research 28 (6): 767–779.
Gvozdenov, Zlata, Janhavi Kolhe, and Brian C. Freeman. 2019. “The Nuclear and DNA-Associated Molecular Chaperone Network.” Cold Spring Harbor Perspectives in Biology 11 (10): a034009.
Harkins, Kelly M., and Anne C. Stone. 2015. “Ancient Pathogen Genomics: Insights into Timing and Adaptation.” Journal of Human Evolution 79: 137–149.
Jackson, Maria, Leah Marks, Gerhard H. W. May, and Joanna B. Wilson. 2018. “The Genetic Basis of Disease.” Essays in Biochemistry 62 (5): 643–723.
Lenormand, Thomas, Jan Engelstadter, Susan E. Johnston, Erik Wijnker, and Christopher R. Haag. 2016. “Evolutionary Mysteries in Meiosis.” Philosophical Transactions of the Royal Society B 371: 1–14.
Levy, Shawn E., and Richard M. Myers. 2016. “Advancements in Next-Generation Sequencing.” Annual Review of Genomics and Human Genetics 17: 95–115.
Lindo, John, Emilia Huerta-Sánchez, Shigeki Nakagome, Morten Rasmussen, Barbara Petzelt, Joycelynn Mitchell, Jerome S. Cybulski, et al. 2016. "A Time Transect of Exomes from a Native American Population Before and After European Contact." Nature Communications 7: 1–11. https://doi.org/10.1038/ncomms13175.
Lu, Mengfei, Cathryn M. Lewis, and Matthew Traylor. 2017. “Pharmacogenetic Testing through the Direct-to-Consumer Genetic Testing Company 23andme.” BMC Medical Genomics 10 (47): 1–8.
Ly, Lundi, Donovan Chan, Mahmoud Aarabi, Mylene Landry, Nathalie A. Behan, Amanda J. MacFarlane, and Jacquetta Trasler. 2017. “Intergenerational Impact of Paternal Lifetime Exposures to Both Folic Acid Deficiency and Supplementation on Reproductive Outcomes and Imprinted Gene Methylation.” Molecular Human Reproduction 23 (7): 461–477.
Ma, Wenxiu, Giancarlo Bonora, Joel B. Berletch, Xinxian Deng, William S. Noble, and Christine M. Disteche. 2018. “X-Chromosome Inactivation and Escape from X Inactivation in Mouse.” Methods in Molecular Biology 1861: 205–219.
Machiela, Mitchell J., Weiyin Zhou, Eric Karlins, Joshua N. Sampson, Neal D. Freedman, Qi Yang, Belynda Hicks, et al. 2016. “Female Chromosome X Mosaicism Is Age-Related and Preferentially Affects the Inactivated X Chromosome.” Nature Communications 7: 1–9. https://doi.org/10.1038/ncomms11843.
Mahdavi, Morteza, Mohammadreza Nassiri, Mohammad M. Kooshyar, Masoume Vakili-Azghandi, Amir Avan, Ryan Sandry, Suja Pillai, Alfred K. Lam, and Vinod Gopalan. 2019. “Hereditary Breast Cancer; Genetic Penetrance and Current Status with BRCA.” Journal of Cellular Physiology 234 (5): 5741–5750.
McDade, Thomas W., Calen P. Ryan, Meaghan J. Jones, Morgan K. Hoke, Judith Borja, Gregory E. Miller, Christopher W. Kuzawa, and Michael S. Kobor. 2019. “Genome-Wide Analysis of DNA Methylation in Relation to Socioeconomic Status During Development and Early Adulthood.” American Journal of Physical Anthropology 169 (1): 3–11.
Migeon, Barbara R. 2017. “Choosing the Active X: The Human Version of X Inactivation.” Trends in Genetics 33 (12): 899–909.
Myerowitz, Rachel. 1997. “Tay-Sachs Disease-Causing Mutations and Neutral Polymorphisms in the Hex A Gene.” Human Mutation 9 (3): 195–208.
National Institute of Environmental Health Sciences. (2024, July 22). Endocrine Disruptors. National Institute of Environmental Health Sciences; United States Government. https://www.niehs.nih.gov/health/topics/agents/endocrine
Onufriev, Alexey V., and Helmut Schiessel. 2019. “The Nucleosome: From Structure to Function through Physics.” Current Opinion in Structural Biology 56: 119–130.
Peedicayil J. (2006). Epigenetic therapy--a new development in pharmacology. The Indian journal of medical research, 123(1), 17–24.
Quillen, Ellen E., Heather L. Norton, Esteban J. Parra, Frida Lona-Durazo, Khai C. Ang, Florin M. Illiescu, Laurel N. Pearson, et al. 2019. “Shades of Complexity: New Perspectives on the Evolution and Genetic Architecture of Human Skin.” American Journal of Physical Anthropology 168 (67): 4–26.
Raspelli, Erica, and Roberta Fraschini. 2019. “Spindle Pole Power in Health and Disease.” Current Genetics 65 (4): 851–855.
Ravinet, M., R. Faria, R. K. Butlin, J. Galindo, N. Bierne, M. Rafajlovic, M. A. F. Noor, B. Mehlig, and A. M. Westram. 2017. “Interpreting the Genomic Landscape of Speciation: A Road Map for Finding Barriers to Gene Flow.” Journal of Evolutionary Biology 30 (8): 1450–1477.
Regev, Aviv, Sarah A. Teichmann, Eric S. Lander, Ido Amit, Christophe Benoist, Ewan Birney, Bernd Bodenmiller, et al. 2017. “The Human Cell Atlas.” Elife 6e27041: 1–30. https://doi.org/10.7554.eLife.27041.
Roberts, Andrea L., Nicole Gladish, Evan Gatev, Meaghan J. Jones, Ying Chen, Julia L. MacIsaac, Shelley S. Tworoger, et al. 2018. “Exposure to Childhood Abuse Is Associated with Human Sperm DNA Methylation.” Translational Psychiatry 8 (194): 1–11.
Roger, Andrew J., Sergio A. Muñoz-Gómez, and Ryoma Kamikawa. 2017. “The Origin and Diversification of Mitochondria.” Current Biology 27 (21): R1177–R1192. https://www.sciencedirect.com/science/article/pii/S096098221731179X?via%3Dihub#!
Ségurel, Laure, and Céline Bon. 2017. “On the Evolution of Lactase Persistence in Humans.” Annual Review of Genomics and Human Genetics 18: 297–319.
Sheth, Bhavisha P., and Vrinda S. Thaker. 2017. “DNA Barcoding and Traditional Taxonomy: An Integrated Approach for Biodiversity Conservation.” Genome 60 (7): 618–628.
Skloot, Rebecca. 2010. The Immortal Life of Henrietta Lacks. New York: Crown Publishing Group.
Snedeker, Jonathan, Matthew Wooten, and Xin Chen. 2017. “The Inherent Asymmetry of DNA Replication.” Annual Review of Cell and Developmental Biology 33: 291–318.
Sullivan-Pyke, Chantae, and Anuja Dokras. 2018. “Preimplantation Genetic Screening and Preimplantation Genetic Diagnosis.” Obstetrics and Gynecology Clinics of North America 45 (1): 113–125.
Sweeney, M. F., Hasan, N., Soto, A. M., & Sonnenschein, C. (2015). Environmental endocrine disruptors: Effects on the human male reproductive system. Reviews in Endocrine and Metabolic Disorders, 16(4), 341–357. https://doi.org/10.1007/s11154-016-9337-4
Szostak, Jack W. 2017. “The Narrow Road to the Deep Past: In Search of the Chemistry of the Origin of Life.” Angewandte Chemie International Edition 56 (37): 11037–11043.
Tessema, Mathewos, Ulrich Lehmann, and Hans Kreipe. 2004. “Cell Cycle and No End.” Virchows Archiv European Journal of Pathology 444 (4): 313–323.
Tishkoff, Sarah A., Floyd A. Reed, Alessia Ranciaro, Benjamin F. Voight, Courtney C. Babbitt, Jesse S. Silverman, Kweli Powell, et al. 2007. “Convergent Adaptation of Human Lactase Persistence in Africa and Europe.” Nature Genetics 39 (1): 31–40.
Visootsak, Jeannie, and John M. Graham, Jr. 2006. “Klinefelter Syndrome and Other Sex Chromosomal Aneuploidies.” Orphanet Journal of Rare Diseases 1:42. https://doi.org/10.1186/1750-1172-1-42.
Wolfe, George C., dir. 2017. The Immortal Life of Henrietta Lacks. HBO Films, April 22, 2017. TV Movie.
Yamamoto, Fumi-ichiro, Henrik Clausen, Thayer White, John Marken, and Sen-itiroh Hakomori. 1990. “Molecular Genetic Basis of the Histo-Blood Group ABO System.” Nature 345 (6272): 229–233.
Yu, X., Zhao, H., Wang, R., Chen, Y., Ouyang, X., Li, W., Sun, Y., & Peng, A. (2024). Cancer epigenetics: from laboratory studies and clinical trials to precision medicine. Cell Death Discovery, 10(1), 1–12. https://doi.org/10.1038/s41420-024-01803-z
Zlotogora, Joël. 2003. “Penetrance and Expressivity in the Molecular Age.” Genetics in Medicine 5 (5): 347–352.
Zorina-Lichtenwalter, Katerina, Ryan N. Lichtenwalter, Dima V. Zaykin, Marc Parisien, Simon Gravel, Andrey Bortsov, and Luda Diatchenko. 2019. “A Study in Scarlet: MC1R as the Main Predictor of Red Hair and Exemplar of the Flip-Flop Effect.” Human Molecular Genetics 28 (12): 2093-2106.
Zwart, Haeh. 2018. “In the Beginning Was the Genome: Genomics and the Bi-Textuality of Human Existence.” New Bioethics 24 (1): 26–43.
Andrea J. Alveshere, Ph.D., Western Illinois University
Student contributors for this chapter: Corin Laberge, Hazel Moorcroft, Isabella Michel, Julian J. Pantoja Quiroz
This chapter is a revision from "Chapter 4: Forces of Evolution” by Andrea J. Alveshere. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Outline a 21st-century perspective of the Modern Synthesis.
- Define populations and population genetics as well as the methods used to study them.
- Identify the forces of evolution and become familiar with examples of each.
- Discuss the evolutionary significance of mutation, genetic drift, gene flow, and natural selection.
- Explain how allele frequencies can be used to study evolution as it happens.
- Contrast micro- and macroevolution.
It’s hard for us, with our typical human life spans of less than 100 years, to imagine all the way back, 3.8 billion years ago, to the origins of life. Scientists still study and debate how life came into being and whether it originated on Earth or in some other region of the universe (including some scientists who believe that studying evolution can reveal the complex processes that were set in motion by God or a higher power). What we do know is that a living single-celled organism was present on Earth during the early stages of our planet’s existence. This organism had the potential to reproduce by making copies of itself, just like bacteria, many amoebae, and our own living cells today. In fact, with modern technologies, we can now trace genetic lineages, or phylogenies, and determine the relationships between all of today’s living organisms—eukaryotes (animals, plants, fungi, etc.), archaea, and bacteria—on the branches of the phylogenetic tree of life (Figure 5.1).
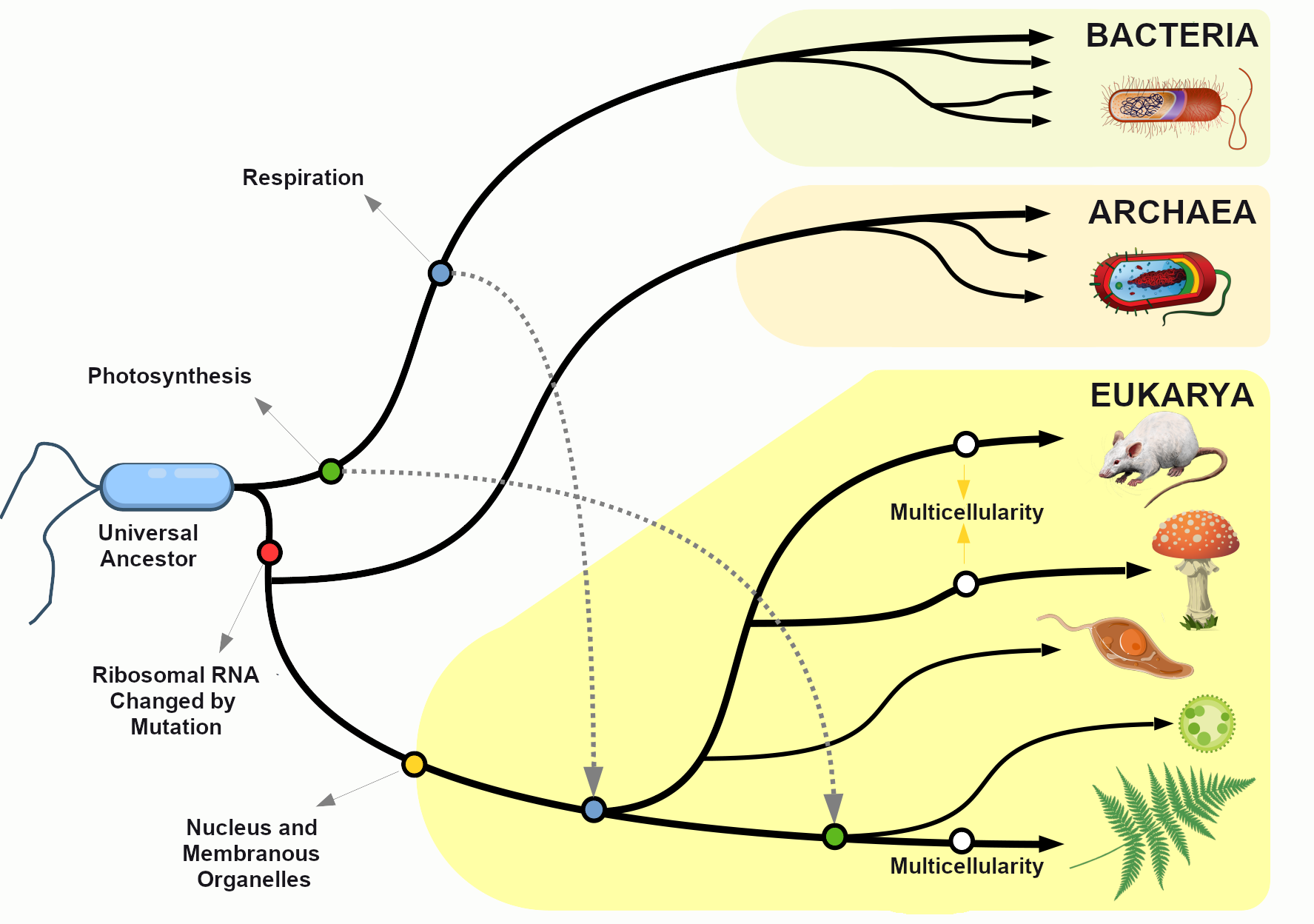
Looking at the common sequences in modern genomes, we can even make educated guesses about the likely genetic sequence of the Last Universal Common Ancestor (LUCA) of all living things. Through a wondrous series of mechanisms and events over nearly four billion years, that ancient single-celled organism gave rise to the rich diversity of species that fill the lands, seas, and skies of our planet. This chapter explores the mechanisms by which that amazing transformation occurred and considers some of the crucial scientific experiments that shaped our current understanding of the evolutionary process.
Population Genetics
Defining Populations and the Variations within Them
One of the major breakthroughs in understanding the mechanisms of evolutionary change came with the realization that evolution takes place at the level of populations, not within individuals. In the biological sciences, a population is defined as a group of individuals of the same species who are geographically near enough to one another that they can breed and produce new generations of individuals.
For the purpose of studying evolution, we recognize populations by their even smaller units: genes. Remember, a gene is the basic unit of information that encodes the proteins needed to grow and function as a living organism. Each gene can have multiple alleles, or variants—each of which may produce a slightly different protein. Each individual, for genetic inheritance purposes, carries a collection of genes that can be passed down to future generations. For this reason, in population genetics, we think of populations as gene pools, which refers to the entire collection of genetic material in a breeding community that can be passed on from one generation to the next.
For genes carried on our human chromosomes (our nuclear DNA), we inherit two copies of each, one from each parent. This means we may carry two of the same alleles (a homozygous genotype) or two different alleles (a heterozygous genotype) for each nuclear gene.
Defining Evolution
In order to understand evolution, it’s crucial to remember that evolution is always studied at the population level. Also, if a population were to stay exactly the same from one generation to the next, it would not be evolving. So evolution requires both a population of breeding individuals and some kind of a genetic change occurring within it. Thus, the simple definition of evolution is a change in the allele frequencies in a population over time. What do we mean by allele frequencies? Allele frequencies refer to the ratio, or percentage, of one allele (one variant of a gene) compared to the other alleles for that gene within the study population (Figure 5.2). By contrast, genotype frequencies are the ratios or percentages of the different homozygous and heterozygous genotypes in the population. Because we carry two alleles per genotype, the total count of alleles in a population will usually be exactly double the total count of genotypes in the same population (with the exception being rare cases in which an individual carries a different number of chromosomes than the typical two; e.g., Down syndrome results when a child carries three copies of Chromosome 21).
The Forces of Evolution
Today, we recognize that evolution takes place through a combination of mechanisms: mutation, genetic drift, gene flow, and natural selection. These mechanisms are called the “forces of evolution”; together they account for all the genotypic variation observed in the world today. Keep in mind that each of these forces was first defined and then tested—and retested—through the experimental work of the many scientists who contributed to the Modern Synthesis.
Mutation
The first force of evolution we will discuss is mutation, and for good reason: mutation is the original source of all the genetic variation found in every living thing. Imagine all the way back in time to the very first single-celled organism, floating in Earth’s primordial sea. Based on what we observe in simple, single-celled organisms today, that organism probably spent its lifetime absorbing nutrients and dividing to produce cloned copies of itself. While the numbers of individuals in that population would have grown (as long as the environment was favorable), nothing would have changed in that perfectly cloned population. There would not have been variety among the individuals. It was only through a copying error—the introduction of a mutation, or change, into the genetic code—that new alleles were introduced into the population.
After many generations have passed in our primordial population, mutations have created distinct chromosomes. The cells are now amoeba-like, larger than many of their tiny bacterial neighbors, who have long since become their favorite source of nutrients. Without mutation to create this diversity, all living things would still be identical to LUCA, our universal ancestor (Figure 5.3).
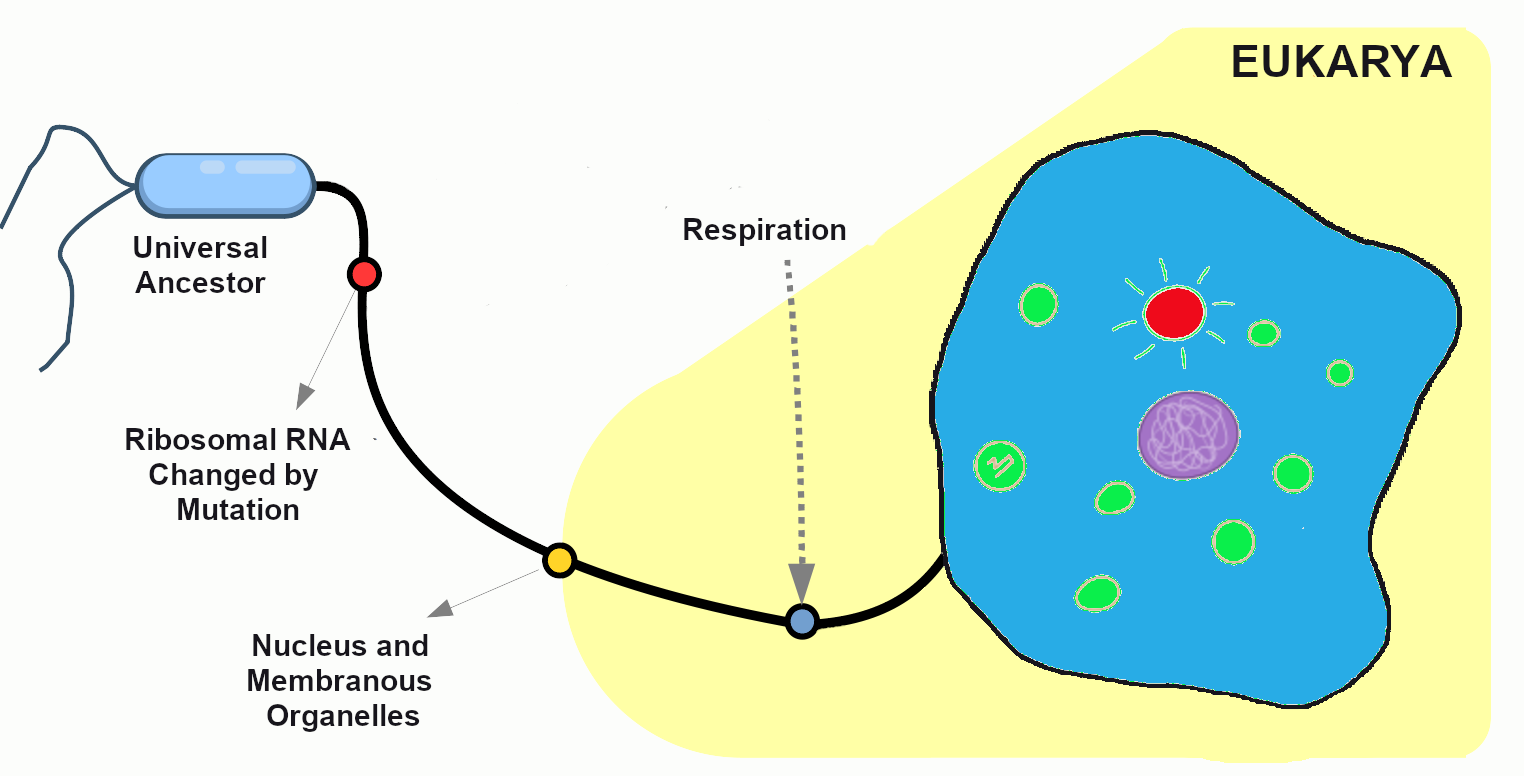
When we think of genetic mutation, we often first think of deleterious mutations—the ones associated with negative effects such as the beginnings of cancers or heritable disorders. The fact is, though, that every genetic adaptation that has helped our ancestors survive since the dawn of life is directly due to beneficial mutations—changes in the DNA that provided some sort of advantage to a given population at a particular moment in time. For example, a beneficial mutation allowed chihuahuas and other tropical-adapted dog breeds to have much thinner fur coats than their cold-adapted cousins the northern wolves, malamutes, and huskies.
Every one of us has genetic mutations. Yes, even you. The DNA in some of your cells today differs from the original DNA that you inherited when you were a tiny, fertilized egg. Mutations occur all the time in the cells of our skin and other organs, due to chemical changes in the nucleotides. Exposure to the UV radiation in sunlight is one common cause of skin mutations. Interaction with UV light causes UV crosslinking, in which adjacent thymine bases bind with one another (Figure 5.4). Many of these mutations are detected and corrected by DNA repair mechanisms, enzymes that patrol and repair DNA in living cells, while other mutations may cause a new freckle or mole or, perhaps, an unusual hair to grow. For people with the autosomal recessive disease xeroderma pigmentosum, these repair mechanisms do not function correctly, resulting in a host of problems especially related to sun exposure, including severe sunburns, dry skin, heavy freckling, and other pigment changes.
Most of our mutations exist in somatic cells, which are the cells of our organs and other body tissues. Those will not be passed onto future generations and so will not affect the population over time. Only mutations that occur in the gametes, the reproductive cells (i.e., the sperm or egg cells), will be passed onto future generations. When a new mutation pops up at random in a family lineage, it is known as a spontaneous mutation. If the individual born with this spontaneous mutation passes it on to his offspring, those offspring receive an inherited mutation. Geneticists have identified many classes of mutations and the causes and effects of many of these.
Point Mutations
A point mutation is a single-letter (single-nucleotide) change in the genetic code resulting in the substitution of one nucleic acid base for a different one. As you learned in Chapter 3, the DNA code in each gene is translated through three-letter “words” known as codons. So depending on how the point mutation changes the “word,” the effect it will have on the protein may be major or minor or may make no difference at all.
If a mutation does not change the resulting protein, then it is called a synonymous mutation. Synonymous mutations do involve a letter (nucleic acid) change, but that change results in a codon that codes for the same “instruction” (the same amino acid or stop code) as the original codon. Mutations that do cause a change in the protein are known as nonsynonymous mutations. Nonsynonymous mutations may change the resulting protein’s amino acid sequence by altering the DNA sequence that encodes the mRNA or by changing how the mRNA is spliced prior to translation (refer to Chapter 3 for more details).
Insertions and Deletions
In addition to point mutations, another class of mutations are insertions and deletions, or indels, for short. As the name suggests, these involve the addition (insertion) or removal (deletion) of one or more coding sequence letters (nucleic acids). These typically first occur as an error in DNA replication, wherein one or more nucleotides are either duplicated or skipped in error. Entire codons or sets of codons may also be removed or added if the indel is a multiple of three nucleotides.
Frameshift mutations are types of indels that involve the insertion or deletion of any number of nucleotides that is not a multiple of three (e.g., adding one or two extra letters to the code). Because these indels are not consistent with the codon numbering, they “shift the reading frame,” causing all the codons beyond the mutation to be misread. Like point mutations, small indels can also disrupt splice sites.
Transposable elements, or transposons, are fragments of DNA that can “jump” around in the genome. There are two types of transposons: retrotransposons are transcribed from DNA into RNA and then “reverse transcribed,” to insert the copied sequence into a new location in the DNA, and DNA transposons, which do not involve RNA. DNA transposons are clipped out of the DNA sequence itself and inserted elsewhere in the genome. Because transposable elements insert themselves into existing DNA sequences, they are frequent gene disruptors. At certain times, and in certain species, it appears that transposons became very active, likely accelerating the mutation rate (and thus, the genetic variation) in those populations during the active periods.
Chromosomal Alterations
The final major category of genetic mutations are changes at the chromosome level: crossover events, nondisjunction events, and translocations. Crossover events occur when DNA is swapped between homologous chromosomes while they are paired up during meiosis I. Crossovers are thought to be so common that some DNA swapping may happen every time chromosomes go through meiosis I. Crossovers don’t necessarily introduce new alleles into a population, but they do make it possible for new combinations of alleles to exist on a single chromosome that can be passed to future generations. This also enables new combinations of alleles to be found within siblings who share the same parents. Also, if the fragments that cross over don’t break at exactly the same point, they can cause genes to be deleted from one of the homologous chromosomes and duplicated on the other.
Nondisjunction events occur when the homologous chromosomes (in meiosis I) or sister chromatids (in meiosis II and mitosis) fail to separate after pairing. The result is that both chromosomes or chromatids end up in the same daughter cell, leaving the other daughter cell without any copy of that chromosome (Figure 5.5). Most nondisjunctions at the gamete level are fatal to the embryo. The most widely known exception is Trisomy 21, or Down syndrome, which results when an embryo inherits three copies of Chromosome 21: two from one parent (due to a nondisjunction event) and one from the other (Figure 5.6). Trisomies (triple chromosome conditions) of Chromosomes 18 (Edwards syndrome) and 13 (Patau syndrome) are also known to result in live births, but the children usually have severe complications and rarely survive beyond the first year of life.
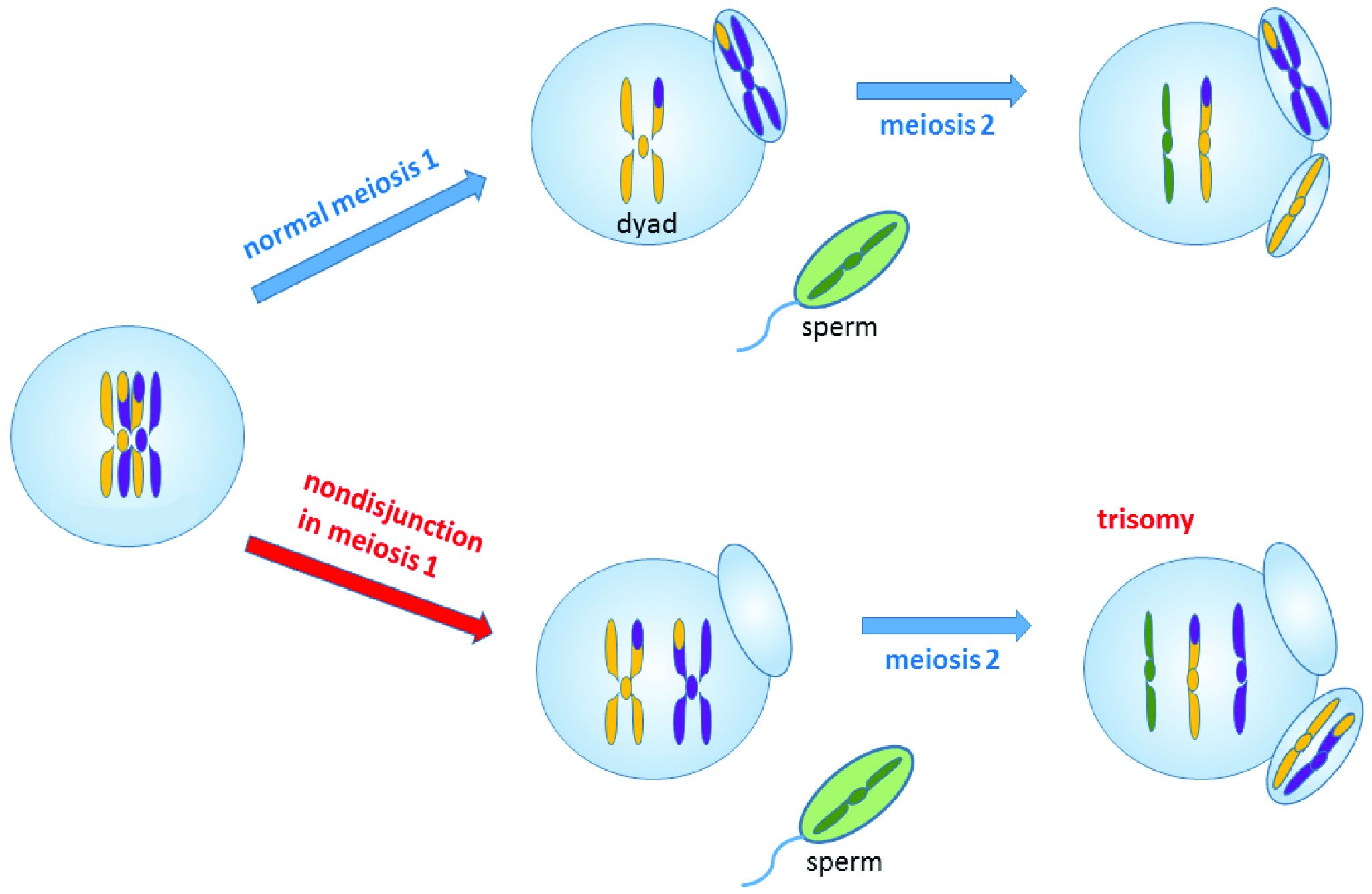

Sex chromosome trisomies (XXX, XXY, XYY) and X chromosome monosomies (inheritance of an X chromosome from one parent and no sex chromosome from the other) are also survivable and fairly common. The symptoms vary but often include atypical sexual characteristics, either at birth or at puberty, and often result in sterility. The X chromosome carries unique genes that are required for survival; therefore, Y chromosome monosomies are incompatible with life.
Chromosomal translocations involve transfers of DNA between nonhomologous chromosomes. This may involve swapping large portions of two or more chromosomes. The exchanges of DNA may be balanced or unbalanced. In balanced translocations, the genes are swapped, but no genetic information is lost. In unbalanced translocations, there is an unequal exchange of genetic material, resulting in duplication or loss of genes. Translocations result in new chromosomal structures called derivative chromosomes, because they are derived or created from two different chromosomes. Translocations are often found to be linked to cancers and can also cause infertility. Even if the translocations are balanced in the parent, the embryo often won’t survive unless the baby inherits both of that parent’s derivative chromosomes (to maintain the balance).
Genetic Drift
The second force of evolution is commonly known as genetic drift. This is an unfortunate misnomer, as this force actually involves the drifting of alleles, not genes. Genetic drift refers to random changes (“drift”) in allele frequencies from one generation to the next. The genes are remaining constant within the population; it is only the alleles of the genes that are changing in frequency. The random nature of genetic drift is a crucial point to understand: it specifically occurs when none of the variant alleles confer an advantage.
Let’s imagine far back in time, again, to that ancient population of amoeba-like cells, subsisting and occasionally dividing, in the primordial sea. A mutation occurs in one of the cells that changes the texture of the cell membrane from a relatively smooth surface to a highly ruffled one (Figure 5.7). This has absolutely no effect on the cell’s quality of life or ability to reproduce. In fact, eyes haven’t evolved yet, so no one in the world at the time would even notice the difference. The cells in the population continue to divide, and the offspring of the ruffled cell inherit the ruffled membrane. The frequency (percentage) of the ruffled allele in the population, from one generation to the next, will depend entirely on how many offspring that first ruffled cell ends up having, and the random events that might make the ruffled alleles more common or more rare (such as population bottlenecks and founder effects, which are discussed below).
Sexual Reproduction and Random Inheritance
Tracking alleles gets a bit more complicated in our primordial cells when, after a number of generations, a series of mutations have created populations that reproduce sexually. These cells now must go through an extra round of cell division (meiosis) to create haploid gametes. The combination of two gametes is now required to produce each new diploid offspring.
In the earlier population, which reproduced via asexual reproduction, a cell either carried the smooth allele or the ruffled allele. With sexual reproduction, a cell inherits one allele from each parent, so there are homozygous cells that contain two smooth alleles, homozygous cells that contain two ruffled alleles, and heterozygous cells that contain one of each allele (Figure 5.8). If the new, ruffled allele happens to be dominant (and we’ll imagine that it is), the heterozygotes will have ruffled cell phenotypes but also will have a 50/50 chance of passing on a smooth allele to each offspring. As long as neither phenotype (ruffled nor smooth) provides any advantage over the other, the variation in the population from one generation to the next will remain completely random.
In sexually reproducing populations (including humans and many other animals and plants in the world today), that 50/50 chance of inheriting one or the other allele from each parent plays a major role in the random nature of genetic drift.
Population Bottlenecks
A population bottleneck occurs when the number of individuals in a population drops dramatically due to some random event. The most obvious, familiar examples are natural disasters. Tsunamis and hurricanes devastating island and coastal populations and forest fires and river floods wiping out populations in other areas are all too familiar. When a large portion of a population is randomly wiped out, the allele frequencies (i.e., the percentages of each allele) in the small population of survivors are often much different from the frequencies in the predisaster, or “parent,” population.
If such an event happened to our primordial ocean cell population—perhaps a volcanic fissure erupted in the ocean floor and only the cells that happened to be farthest from the spewing lava and boiling water survived—we might end up, by random chance, with a surviving population that had mostly ruffled alleles, in contrast to the parent population, which had only a small percentage of ruffles (Figure 5.9).
One of the most famous examples of a population bottleneck is the prehistoric disaster that led to the extinction of dinosaurs, the Cretaceous–Paleogene extinction event (often abbreviated K–Pg; previously K-T). This occurred approximately 66 million years ago. Dinosaurs and all their neighbors were going about their ordinary routines when a massive asteroid zoomed in from space and crashed into what is now the Gulf of Mexico, creating an impact so enormous that populations within hundreds of miles of the crash site were likely immediately wiped out. The skies filled with dust and debris, causing temperatures to plummet worldwide. It’s estimated that 75% of the world’s species went extinct as a result of the impact and the deep freeze that followed (Jablonski and Chaloner 1994).

The populations that emerged from the K-Pg extinction were markedly different from their pre-disaster communities. Surviving mammal populations expanded and diversified, and other new creatures appeared. The ecosystems of Earth were filled with new organisms and have never been the same (Figure 5.10).
Much more recently in geological time, during the colonial period, many human populations experienced bottlenecks as a result of the fact that imperial powers were inclined to slaughter communities who were reluctant to give up their lands and resources. This effect was especially profound in the Americas, where Indigenous populations faced the compounded effects of brutal warfare, exposure to new bacteria and viruses (against which they had no immunity), and ultimately segregation on resource-starved reservations. The populations in Europe, Asia, and Africa had experienced regular gene flow during the 10,000-year period in which most kinds of livestock were being domesticated, giving them many generations of experience building up immunity against zoonotic diseases (those that can pass from animals to humans). In contrast, the residents of the Americas had been almost completely isolated during those millennia, so all these diseases swept through the Americas in rapid succession, creating a major loss of genetic diversity in the Indigenous American population. It is estimated that between 50% and 95% of the Indigenous American populations died during the first decades after European contact, around 500 years ago (Livi-Bacci 2006).
An urgent health challenge facing humans today involves human-induced population bottlenecks that produce antibiotic-resistant bacteria. Antibiotics are medicines prescribed to treat bacterial infections. The typical prescription includes enough medicine for ten days. People often feel better much sooner than ten days and sometimes decide to quit taking the medicine ahead of schedule. This is often a big mistake. The antibiotics have quickly killed off a large percentage of the bacteria—enough to reduce the symptoms and make you feel much better. However, this has created a bacterial population bottleneck. There are usually a small number of bacteria that survive those early days. If you take the medicine as prescribed for the full ten days, it’s quite likely that there will be no bacterial survivors. If you quit early, though, the survivors—who were the members of the original population who were most resistant to the antibiotic—will begin to reproduce again. Soon the infection will be back, possibly worse than before, and now all of the bacteria are resistant to the antibiotic that you had been prescribed.
Other activities that have contributed to the rise of antibiotic-resistant bacteria include the use of antibacterial cleaning products and the inappropriate use of antibiotics as a preventative measure in livestock or to treat infections that are viral instead of bacterial (viruses do not respond to antibiotics). In 2017, the World Health Organization published a list of twelve antibiotic-resistant pathogens that are considered top priority targets for the development of new antibiotics (World Health Organization 2017).
Dig Deeper: The North American Elephant Seal: Thriving Bottleneck Populations That Still Face Genetic Defects
In 1892, the Northern Elephant Seal underwent a severe population bottleneck caused by commercial hunting, reducing the species to an estimated 20 individuals at the time. This drastic decline led to a substantial loss of genetic diversity–a common consequence of extreme population bottlenecks (Hoelzel et al., 2024 & Weber et al., 2000). While the population has since recovered to over 200,000 individuals, its genetic variability remains significantly low. Analyses of genetic markers, including allozymes, mitochondrial DNA, and microsatellites, consistently reflect this reduced diversity (Hoelzel et al., 2024). Comparative studies further underscore this loss by highlighting the higher genetic variation observed in the Southern Elephant Seal, which did not experience similar population constraints (2024).

In a 2024 study for Nature, Ecology, and Evolution, Hoelzel and colleagues sequenced 260 modern and 8 historical genomes of the northern elephant seal. This comparison revealed a decrease in average heterozygosity from 0.00142 before the bottleneck to 0.000176 in the contemporary population, confirming the decline in genetic variation (2024). Hoelzel’s mitogenome tree further illustrates this loss, revealing only two significant lineages remaining post-bottleneck, with limited diversity within each. Among the issues of diversity, the population has shown an increased number of loss-of-function (LOF) alleles, suggesting that increased inbreeding has amplified the frequency of these detrimental alleles; this reduced genetic diversity negatively affects both male and female reproductive fitness. Females who practiced repetitive inbreeding had higher LOF alleles and subsequently weaned fewer pups per year over their lifetime, while male reproductive success was linked to specific LOF loci associated with sperm production (2024). Hoelzel uses the example of “Alpha-Male M12”–known for low paternity success despite frequent copulations–which was homozygous for non-functional versions of four out of five LOF loci related to sperm function (2024, p. 688). The species' mating system, characterized by extreme polygyny, further exacerbates the loss of genetic variation even with countless copulatory partners
Prior research published in Current Biology presents an empirical genetic assessment of this population bottleneck, highlighting its long-term genetic consequences, particularly the loss of mitochondrial diversity (Weber et al., 2000). In this research, Weber and colleagues note that random lineage sampling during the bottleneck led to the persistence of specific genetic variants by chance rather than through natural selection (2000). This research emphasizes that the loss of diversity poses potential future genetic vulnerabilities for the seals, and that further studies are crucial for understanding the full scope of these impacts on the seals' overall fitness (2000). In 2024, the work led by Hoelzen and company provided the missing data that the previous study had left unanswered. Their previously explored findings indicate that, although the seals have recovered in numbers, their genetic resilience remains compromised, leaving the population more vulnerable to future environmental pressures, such as climate change or resource shortages (Hoelzel et al., 2024). Ultimately, while the population's size remains stable, the genetic consequences of the bottleneck indicate that past stochastic events continue to influence the seals' long-term fitness and adaptability.
This research indicates that the historical bottleneck continues to affect the seals' health and fitness, despite the population's recovery. Limited genetic diversity and the persistence of harmful alleles due to inbreeding have continued to handicap the species' ability to thrive in environmental challenges such as climate change and resource fluctuations (2024). This emphasizes the importance of incorporating genetic factors into conservation strategies, as populations that have rebounded may still harbour long-term genetic weaknesses. Moreover, the elephant seal’s history serves as a powerful example of how human actions —such as overhunting — can have long-lasting impacts on biodiversity, reinforcing the importance of understanding human-environment interactions in ecological and conservation contexts.
Founder Effects
Founder effects occur when members of a population leave the main or “parent” group and form a new population that no longer interbreeds with the other members of the original group. Similar to survivors of a population bottleneck, the newly founded population often has allele frequencies that are different from the original group. Alleles that may have been relatively rare in the parent population can end up being very common due to the founder effect. Likewise, recessive traits that were seldom seen in the parent population may be seen frequently in the descendants of the offshoot population.
One striking example of the founder effect was first noted in the Dominican Republic in the 1970s. During a several-year period, eighteen children who had been born with female genitalia and raised as girls suddenly grew penises at puberty. This culture tended to value sons over daughters, so these transitions were generally celebrated. They labeled the condition guevedoces, which translates to “penis at twelve,” due to the average age at which this occurred. Scientists were fascinated by the phenomenon.
Genetic and hormonal studies revealed that the condition, scientifically termed 5-alpha reductase deficiency, is an autosomal recessive syndrome that manifests when a child having both X and Y sex chromosomes inherits two nonfunctional (mutated) copies of the SRD5A2 gene (Imperato-McGinley and Zhu 2002). These children develop testes internally, but the 5-alpha reductase 2 steroid, which is necessary for development of male genitals in babies, is not produced. In absence of this male hormone, the baby develops female-looking genitalia (in humans, “female” is the default infant body form, if the full set of the necessary male hormones are not produced). At puberty, however, a different set of male hormones are produced by other fully functional genes. These hormones complete the male genital development that did not happen in infancy. This condition became quite common in the Dominican Republic during the 1970s due to founder effect—that is, the mutated SRD5A2 gene happened to be much more common among the Dominican Republic’s founding population than in the parent populations. (The Dominican population derives from a mixture of Indigenous Americans [Taino] peoples, West Africans, and Western Europeans.) Five-alpha reductase syndrome has since been observed in other small, isolated populations around the world.
Founder effect is closely linked to the concept of inbreeding, which in population genetics does not necessarily mean breeding with immediate family relatives. Instead, inbreeding refers to the selection of mates exclusively from within a small, closed population—that is, from a group with limited allelic variability. This can be observed in small, physically isolated populations but also can happen when cultural practices limit mates to a small group. As with the founder effect, inbreeding increases the risk of inheriting two copies of any nonfunctional (mutant) alleles.
The Amish in the United States are a population that, due to their unique history and cultural practices, emerged from a small founding population and have tended to select mates from within their groups. The Old Order Amish population of Lancaster County, Pennsylvania, has approximately 50,000 current members, all of whom can trace their ancestry back to a group of approximately 80 individuals. This small founding population immigrated to the United States from Switzerland in the mid-1700s to escape religious persecution. Since the Amish keep to themselves and almost exclusively select mates from within their own communities, they have more recessive traits compared to their parent population.

One of the genetic conditions that has been observed much more frequently in the Lancaster County Amish population is Ellis-van Creveld syndrome, which is an autosomal recessive disorder characterized by short stature (dwarfism), polydactyly (the development of more than five digits [fingers or toes] on the hands or feet], abnormal tooth development, and heart defects (Figure 5.11). Among the general world population, Ellis-van Creveld syndrome is estimated to affect approximately 1 in 60,000 individuals; among the Old Order Amish of Lancaster County, the rate is estimated to be as high as 1 in every 200 births (D’Asdia et al. 2013).
One important insight that has come from the study of founder effects is that a limited gene pool carries a much higher risk for genetic diseases. Genetic diversity in a population greatly reduces these risks.
Gene Flow
The third force of evolution is traditionally called gene flow. As with genetic drift, this is a misnomer, because it refers to flowing alleles, not genes. (All members of the same species share the same genes; it is the alleles of those genes that may vary.) Gene flow refers to the movement of alleles from one population to another. In most cases, gene flow can be considered synonymous with migration.
Returning again to the example of our primordial cell population, let’s imagine that, after the volcanic fissure opened up in the ocean floor, wiping out the majority of the parent population, two surviving populations developed in the waters on opposite sides of the fissure. Ultimately, the lava from the fissure cooled into a large island that continued to provide a physical barrier between the populations (Figure 5.12).

In the initial generations after the eruption, due to founder effect, isolation, and random inheritance (genetic drift), the population to the west of the islands contained a vast majority of the ruffled membrane alleles while the eastern population carried only the smooth alleles. Ocean currents in the area typically flowed from east to west, sometimes carrying cells (facilitating gene flow) from the eastern (smooth) population to the western (ruffled) population. Due to the ocean currents, it was almost impossible for any cells from the western population to be carried eastward. Thus, for inheritance purposes, the eastern (smooth) population remained isolated. In this case, the gene flow is unidirectional (going only in one direction) and unbalanced (only one population is receiving the new alleles).
Among humans, gene flow is often described as admixture. In forensic cases, anthropologists and geneticists are often asked to estimate the ancestry of unidentified human remains to help determine whether they match any missing persons’ reports. This is one of the most complicated tasks in these professions because, while “race” or “ancestry” involves simple checkboxes on a missing person’s form, among humans today there are no truly distinct genetic populations. All modern humans are members of the same fully breeding compatible species, and all human communities have experienced multiple episodes of gene flow (admixture), leading all humans today to be so genetically similar that we are all members of the same (and only surviving) human subspecies: Homo sapiens sapiens.
Gene flow between otherwise isolated nonhuman populations is often termed hybridization.. One example of this involves the hybridization and spread of Scutellata honey bees (a.k.a. “killer bees”) in the Americas. All honey bees worldwide are classified as Apis mellifera. Due to distinct adaptations to various environments around the world, there are 28 different subspecies of Apis mellifera.
During the 1950s, a Brazilian biologist named Warwick E. Kerr experimented with hybridizing African and European subspecies of honey bees to try to develop a strain that was better suited to tropical environments than the European honey bees that had long been kept by North American beekeepers. Dr. Kerr was careful to contain the reproductive queens and drones from the African subspecies, but in 1957, a visiting beekeeper accidentally released 26 queen bees of the Scutellata subspecies (Apis mellifera scutellata) from southern Africa into the Brazilian countryside. The Scutellata bees quickly interbred with local European honey bee populations. The hybridized bees exhibited a much more aggressively defensive behavior, fatally or near-fatally attacking many humans and livestock that ventured too close to their hives. The hybridized bees spread throughout South America and reached Mexico and California by 1985. By 1990, permanent colonies had been established in Texas, and by 1997, 90% of trapped bee swarms around Tucson, Arizona, were found to be Scutellata hybrids (Sanford 2006).
Another example involves the introduction of the Harlequin ladybeetle, Harmonia axyridis, native to East Asia, to other parts of the world as a “natural” form of pest control. Harlequin ladybeetles are natural predators of some of the aphids and other crop-pest insects. First introduced to North America in 1916, the “biocontrol” strains of Harlequin ladybeetles were considered to be quite successful in reducing crop pests and saving farmers substantial amounts of money. After many decades of successful use in North America, biocontrol strains of Harlequin ladybeetles were also developed in Europe and South America in the 1980s.
Over the seven decades of biocontrol use, the Harlequin ladybeetle had never shown any potential for development of wild colonies outside of its native habitat in China and Japan. New generations of beetles always had to be reared in the lab. That all changed in 1988, when a wild colony took root near New Orleans, Louisiana. Either through admixture with a native ladybeetle strain, or due to a spontaneous mutation, a new allele was clearly introduced into this population that suddenly enabled them to survive and reproduce in a wide range of environments. This population spread rapidly across the Americas and had reached Africa by 2004.
In Europe, the invasive, North American strain of Harlequin ladybeetle admixed with the European strain (Figure 5.13), causing a population explosion (Lombaert et al. 2010). Even strains specifically developed to be flightless (to curtail the spreading) produced flighted offspring after admixture with members of the North American population (Facon et al. 2011). The fast-spreading, invasive strain has quickly become a disaster, out-competing native ladybeetle populations (some to the point of extinction), causing home infestations, decimating fruit crops, and contaminating many batches of wine with their bitter flavor after being inadvertently harvested with the grapes (Pickering et al. 2004).
Natural Selection
The final force of evolution is natural selection. This is the evolutionary process that Charles Darwin first brought to light, and it is what the general public typically evokes when considering the process of evolution. Natural selection occurs when certain phenotypes confer an advantage or disadvantage in survival and/or reproductive success. The alleles associated with those phenotypes will change in frequency over time due to this selective pressure. It’s also important to note that the advantageous allele may change over time (with environmental changes) and that an allele that had previously been benign may become advantageous or detrimental. Of course, dominant, recessive, and codominant traits will be selected upon a bit differently from one another. Because natural selection acts on phenotypes rather than the alleles themselves, deleterious (disadvantageous) alleles can be retained by heterozygotes without any negative effects.
In the case of our primordial ocean cells, up until now, the texture of their cell membranes has been benign. The frequencies of smooth to ruffled alleles, and smooth to ruffled phenotypes, has changed over time, due to genetic drift and gene flow. Let’s now imagine that the Earth’s climate has cooled to a point that the waters frequently become too cold for survival of the tiny bacteria that are the dietary staples of our smooth and ruffled cell populations. The way amoeba-like cells “eat” is to stretch out the cell membrane, almost like an arm, to encapsulate, then ingest, the tiny bacteria. When the temperatures plummet, the tiny bacteria populations plummet with them. Larger bacteria, however, are better able to withstand the temperature change.
The smooth cells were well-adapted to ingesting tiny bacteria but poorly suited to encapsulating the larger bacteria. The cells with the ruffled membranes, however, are easily able to extend their ruffles to encapsulate the larger bacteria. They also find themselves able to stretch their entire membrane to a much larger size than their smooth-surfaced neighbors, allowing them to ingest more bacteria at a given time and to go for longer periods between feedings (Figure 5.14).
The smooth and ruffled traits, which had previously offered no advantage or disadvantage while food was plentiful, now are subject to natural selection. During the cold snaps, at least, the ruffled cells have a definite advantage. We can imagine that the western population that has mostly ruffled alleles will continue to do well, while the eastern population is at risk of dying out if the smaller bacteria remain scarce and no ruffled alleles are introduced.
A classic example of natural selection involves the study of an insect called the peppered moth (Biston betularia) in England during the Industrial Revolution in the 1800s. Prior to the Industrial Revolution, the peppered moth population was predominantly light in color, with dark (pepper-like) speckles on the wings. The “peppered” coloration was very similar to the appearance of the bark and lichens that grew on the local trees (Figure 5.15). This helped to camouflage the moths as they rested on a tree, making it harder for moth-eating birds to find and snack on them. There was another phenotype that popped up occasionally in the population. These individuals were heterozygotes that carried an overactive, dominant pigment allele, producing a solid black coloration. As you can imagine, the black moths were much easier for birds to spot, making this phenotype a real disadvantage.
The situation changed, however, as the Industrial Revolution took off. Large factories began spewing vast amounts of coal smoke into the air, blanketing the countryside, including the lichens and trees, in black soot. Suddenly, it was the light-colored moths that were easy for birds to spot and the black moths that held the advantage. The frequency of the dark pigment allele rose dramatically. By 1895, the black moth phenotype accounted for 98% of observed moths (Grant 1999).

Thanks to new environmental regulations in the 1960s, the air pollution in England began to taper off. As the soot levels decreased, returning the trees to their former, lighter color, this provided the perfect opportunity to study how the peppered moth population would respond. Repeated follow-up studies documented the gradual rise in the frequency of the lighter-colored phenotype. By 2003, the maximum frequency of the dark phenotype was 50% and in most parts of England had decreased to less than 10% (Cook 2003).
Directional, Balancing/Stabilizing, and Disruptive/Diversifying Selection
Natural selection can be classified as directional, balancing/stabilizing, or disruptive/diversifying, depending on how the pressure is applied to the population (Figure 5.16).
Both of the above examples of natural selection involve directional selection: the environmental pressures favor one phenotype over the other and cause the frequencies of the associated advantageous alleles (ruffled membranes, dark pigment) to gradually increase. In the case of the peppered moths, the direction shifted three times: first, it was selecting for lighter pigment; then, with the increase in pollution, the pressure switched to selection for darker pigment; finally, with reduction of the pollution, the selection pressure shifted back again to favoring light-colored moths.
Balancing selection (a.k.a. stabilizing selection) occurs when selection works against the extremes of a trait and favors the intermediate phenotype. For example, humans maintain an average birth weight that balances the need for babies to be small enough not to cause complications during pregnancy and childbirth but big enough to maintain a safe body temperature after they are born. Another example of balancing selection is found in the genetic disorder called sickle cell anemia (see “Special Topic: Sickle Cell Anemia”).
Disruptive selection (a.k.a. diversifying selection), the opposite of balancing selection, occurs when both extremes of a trait are advantageous. Since individuals with traits in the mid-range are selected against, disruptive selection can eventually lead to the population evolving into two separate species. Darwin believed that the many species of finches (small birds) found in the remote Galapagos Islands provided a clear example of disruptive selection leading to speciation. He observed that seed-eating finches either had large beaks, capable of eating very large seeds, or small beaks, capable of retrieving tiny seeds. The islands did not have many plants that produced medium-size seeds. Thus, birds with medium-size beaks would have trouble eating the very large seeds and would also have been inefficient at picking up the tiny seeds. Over time, Darwin surmised, this pressure against mid-size beaks may have led the population to divide into two separate species.
Sexual Selection
Sexual selection is an aspect of natural selection in which the selective pressure specifically affects reproductive success (the ability to successfully breed and raise offspring) rather than survival. Sexual selection favors traits that will attract a mate. Sometimes these sexually appealing traits even carry greater risks in terms of survival.

A classic example of sexual selection involves the brightly colored feathers of the peacock. The peacock is the male sex of the peafowl genera Pavo and Afropavo. During mating season, peacocks will fan their colorful tails wide and strut in front of the peahens in a grand display. The peahens will carefully observe these displays and will elect to mate with the male that they find the most appealing. Many studies have found that peahens prefer the males with the fullest, most colorful tails. While these large, showy tails provide a reproductive advantage, they can be a real burden in terms of escaping predators. The bright colors and patterns as well as the large size of the peacock tail make it difficult to hide. Once predators spot them, peacocks also struggle to fly away, with the heavy tail trailing behind and weighing them down (Figure 5.17). Some researchers have argued that the increased risk is part of the appeal for the peahens: only an especially strong, alert, and healthy peacock would be able to avoid predators while sporting such a spectacular tail.
It’s important to keep in mind that sexual selection relies on the trait being present throughout mating years. Reflecting on the NF1 genetic disorder (see “Special Topic: Neurofibromatosis Type 1 [NF1]”), given how disfiguring the symptoms can become, some might find it surprising that half of the babies born with NF1 inherited it from a parent. Given that the disorder is autosomal dominant and fully penetrant (meaning it has no unaffected carriers), it may seem surprising that sexual selection doesn’t exert more pressure against the mutated alleles. One important factor is that, while the neurofibromas typically begin to appear during puberty, they usually emerge only a few at a time and may grow very slowly. Many NF1 patients don’t experience the more severe or disfiguring symptoms until later in life, long after they have started families of their own.
Some researchers prefer to classify sexual selection separately, as a fifth force of evolution. The traits that underpin mate selection are entirely natural, of course. Research has shown that subtle traits, such as the type of pheromones (hormonal odors related to immune system alleles) someone emits and how those are perceived by the immune system genotype of the “sniffer,” may play crucial and subconscious roles in whether we find someone attractive or not (Chaix, Cao, and Donnelly 2008).
Special Topic: Neurofibromatosis Type 1 (NF1)
Neurofibromatosis Type 1, also known as NF1, is a genetic disorder that illustrates how a mutation in a single gene can affect multiple systems in the body. Surprisingly common, more people have NF1 than cystic fibrosis and muscular dystrophy combined. Even more surprising, given how common it is, is how few people have heard of it. One in every 3,000 babies is born with NF1, and this holds true for all populations worldwide (Riccardi 1992). This means that, for every 3,000 people in your community, there is likely at least one person living with this disorder. NF1 is an autosomal dominant condition, which means that everyone born with a mutation in the gene, whether inherited or spontaneous, has a 50/50 chance of passing it on to each of their own children.
The NF1 disorder results from mutation of the NF1 gene on Chromosome 17. Almost any mutation that affects the sequence of the gene’s protein product, neurofibromin, will cause the disorder. Studies of individuals with NF1 have identified over 3,000 different mutations of all kinds (including point mutations, small and large indels, and translocations). The NF1 gene is one of the largest known genes, containing at least 60 exons (protein-encoding sequences) in a span of about 300,000 nucleotides.
We know that neurofibromin plays an important role in preventing tumor growth because one of the most common symptoms of the NF1 disorder is the growth of benign (noncancerous) tumors, called neurofibromas. Neurofibromas sprout from nerve sheaths—the tissues that encase our nerves—throughout the body, usually beginning around puberty. There is no way to predict where the tumors will occur, or when or how quickly they will grow, although only about 15% turn malignant (cancerous). The two types of neurofibromas that are typically most visible are cutaneous neurofibromas, which are spherical bumps on, or just under, the surface of the skin (Figure 5.18), and plexiform neurofibromas, growths involving whole branches of nerves, often giving the appearance that the surface of the skin is “melting” (Figure 5.19).


Unfortunately, there is currently no cure for NF1. Surgical removal of neurofibromas risks paralysis, due to the high potential for nerve damage, and often results in the tumors growing back even more vigorously. This means that patients are often forced to live with disfiguring and often painful neurofibromas. People who are not familiar with NF1 often mistake neurofibromas for something contagious. This makes it especially hard for people living with NF1 to get jobs working with the public or even to enjoy spending time away from home. Raising public awareness about NF1 and its symptoms can be a great help in improving the quality of life for people living with this condition.

One of the first symptoms of NF1 in a small child is usually the appearance of café-au-lait spots, or CALS, which are flat, brown birthmark-like spots on the skin (Figure 5.20). CALS are often light brown, similar to the color of coffee with cream, which is the reason for the name, although the shade of the pigment depends on a person’s overall complexion. Some babies are born with CALS, but for others the spots appear within the first few years of life. Having six or more CALS larger than five millimeters (mm) across is a strong indicator that a child may have NF1.
Other common symptoms include the following: gliomas (tumors) of the optic nerve, which can cause vision loss; thinning of bones and failure to heal if they break (often requiring amputation); low muscle tone (poor muscle development, often delaying milestones such as sitting up, crawling, and walking); hearing loss, due to neurofibromas on auditory nerves; and learning disabilities, especially those involving spatial reasoning. Approximately 50% of people with NF1 have some type of speech and/or learning disability and often benefit greatly from early intervention services. Generalized developmental disability, however, is not common with NF1, so most people with NF1 live independently as adults. Many people with NF1 live full and successful lives, as long as their symptoms can be managed.
Based on the wide variety of symptoms, it’s clear that the neurofibromin protein plays important roles in many biochemical pathways. While everyone who has NF1 will exhibit some symptoms during their lifetime, there is a great deal of variation in the types and severity of symptoms, even between individuals from the same family who share the exact same NF1 mutation. It seems crazy that a gene with so many important functions would be so susceptible to mutation. Part of this undoubtedly has to do with its massive size—a gene with 300,000 nucleotides has ten times more nucleotides available for mutation than does a gene of 30,000 bases. This also suggests that the mutability of this gene might provide some benefits, which is a possibility that we will revisit later in this chapter.
Special Topic: Sickle Cell Anemia
Sickle cell anemia is an autosomal recessive genetic disorder that affects millions of people worldwide. It is most common in Africa, countries around the Mediterranean Sea, and eastward as far as India. Populations in the Americas that have high percentages of ancestors from these regions also have high rates of sickle cell anemia. In the United States, it’s estimated that 72,000 people live with the disease, with one in approximately 1,200 Hispanic-American babies and one in every 500 African-American babies inheriting the condition (World Health Organization 1996).
Sickle cell anemia affects the hemoglobin protein in red blood cells. Normal red blood cells are somewhat doughnut-shaped—round with a depression on both sides of the middle. They carry oxygen around the bloodstream to cells throughout the body. Red blood cells produced by the mutated form of the gene take on a stiff, sickle-like crescent shape when stressed by low oxygen or dehydration (Figure 5.21). Because of their elongated shape and the fact that they are stiff rather than flexible, they tend to form clumps in the blood vessels, inhibiting blood flow to adjacent areas of the body. This causes episodes of extreme pain and can cause serious problems in the oxygen-deprived tissues. The sickle cells also break down much more quickly than normal cells, often lasting only 20 days rather than the 120 days of normal cells. This causes an overall shortage of blood cells in the sickle cell patient, resulting in low iron (anemia) and problems associated with it such as extreme fatigue, shortness of breath, and hindrances to children’s growth and development.
The devastating effects of sickle cell anemia made its high frequency a pressing mystery. Why would an allele that is so deleterious in its homozygous form be maintained in a population at levels as high as the one in twelve African Americans estimated to carry at least one copy of the allele? The answer turned out to be one of the most interesting cases of balancing selection in the history of genetic study.
While looking for an explanation, scientists noticed that the countries with high rates of sickle cell disease also shared a high risk for another disease called malaria, which is caused by infection of the blood by a Plasmodium parasite. These parasites are carried by mosquitoes and enter the human bloodstream via a mosquito bite. Once infected, the person will experience flu-like symptoms that, if untreated, can often lead to death. Researchers discovered that many people living in these regions seemed to have a natural resistance to malaria. Further study revealed that people who carry the sickle cell allele are far less likely to experience a severe case of malaria. This would not be enough of a benefit to make the allele advantageous for the sickle cell homozygotes, who face shortened life spans due to sickle cell anemia. The real benefit of the sickle cell allele goes to the heterozygotes.
People who are heterozygous for sickle cell carry one normal allele, which produces the normal, round, red blood cells, and one sickle cell allele, which produces the sickle-shaped red blood cells. Thus, they have both the sickle and round blood cell types in their bloodstream. They produce enough of the round red blood cells to avoid the symptoms of sickle cell anemia, but they have enough sickle cells to provide protection from malaria.
When the Plasmodium parasites infect an individual, they begin to multiply in the liver, but then must infect the red blood cells to complete their reproductive cycle. When the parasites enter sickle-type cells, the cells respond by taking on the sickle shape. This prevents the parasite from circulating through the bloodstream and completing its life cycle, greatly inhibiting the severity of the infection in the sickle cell heterozygotes compared to non–-sickle cell homozygotes. See Chapter 14 for more discussion of sickle cell anemia.
Special Topic: The Real Primordial Cells—Dictyostelium Discoideum
The amoeba-like primordial cells that were used as recurring examples throughout this chapter are inspired by actual research that is truly fascinating. In 2015, Gareth Bloomfield and colleagues reported on their genomic study of the social amoeba Dictyostelium discoideum (a.k.a. “slime molds,” although technically they are amoebae, not molds). Strains of these amoebae have been grown in research laboratories for many decades and are useful in studying the mechanisms that amoeboid single-celled organisms use to ingest food and liquid. For simplification of our examples in this chapter, our amoeba-like cells remained ocean dwellers. Wild Dictyostelium discoideum, however, live in soil and feed on soil bacteria by growing ruffles in their membranes that reach out to encapsulate the bacterial cell. Laboratory strains, however, are typically raised on liquid media (agar) in Petri dishes, which is not suitable for the wild-type amoebae. It was widely known that the laboratory strains must have developed mutations in one or more genes to allow them to ingest the larger nutrient particles in the agar and larger volumes of liquid, but the genes involved were not known.
Bloomfield and colleagues performed genomic testing on both the wild and the laboratory strains of Dictyostelium discoideum. Their discovery was astounding: every one of the laboratory strains carried a mutation in the NF1 gene, the very same gene associated with Neurofibromatosis Type 1 (NF1) in humans. The antiquity of this massive, easily mutated gene is incredible. It originated in an ancestor common to both humans and these amoebae, and it has been retained in both lineages ever since. As seen in Dictyostelium discoideum, breaking the gene can be advantageous. Without a functioning copy of the neurofibromin protein, the cell membrane is able to form much-larger feeding structures, allowing the NF1 mutants to ingest larger particles and larger volumes of liquid. For these amoebae, this may provide dietary flexibility that functions somewhat like an insurance policy for times when the food supply is limited.
Dictyostelium discoideum are also interesting in that they typically reproduce asexually, but under certain conditions, one cell will convert into a “giant” cell, which encapsulates surrounding cells, transforming into one of three sexes. This cell will undergo meiosis, producing gametes that must combine with one of the other two sexes to produce viable offspring. This ability for sexual reproduction may be what allows Dictyostelium discoideum to benefit from the advantages of NF1 mutation, while also being able to restore the wild type NF1 gene in future generations.
What does this mean for humans living with NF1? Well, understanding the role of the neurofibromin protein in the membranes of simple organisms like Dictyostelium discoideum may help us to better understand how it functions and malfunctions in the sheaths of human neurons. It’s also possible that the mutability of the NF1 gene confers certain advantages to humans as well. Alleles of the NF1 gene have been found to reduce one’s risk for alcoholism (Repunte-Canonigo Vez et al. 2015), opiate addiction (Sanna et al. 2002), Type 2 diabetes (Martins et al. 2016), and hypomusicality (a lower-than-average musical aptitude; Cota et al. 2018). This research is ongoing and will be exciting to follow in the coming years.
Studying Evolution in Action
The Hardy-Weinberg Equilibrium
This chapter has introduced you to the forces of evolution, the mechanisms by which evolution occurs. How do we detect and study evolution, though, in real time, as it happens? One tool we use is the Hardy-Weinberg Equilibrium: a mathematical formula that allows estimation of the number and distribution of dominant and recessive alleles in a population. This aids in determining whether allele frequencies are changing and, if so, how quickly over time, and in favor of which allele? It’s important to note that the Hardy-Weinberg formula only gives us an estimate based on the data for a snapshot in time. We will have to calculate it again later, after various intervals, to determine if our population is evolving and in what way the allele frequencies are changing.
Calculating the Hardy-Weinberg Equilibrium
In the Hardy-Weinberg formula, p represents the frequency of the dominant allele, and q represents the frequency of the recessive allele. Remember, an allele’s frequency is the proportion, or percentage, of that allele in the population. For the purposes of Hardy-Weinberg, we give the allele percentages as decimal numbers (e.g., 42% = 0.42), with the entire population (100% of alleles) equaling 1. If we can figure out the frequency of one of the alleles in the population, then it is simple to calculate the other. Simply subtract the known frequency from 1 (the entire population): 1 – p = q and 1 – q = p.
The Hardy-Weinberg formula is p2 + 2pq + q2, where:
p2 represents the frequency of the homozygous dominant genotype;
2pq represents the frequency of the heterozygous genotype; and
q2 represents the frequency of the homozygous recessive genotype.
It is often easiest to determine q2 first, simply by counting the number of individuals with the unique, homozygous recessive phenotype (then dividing by the total individuals in the population to arrive at the “frequency”). Once we have this number, we simply need to calculate the square root of the homozygous recessive phenotype frequency. That gives us q. Remember, 1 – q equals p, so now we have the frequencies for both alleles in the population. If we needed to figure out the frequencies of heterozygotes and homozygous dominant genotypes, we’d just need to plug the p and q frequencies back into the p2 and 2pq formulas.

Let’s imagine we have a population of ladybeetles that carries two alleles: a dominant allele that produces red ladybeetles and a recessive allele that produces orange ladybeetles. Since red is dominant, we’ll use R to represent the red allele, and r to represent the orange allele. Our population has ten beetles, and seven are red and three are orange (Figure 5.24). Let’s calculate the number of genotypes and alleles in this population.
Of ten total beetles, we have three orange beetles3/10 = .30 (30%) frequency—and we know they are homozygous recessive (rr). So:
rr = .3; therefore, r = √.3 = .5477
R = 1 – .5477 = .4523
Using the Hardy-Weinberg formula:
1=.45232 + 2 x .4523 x .5477 +.54772 = .20 + .50 + .30 = 1
Thus, the genotype breakdown is 20% RR, 50% Rr, and 30% rr
(2 red homozygotes, 5 red heterozygotes, and 3 orange homozygotes).
Since we have 10 individuals, we know we have 20 total alleles: 4 red from the RR group, 5 red and 5 orange from the Rr group, and 6 orange from the rr group, for a grand total of 9 red and 11 orange (45% red and 55% orange, just like we estimated in the 1 – q step).
Reminder: The Hardy-Weinberg formula only gives us an estimate for a snapshot in time. We will have to calculate it again later, after various intervals, to determine if our population is evolving and in what way the allele frequencies are changing.
Interpreting Evolutionary Change: Nonrandom Mating
Once we have detected change occurring in a population, we need to consider which evolutionary processes might be the cause of the change. It is important to watch for nonrandom mating patterns, to see if they can be included or excluded as possible sources of variation in allele frequencies.
Nonrandom mating (also known as assortative mating) occurs when mate choice within a population follows a nonrandom pattern.
Positive assortative mating patterns result from a tendency for individuals to mate with others who share similar phenotypes. This often happens based on body size. Taking as an example dog breeds, it is easier for two Chihuahuas to mate and have healthy offspring than it is for a Chihuahua and a St. Bernard to do so. This is especially true if the Chihuahua is the female and would have to give birth to giant St. Bernard pups.
Negative assortative mating patterns occur when individuals tend to select mates with qualities different from their own. This is what is at work when humans choose partners whose pheromones indicate that they have different and complementary immune alleles, providing potential offspring with a better chance at a stronger immune system.
Among domestic animals, such as pets and livestock, assortative mating is often directed by humans who decide which pairs will mate to increase the chances of offspring having certain desirable traits. This is known as artificial selection.
Among humans, in addition to phenotypic traits, cultural traits such as religion and ethnicity may also influence assortative mating patterns.
Defining a Species
Species are organisms whose individuals are capable of breeding because they are biologically and behaviorally compatible to produce viable, fertile offspring. Viable offspring are those offspring that are healthy enough to survive to adulthood. Fertile offspring are able to reproduce successfully, resulting in offspring of their own. Both conditions must be met for individuals to be considered part of the same species. As you can imagine, these criteria complicate the identification of distinct species in fossilized remains of extinct populations. In those cases, we must examine how much phenotypic variation is typically found within a comparable modern-day species; we can then determine whether the fossilized remains fall within the expected range of variation for a single species.
Some species have subpopulations that are regionally distinct. These are classified as separate subspecies because they have their own unique phenotypes and are geographically isolated from one another. However, if they do happen to encounter one another, they are still capable of successful interbreeding.
There are many examples of sterile hybrids that are offspring of parents from two different species. For example, horses and donkeys can breed and have offspring together. Depending on which species is the mother and which is the father, the offspring are either called mules, or hennies. Mules and hennies can live full life spans but are not able to have offspring of their own. Likewise, tigers and lions have been known to mate and have viable offspring. Again, depending on which species is the mother and which is the father, these offspring are called either ligers or tigons. Like mules and hennies, ligers and tigons are unable to reproduce. In each of these cases, the mismatched set of chromosomes that the offspring inherit produce an adequate set of functioning genes for the hybrid offspring; however, once mixed and divided in meiosis, the gametes don’t contain the full complement of genes needed for survival in the third generation.
Micro- to Macroevolution
Microevolution refers to changes in allele frequencies within breeding populations—that is, within single species. Macroevolution describes how the similarities and differences between species, as well as the phylogenetic relationships with other taxa, lead to changes that result in the emergence of new species. Consider our example of the peppered moth that illustrated microevolution over time, via directional selection favoring the peppered allele when the trees were clean and the dark pigment allele when the trees were sooty. Imagine that environmental regulations had cleaned up the air pollution in one part of the nation, while the coal-fired factories continued to spew soot in another area. If this went on long enough, it’s possible that two distinct moth populations would eventually emerge—one containing only the peppered allele and the other only harboring the dark pigment allele.
When a single population divides into two or more separate species, it is called speciation. The changes that prevent successful breeding between individuals who descended from the same ancestral population may involve chromosomal rearrangements, changes in the ability of the sperm from one species to permeate the egg membrane of the other species, or dramatic changes in hormonal schedules or mating behaviors that prevent members from the new species from being able to effectively pair up.
There are two types of speciation: allopatric and sympatric. Allopatric speciation is caused by long-term isolation (physical separation) of subgroups of the population (Figure 5.22). Something occurs in the environment—perhaps a river changes its course and splits the group, preventing them from breeding with members on the opposite riverbank. Over many generations, new mutations and adaptations to the different environments on each side of the river may drive the two subpopulations to change so much that they can no longer produce fertile, viable offspring, even if the barrier is someday removed.
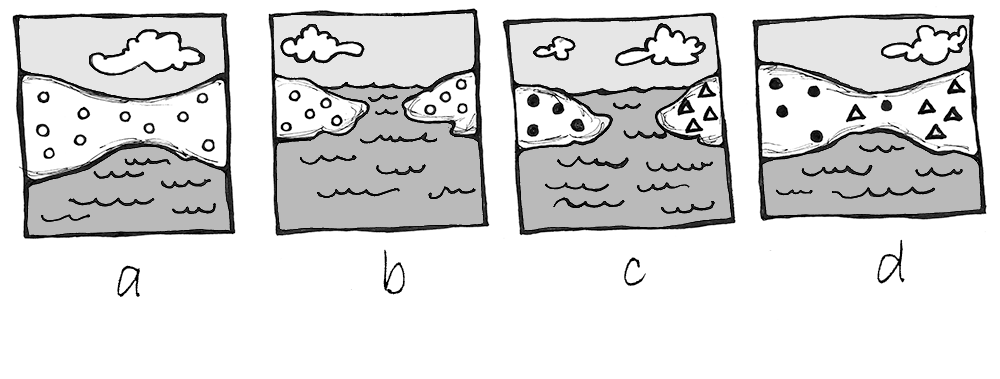
Sympatric speciation occurs when the population splits into two or more separate species while remaining located together without a physical barrier. This typically results from a new mutation that pops up among some members of the population that prevents them from successfully reproducing with anyone who does not carry the same mutation. This is seen particularly often in plants, as they have a higher frequency of chromosomal duplications.
One of the quickest rates of speciation is observed in the case of adaptive radiation. Adaptive radiation refers to the situation in which subgroups of a single species rapidly diversify and adapt to fill a variety of ecological niches. An ecological niche is a set of constraints and resources that is available in an environmental setting. Evidence for adaptive radiations is often seen after population bottlenecks. A mass disaster kills off many species, and the survivors have access to a new set of territories and resources that were either unavailable or much coveted and fought over before the disaster. The offspring of the surviving population will often split into multiple species, each of which stems from members in that first group of survivors who happened to carry alleles that were advantageous for a particular niche.
The classic example of adaptive radiation brings us back to Charles Darwin and his observations of the many species of finches on the Galapagos Islands. We are still not sure how the ancestral population of finches first arrived on that remote Pacific Island chain, but they found themselves in an environment filled with various insects, large and tiny seeds, fruit, and delicious varieties of cactus. Some members of that initial population carried alleles that gave them advantages for each of these dietary niches. In subsequent generations, others developed new mutations, some of which were beneficial. These traits were selected for, making the advantageous alleles more common among their offspring. As the finches spread from one island to the next, they would be far more likely to find mates among the birds on their new island. Birds feeding in the same area were then more likely to mate together than birds who have different diets, contributing to additional assortative mating. Together, these evolutionary mechanisms caused rapid speciation that allowed the new species to make the most of the various dietary niches (Figure 5.23).
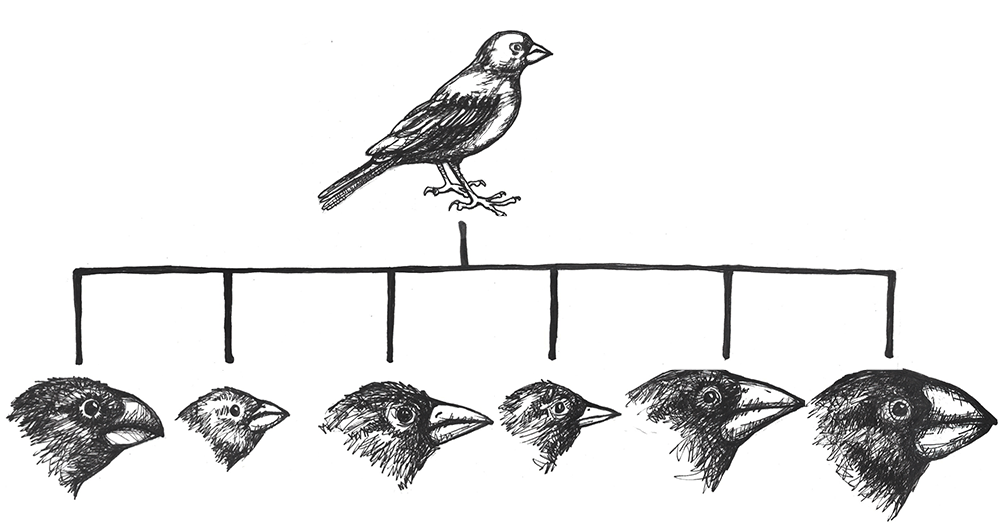
In today’s modern world, understanding these evolutionary processes is crucial for developing immunizations and antibiotics that can keep up with the rapid mutation rate of viruses and bacteria. This is also relevant to our food supply, which relies, in large part, on the development of herbicides and pesticides that keep up with the mutation rates of pests and weeds. Viruses, bacteria, agricultural pests, and weeds have all shown great flexibility in developing alleles that make them resistant to the latest medical treatment, pesticide, or herbicide. Billion-dollar industries have specialized in trying to keep our species one step ahead of the next mutation in the pests and infectious diseases that put our survival at risk.
Review Questions
- Summarize the Modern Synthesis and provide several examples of how it is relevant to questions and problems in our world today.
- You inherit a house from a long-lost relative that contains a fancy aquarium, filled with a variety of snails. The phenotypes include large snails and small snails; red, black, and yellow snails; and solid, striped, and spotted snails. Devise a series of experiments that would help you determine how many snail species are present in your aquarium.
- Match the correct force of evolution with the correct real-world example:
a. Mutationi. 5-alpha reductase deficiency
b. Genetic Driftii. Peppered Moths
c. Gene Flowiii. Neurofibromatosis Type 1
d. Natural Selectioniv. Scutellata Honey Bees - Imagine a population of common house mice (Mus musculus). Draw a comic strip illustrating how mutation, genetic drift, gene flow, and natural selection might transform this population over several (or more) generations.
-
The many breeds of the single species of domestic dog (Canis familiaris) provide an extreme example of microevolution. Discuss why this is the case. What future scenarios can you imagine that could potentially transform the domestic dog into an example of macroevolution?
-
The ability to roll one’s tongue (lift the outer edges of the tongue to touch each other, forming a tube) is a dominant trait. In a small town of 1,500 people, 500 can roll their tongues. Use the Hardy-Weinberg formula to determine how many individuals in the town are homozygous dominant, heterozygous, and homozygous recessive.
Key Terms
5-alpha reductase deficiency: An autosomal recessive syndrome that manifests when a child having both X and Y sex chromosomes inherits two nonfunctional (mutated) copies of the SRD5A2 gene, producing a deficiency in a hormone necessary for development in infancy of typical male genitalia. These children often appear at birth to have female genitalia, but they develop a penis and other sexual characteristics when other hormones kick in during puberty.
Adaptive radiation: The situation in which subgroups of a single species rapidly diversify and adapt to fill a variety of ecological niches.
Admixture: A term often used to describe gene flow between human populations. Sometimes also used to describe gene flow between nonhuman populations.
Allele frequency: The ratio, or percentage, of one allele compared to the other alleles for that gene within the study population.
Alleles: Variant forms of genes.
Allopatric speciation: Speciation caused by long-term isolation (physical separation) of subgroups of the population.
Antibiotics: Medicines prescribed to treat bacterial infections.
Artificial selection: Human-directed assortative mating among domestic animals, such as pets and livestock, designed to increase the chances of offspring having certain desirable traits.
Asexual reproduction: Reproduction via mitosis, whereby offspring are clones of the parents.
Autosomal dominant: A phenotype produced by a gene on an autosomal chromosome that is expressed, to the exclusion of the recessive phenotype, in heterozygotes.
Autosomal recessive: A phenotype produced by a gene on an autosomal chromosome that is expressed only in individuals homozygous for the recessive allele.
Balanced translocations: Chromosomal translocations in which the genes are swapped but no genetic information is lost.
Balancing selection: A pattern of natural selection that occurs when the extremes of a trait are selected against, favoring the intermediate phenotype (a.k.a. stabilizing selection).
Beneficial mutations: Mutations that produce some sort of an advantage to the individual.
Benign: Noncancerous. Benign tumors may cause problems due to the area in which they are located (e.g., they might put pressure on a nerve or brain area), but they will not release cells that aggressively spread to other areas of the body.
Café-au-lait spots (CALS): Flat, brown birthmark-like spots on the skin, commonly associated with Neurofibromatosis Type 1.
Chromosomal translocations: The transfer of DNA between nonhomologous chromosomes.
Chromosomes: Molecules that carry collections of genes.
Codons: Three-nucleotide units of DNA that function as three-letter “words,” encoding instructions for the addition of one amino acid to a protein or indicating that the protein is complete.
Cretaceous–Paleogene extinction: A mass disaster caused by an asteroid that struck the earth approximately 66 million years ago and killed 75% of life on Earth, including all terrestrial dinosaurs. (a.k.a. K-Pg Extinction, Cretatious-Tertiary Extinction, and K-T Extinction).
Crossover events: Chromosomal alterations that occur when DNA is swapped between homologous chromosomes while they are paired up during meiosis I.
Cutaneous neurofibromas: Neurofibromas that manifest as spherical bumps on or just under the surface of the skin.
Deleterious mutation: A mutation producing negative effects to the individual such as the beginnings of cancers or heritable disorders.
Deletions: Mutations that involve the removal of one or more nucleotides from a DNA sequence.
Derivative chromosomes: New chromosomal structures resulting from translocations.
Dictyostelium discoideum: A species of social amoebae that has been widely used for laboratory research. Laboratory strains of Dictyostelium discoideum all carry mutations in the NF1 gene, which is what allows them to survive on liquid media (agar) in Petri dishes.
Directional selection: A pattern of natural selection in which one phenotype is favored over the other, causing the frequencies of the associated advantageous alleles to gradually increase.
Disruptive selection: A pattern of natural selection that occurs when both extremes of a trait are advantageous and intermediate phenotypes are selected against (a.k.a. diversifying selection).
DNA repair mechanisms: Enzymes that patrol and repair DNA in living cells.
DNA transposons: Transposons that are clipped out of the DNA sequence itself and inserted elsewhere in the genome.
Ecological niche: A set of constraints and resources that are available in an environmental setting.
Ellis-van Creveld syndrome: An autosomal recessive disorder characterized by short stature (dwarfism), polydactyly (the development of more than five digits [fingers or toes] on the hands or feet), abnormal tooth development, and heart defects. Estimated to affect approximately one in 60,000 individuals worldwide, among the Old Order Amish of Lancaster County, the rate is estimated to be as high as one in every 200 births.
Evolution: A change in the allele frequencies in a population over time.
Exons: The DNA sequences within a gene that directly encode protein sequences. After being transcribed into messenger RNA, the introns (DNA sequences within a gene that do not directly encode protein sequences) are clipped out, and the exons are pasted together prior to translation.
Fertile offspring: Offspring that can successfully reproduce, resulting in offspring of their own.
Founder effect: A type of genetic drift that occurs when members of a population leave the main or “parent” group and form a new population that no longer interbreeds with the other members of the original group.
Frameshift mutations: Types of indels that involve the insertion or deletion of any number of nucleotides that is not a multiple of three. These “shift the reading frame” and cause all codons beyond the mutation to be misread.
Gametes: The reproductive cells, produced through meiosis (a.k.a. germ cells or sperm or egg cells).
Gene: A sequence of DNA that provides coding information for the construction of proteins.
Gene flow: The movement of alleles from one population to another. This is one of the forces of evolution.
Gene pool: The entire collection of genetic material in a breeding community that can be passed on from one generation to the next.
Genetic drift: Random changes in allele frequencies within a population from one generation to the next. This is one of the forces of evolution.
Genotype: The set of alleles that an individual has for a given gene.
Genotype frequencies: The ratios or percentages of the different homozygous and heterozygous genotypes in the population.
Guevedoces: The term coined locally in the Dominican Republic for the condition scientifically known as 5-alpha reductase deficiency. The literal translation is “penis at twelve.”
Hardy-Weinberg Equilibrium: A mathematical formula (1=p2 + 2pq + q2 ) that allows estimation of the number and distribution of dominant and recessive alleles in a population.
Harlequin ladybeetle: A species of ladybeetle, native to East Asia, that was introduced to Europe and the Americas as a form of pest control. After many decades of use, one of the North American strains developed the ability to reproduce in diverse environments, causing it to spread rapidly throughout the Americas, Europe, and Africa. It has hybridized with European strains and is now a major pest in its own right.
Heterozygous genotype: A genotype comprising two different alleles.
Homozygous genotype: A genotype comprising an identical set of alleles.
Hybridization: A term often used to describe gene flow between nonhuman populations.
Inbreeding: The selection of mates exclusively from within a small, closed population.
Indels: A class of mutations that includes both insertions and deletions.
Inherited mutation: A mutation that has been passed from parent to offspring.
Insertions: Mutations that involve the addition of one or more nucleotides into a DNA sequence.
Isolation: Prevention of a population subgroup from breeding with other members of the same species due to a physical barrier or, in humans, a cultural rule.
Last Universal Common Ancestor (LUCA): The ancient organism from which all living things on Earth are descended.
Macroevolution: Changes that result in the emergence of new species, how the similarities and differences between species, as well as the phylogenetic relationships with other taxa, lead to changes that result in the emergence of new species.
Malaria: A frequently deadly mosquito-borne disease caused by infection of the blood by a Plasmodium parasite.
Malignant: Cancerous. Malignant tumors grow aggressively and their cells may metastasize (travel through the blood or lymph systems) to form new, aggressive tumors in other areas of the body.
Microevolution: Changes in allele frequencies within breeding populations—that is, within a single species.
Modern Synthesis: The integration of Darwin’s, Mendel’s, and subsequent research into a unified theory of evolution.
Monosomies: Conditions resulting from a nondisjunction event, in which a cell ends up with only one copy of a chromosome. In humans, a single X chromosome is the only survivable monosomy.
Mutation: A change in the nucleotide sequence of the genetic code. This is one of the forces of evolution.
Natural selection: An evolutionary process that occurs when certain phenotypes confer an advantage or disadvantage in survival and/or reproductive success. This is one of the forces of evolution, and it was first identified by Charles Darwin.
Negative assortative mating: A pattern that occurs when individuals tend to select mates with qualities different from their own.
Neurofibromas: Nerve sheath tumors that are common symptoms of Neurofibromatosis Type 1.
Neurofibromatosis Type 1: An autosomal dominant genetic disorder affecting one in every 3,000 people. It is caused by mutation of the NF1 gene on Chromosome 17, resulting in a defective neurofibromin protein. The disorder is characterized by neurofibromas, café-au-lait spots, and a host of other potential symptoms.
NF1: An abbreviation for Neurofibromatosis Type 1. When italicized, NF1 refers to the gene on Chromosome 17 that encodes the neurofibromin protein.
Nondisjunction events: Chromosomal abnormalities that occur when the homologous chromosomes (in meiosis I) or sister chromatids (in meiosis II and mitosis) fail to separate after pairing. The result is that both chromosomes or chromatids end up in the same daughter cell, leaving the other daughter cell without any copy of that chromosome.
Nonrandom mating: A scenario in which mate choice within a population follows a nonrandom pattern (a.k.a. assortative mating).
Nonsynonymous mutation: A point mutation that causes a change in the resulting protein.
Old Order Amish: A culturally isolated population in Lancaster County, Pennsylvania, that has approximately 50,000 current members, all of whom can trace their ancestry back to a group of approximately eighty individuals. This group has high rates of certain genetics disorders, including Ellis-van Creveld syndrome.
Origins of life: How the first living organism came into being.
Peacock: The male sex of the peafowl, famous for its large, colorful tail, which it dramatically displays to attract mates. (The female of the species is known as a peahen.)
Peppered moth: A species of moth (Biston betularia) found in England that has light and dark phenotypes. During the Industrial Revolution, when soot blackened the trees, the frequency of the previously rare dark phenotype dramatically increased, as lighter-colored moths were easier for birds to spot against the sooty trees. After environmental regulations eliminated the soot, the lighter-colored phenotype gradually became most common again.
Phenotype: The observable traits that are produced by a genotype.
Phylogenetic tree of life: A family tree of all living organisms, based on genetic relationships.
Phylogenies: Genetically determined family lineages.
Plasmodium: A genus of mosquito-borne parasite. Several Plasmodium species cause malaria when introduced to the human bloodstream via a mosquito bite.
Plexiform neurofibromas: Neurofibromas that involve whole branches of nerves, often giving the appearance that the surface of the skin is “melting.”
Point mutation: A single-letter (single-nucleotide) change in the genetic code, resulting in the substitution of one nucleic acid base for a different one.
Polymorphisms: Multiple forms of a trait; alternative phenotypes within a given species.
Population: A group of individuals who are genetically similar enough and geographically near enough to one another that they can breed and produce new generations of individuals.
Population bottleneck: A type of genetic drift that occurs when the number of individuals in a population drops dramatically due to some random event.
Positive assortative mating: A pattern that results from a tendency for individuals to mate with others who share similar phenotypes.
Retrotransposons: Transposons that are transcribed from DNA into RNA, and then are “reverse transcribed,” to insert the copied sequence into a new location in the DNA.
Scutellata honey bees: A strain of honey bees that resulted from the hybridization of African and European honey bee subspecies. These bees were accidentally released into the wild in 1957 in Brazil and have since spread throughout South and Central America and into the United States. Also known as “killer bees,” they tend to be very aggressive in defense of their hives and have caused many fatal injuries to humans and livestock.
Sexual reproduction: Reproduction via meiosis and combination of gametes. Offspring inherit genetic material from both parents.
Sexual selection: An aspect of natural selection in which the selective pressure specifically affects reproductive success (the ability to successfully breed and raise offspring).
Sickle cell anemia: An autosomal recessive genetic disorder that affects millions of people worldwide. It is most common in Africa, countries around the Mediterranean Sea, and eastward as far as India. Homozygotes for the recessive allele develop the disorder, which produce misshapen red blood cells that cause iron deficiency, painful episodes of oxygen-deprivation in localized tissues, and a host of other symptoms. In heterozygotes, though, the sickle cell allele confers a greater resistance to malaria.
Somatic cells: The cells of our organs and other body tissues (all cells except gametes) that replicate by mitosis.
Speciation: The process by which a single population divides into two or more separate species.
Species: Organisms whose individuals are capable of breeding because they are biologically and behaviorally compatible to produce viable, fertile offspring.
Spontaneous mutation: A mutation that occurs due to random chance or unintentional exposure to mutagens. In families, a spontaneous mutation is the first case, as opposed to mutations that are inherited from parents.
Subspecies: A distinct subtype of a species. Most often, this is a geographically isolated population with unique phenotypes; however, it remains biologically and behaviorally capable of interbreeding with other populations of the same species.
Sympatric speciation: When a population splits into two or more separate species while remaining located together without a physical (or cultural) barrier.
Synonymous mutation: A point mutation that does not change the resulting protein.
Transposable elements: Fragments of DNA that can “jump” around in the genome.
Transposon: Another term for “transposable element.”
Trisomies: Conditions in which three copies of the same chromosome end up in a cell, resulting from a nondisjunction event. Down syndrome, Edwards syndrome, and Patau syndrome are trisomies.
Unbalanced translocations: Chromosomal translocations in which there is an unequal exchange of genetic material, resulting in duplication or loss of genes.
UV crosslinking: A type of mutation in which adjacent thymine bases bind to one another in the presence of UV light.
Viable offspring: Offspring that are healthy enough to survive to adulthood.
Xeroderma pigmentosum: An autosomal recessive disease in which DNA repair mechanisms do not function correctly, resulting in a host of problems especially related to sun exposure, including severe sunburns, dry skin, heavy freckling, and other pigment changes.
For Further Exploration
Explore Evolution on HHMI’s Biointeractive website.
Teaching Evolution through Human Examples, Smithsonian Museum of Natural History websites.
References
Bloomfield, Gareth, David Traynor, Sophia P. Sander, Douwe M. Veltman, Justin A. Pachebat, and Robert R. Kay. 2015. “Neurofibromin Controls Macropinocytosis and Phagocytosis in Dictyostelium.” eLife 4:e04940.
Chaix, Raphaëlle, Chen Cao, and Peter Donnelly. 2008. “Is Mate Choice in Humans MHC-Dependent?” PLoS Genetics 4 (9): e1000184.
Cook, Laurence M. 2003. "The Rise and Fall of the Carbonaria Form of the Peppered Moth." The Quarterly Review of Biology 78 (4): 399–417.
Cota, Bruno Cézar Lage, João Gabriel Marques Fonseca, Luiz Oswaldo Carneiro Rodrigues, Nilton Alves de Rezende, Pollyanna Barros Batista, Vincent Michael Riccardi, and Luciana Macedo de Resende. 2018. “Amusia and Its Electrophysiological Correlates in Neurofibromatosis Type 1.” Arquivos de Neuro-Psiquiatria 76 (5): 287–295.
D’Asdia, Maria Cecilia, Isabella Torrente, Federica Consoli, Rosangela Ferese, Monia Magliozzi, Laura Bernardini, Valentina Guida, et al. 2013. “Novel and Recurrent EVC and EVC2 Mutations in Ellis-van Creveld Syndrome and Weyers Acrofacial Dyostosis.” European Journal of Medical Genetics 56 (2): 80–87.
Dobzhansky, Theodosius. 1937. Genetics and the Origin of Species. Columbia University Biological Series. New York: Columbia University Press.
Facon, Benoît, Laurent Crespin, Anne Loiseau, Eric Lombaert, Alexandra Magro, and Arnaud Estoup. 2011. “Can Things Get Worse When an Invasive Species Hybridizes? The Harlequin Ladybird Harmonia axyridis in France as a Case Study.” Evolutionary Applications 4 (1): 71–88.
Fisher, Ronald A. 1919. "The Correlation between Relatives on the Supposition of Mendelian Inheritance." Transactions of the Royal Society of Edinburgh 52 (2): 399–433.
Ford, E. B. 1942. Genetics for Medical Students. London: Methuen.
Ford, E. B. 1949. Mendelism and Evolution. London: Methuen.
Grant, Bruce S. 1999. “Fine-tuning the Peppered Moth Paradigm.” Evolution 53 (3): 980–984.
Haldane, J. B. S. 1924. “A Mathematical Theory of Natural and Artificial Selection (Part 1).” Transactions of the Cambridge Philosophical Society 23 (2):19–41.
Hoelzel, A. R., Gkafas, G. A., Kang, H., Sarigol, F., Le Boeuf, B., Costa, D. P., Beltran, R. S., Reiter, J., Robinson, P. W., McInerney, N., Seim, I., Sun, S., Fan, G., & Li, S. (2024). Genomics of post-bottleneck recovery in the northern elephant seal. Nature Ecology & Evolution, 8, 686–694. https://doi.org/10.1038/s41559-024-02337-4
Imperato-McGinley, J., and Y.-S. Zhu. 2002. “Androgens and Male Physiology: The Syndrome of 5 Alpha-Reductase-2 Deficiency.” Molecular and Cellular Endocrinology 198 (1-2): 51–59.
Jablonski, David, and W. G. Chaloner. 1994. "Extinctions in the Fossil Record.” Philosophical Transactions of the Royal Society of London B: Biological Sciences 344 (1307): 11–17.
Livi-Bacci, Massimo. 2006. “The Depopulation of Hispanic America after the Conquest.” Population Development and Review 32 (2): 199–232.
Lombaert, Eric, Thomas Guillemaud, Jean-Marie Cornuet, Thibaut Malausa, Benoît Facon, and Arnaud Estoup. 2010. "Bridgehead Effect in the Worldwide Invasion of the Biocontrol Harlequin Ladybird.” PLoS ONE 5 (3): e9743.
Martins, Aline Stangherlin, Ann Kristine Jansen, Luiz Oswaldo Carneiro Rodrigues, Camila Maria Matos, Marcio Leandro Ribeiro Souza, Juliana Ferreira de Souza, Maria de Fátima Haueisen Sander Diniz, et al. 2016. “Lower Fasting Blood Glucose in Neurofibromatosis Type 1.” Endocrine Connections 5 (1): 28–33.
Pickering, Gary, James Lin, Roland Riesen, Andrew Reynolds, Ian Brindle, and George Soleas. 2004. "Influence of Harmonia axyridis on the Sensory Properties of White and Red Wine." American Journal of Enology and Viticulture 55 (2): 153–159.
Repunte-Canonigo Vez, Melissa A. Herman, Tomoya Kawamura, Henry R. Kranzler, Richard Sherva, Joel Gelernter, Lindsay A. Farrer, Marisa Roberto, and Pietro Paolo Sanna. 2015. “NF1 Regulates Alcohol Dependence-Associated Excessive Drinking and Gamma-Aminobutyric Acid Release in the Central Amygdala in Mice and Is Associated with Alcohol Dependence in Humans.” Biological Psychiatry 77 (10): 870–879.
Riccardi, Vincent M. 1992. Neurofibromatosis: Phenotype, Natural History, and Pathogenesis. Baltimore: Johns Hopkins University Press.
Sanford, Malcolm T. 2006. "The Africanized Honey Bee in the Americas: A Biological Revolution with Human Cultural Implications, Part V—Conclusion." American Bee Journal 146 (7): 597–599.
Sanna, Pietro Paolo, Cindy Simpson, Robert Lutjens, and George Koob. 2002. “ERK Regulation in Chronic Ethanol Exposure and Withdrawal.” Brain Research 948 (1–2): 186–191.
Weber, DianaS., Stewart, B. S., Garza, J. Carlos., & Lehman, N. (2000). An empirical genetic assessment of the severity of the northern elephant seal population bottleneck. Current Biology, 10(20), 1287–1290. https://doi.org/10.1016/s0960-9822(00)00759-4
World Health Organization. 1996. “Control of Hereditary Disorders: Report of WHO Scientific meeting (1996).” WHO Technical Reports 865. Geneva: World Health Organization.
World Health Organization. 2017. “Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics.” Global Priority Pathogens List, February 27. Geneva: World Health Organization. https://www.who.int/medicines/publications/WHO-PPL-Short_Summary_25Feb-ET_NM_WHO.pdf.
Wright, Sewall. 1932. "The Roles of Mutation, Inbreeding, Crossbreeding, and Selection in Evolution." Proceedings of the Sixth International Congress on Genetics 1 (6): 356–366.
Acknowledgment
Many thanks to Dr. Vincent M. Riccardi for sharing his vast knowledge of neurofibromatosis and for encouraging me to explore it from an anthropological perspective.
Karin Enstam Jaffe, Ph.D., Sonoma State University
This chapter is a revision from "Chapter 6: Primate Ecology and Behavior” by Karin Enstam Jaffe. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Describe the variables that affect primate diets.
- Explain how primates interact with other organisms in their environment.
- Discuss why primates live in groups, types of primate groups, and components of their social systems.
- Describe the reproductive strategies of males and females.
- Explain the ways in which primates communicate.
- Discuss the evidence for primate cultural traditions.
Nonhuman primates (hereafter, “primates”) are a fascinating group of animals, whose similarity to humans can be striking. Because of this similarity, studying primates helps anthropologists to gain insight into how our human ancestors may have behaved. It also allows us to better understand our own behavior through comparison (examining similarities and differences) with other primates as well as by comparing different species of primates to one another. In this way, studying primates helps anthropologists comprehend humanity from a biological perspective, which contributes to anthropology’s commitment to holism, the idea that the parts of a system interconnect and interact to make up the whole.

Ethology is the study of animal behavior, while primatology is the study of primate behavior. People who study primates are called primatologists. Research on primates can be conducted in the field (i.e., on wild primates) or in captivity (i.e., zoos) and may or may not involve experiments, such as playing recorded alarm calls to see how individuals react. Unlike some other Science, Technology, Engineering, and Math (STEM) fields, primatology has a long history of research conducted by women (see “Special Topic: Women in Primatology”). Primatologists come from many different disciplines, have diverse backgrounds, and study primates for different reasons. Biologists study primates as examples of evolutionary theories like natural selection, and to understand behaviors as adaptations, or traits with a function that increases fitness, i.e. an individual’s survival and/or reproduction. Primate intelligence is of interest to psychologists who want to learn more about deception or cooperation and to linguists interested in the principles of communication and language. Ecologists consider how primates interact with the habitats they occupy, and conservationists examine how primates are affected by deforestation, poaching, or illegal animal trade (see Appendix B: Primate Conservation for more information on these topics). Biological anthropologists, like myself (Figure 7.1), who study primates are interested in learning about their social complexity, and ecological and behavioral variation, to better understand the biological basis of human behavior. And, similar to biologists, we also explore how primate behavior is adaptive and contributes to individual fitness. Like other sciences, primatology is only as strong as its researchers, methods, and theories, and the field has benefitted recently from efforts to increase diversity and reckon with its colonialist past, as discussed below in “Special Topic: Women in Primatology.”
Humans share many traits in common with primates. As you learned in Chapter 5, some of these traits are similar due to homology, traits both species inherited from a common primate ancestor. For example, like most other primates, humans are social animals who live in groups. Group living did not evolve independently in humans and other primates. Rather, group living is a trait that evolved in a primate ancestor, and because it benefited survival, it was retained in the species’ descendants (or the species that come after the ancestor species). In contrast, humans and other primates can have similar traits that evolved independently, which is called analogy. For example, both humans and Japanese macaques (Macaca fuscata) use natural hot springs (Figures 7.2a-b). Research on these monkeys indicates that sitting in hot springs reduces stress and helps keep them warm, much as it does for humans (Takeshita et al. 2018). But this behavior is not the result of humans and Japanese macaques having a shared ancestor who used hot springs. Rather, the behavior arose independently in two species that both occupy northerly environments and adapted to cold climates using a similar behavior. Studying the homologous traits we share with other primates, like living in groups, helps us develop hypotheses about human behaviors as adaptations, which in turn helps us develop models for the behavior of our human ancestors. Studying analogous traits, like hot springs use, allows us to better understand the effects of ecological variables on morphology and behavior of both primates and humans, living and extinct.

Special Topic: Women in Primatology
While many STEM fields have traditionally been, and continue to be, dominated by men, primatology has a long history of significant research conducted by women. This is due, in part, to the fact that three of the most well-known primatologists are women. In the early 1960s, British paleoanthropologist Louis Leakey (discussed in Chapters 9 and 10) was looking for students to study the great apes in hopes of shedding light on the behaviors of our early ancestors. He chose Jane Goodall (Figure 7.3a) to study chimpanzees (Pan troglodytes), Birute Galdikas (Figure 7.3b) to study Bornean orangutans (Pongo pygmaeus), and Dian Fossey (Figure 7.3c) to study mountain gorillas (Gorilla beringei beringei). The work of these three women, sometimes referred to as Leakey’s “Trimates,” has transformed our understanding of ape (and primate) behavior.

Arriving at the Gombe Stream Reserve in Tanzania in 1960, Jane Goodall was one of the first scientists to conduct a long-term study of wild nonhuman primates. Before then, most studies lasted less than a year and were often zoo-based. By 1961, she had made two astounding observations that forced us to reconsider what differentiates humans from the rest of the primate order. She observed chimpanzees eating a colobus monkey, the first reported evidence of meat eating in our closest relatives (she later observed them hunting and sharing meat). And she discovered that chimpanzees make and use tools by stripping leaves off twigs to “fish” for termites. Her work, spanning several decades, has produced long-term data on chimpanzee mating strategies, mother-infant bonds, and aggression. In the mid-1980s, Goodall transitioned from field researcher to conservationist and activist, advocating for the humane use of nonhuman animals (Stanford 2017).
Birute Galdikas began her study of orangutans in Kalimantan, Borneo, in 1971. Hers was the first long-term study conducted on the Bornean orangutan. Galdikas and her colleagues have collected over 150,000 hours of observational data, focusing on the life histories of individual orangutans. While conducting behavioral research, Galdikas discovered that the pet trade and habitat loss were adversely affecting the orangutan population. Eventually, Galdikas’s conservation efforts began to extend beyond advocacy and into rehabilitation and forest preservation (Bell 2017). If you would like to learn more about primate conservation efforts, please see Appendix B: Primate Conservation.
In 1967, Dian Fossey began her long-term study of mountain gorillas and founded the Karisoke Research Center in Rwanda. Her and her colleagues’ research, over several decades, revealed much about gorilla social behavior, ecology, and life history. Her efforts also led to the development of mountain gorilla conservation programs. However, she was a controversial figure, as discussed below. Fossey was murdered in December 1985; the case remains unsolved (Stewart 2017).
Decolonizing Primatology
Recently, the movement to decolonize primatology, by understanding and highlighting the theories and research of non-Western individuals and perspectives, has gathered steam. This movement draws attention to the maltreatment of local people by Western primatologists. For example, Michelle Rodrigues (2019) argues that it's time we stop focusing on the scientific and conservation contributions of Dian Fossey and acknowledge that her "active conservation" techniques included kidnapping and torturing local Rwandans who were known as, or suspected to be, gorilla poachers. Rodrigues (2019) argues:
The image of Fossey, a white American woman, whipping and torturing black African poachers is evocative of the behavior of white slaveholders in the American South. It is appalling enough to think of that behavior occurring in the 1850s; there is no way we can explain Fossey’s behavior in the 1970s as the product of “a different time.” Yet, almost three decades later, the romantic notion of a noble martyr who died for her devotion to gorillas prevails, and these terrifying actions are often described as simply unorthodox methods. Perhaps these truths are softened due to fears that the reality of this legacy would harm gorilla conservation efforts. But memorializing her as a martyr and patron saint of gorilla conservation demands that we forget the cruel acts she advocated for and performed.
Further, Louis Leakey’s installment of Goodall, Galdikas, and Fossey to study chimpanzees, orangutans, and mountain gorillas, respectively, is itself viewed as recapitulating the colonial legacy in Africa and Asia. Given that Leakey was the offspring of British missionaries, Rodrigues (2019) argues, it is no accident that he was willing to mentor British and American women, while overlooking women from Africa and Asia as potential researchers. This leads us to another level of the decolonizing movement, which aims to highlight the research of non-Western primatologists, particularly those living in what primatologists refer to as “habitat countries” that are home to living primates. As you will see in this chapter, scientists from diverse backgrounds are active contributors to exciting research on primates around the world.
Ecology
The more than 600 species and subspecies of living primates are highly diverse in their dietary preferences and the habitats they occupy. In this section we’ll briefly discuss aspects of ecology, or the relationship between organisms and their physical surroundings, that impact a primate’s life, the foods they eat, and the other species with whom they interact.
Primate Diets
Diet may be the most important variable influencing variation in primate morphology, behavior, and ecology. Most primates are omnivores who ingest a variety of foods in order to obtain appropriate levels of protein, carbohydrates, fats, and fluids, but one type of food often makes up the majority of each species’ diet. You learned about the dental and digestive adaptations of frugivores (who feed primarily on fruit), folivores (whose diet consists mostly of leaves), and insectivores (who eat mainly insects) in Chapter 5, so we will not discuss them again here.
Body Size and Diet

Insects are a high-quality food, full of easily digestible protein and high in calories that meet most of a primate’s dietary needs. Although all primates will eat insects if they come upon them, those species that rely most heavily on insects tend to be the smallest. Why? Because larger primates simply cannot capture and consume enough insects every day to survive. Because of their small size (less than 150 g), spectral tarsiers (Tarsius spectrum) have a fast metabolism, which means they turn food to energy quickly, but they do not need to consume large amounts of food each day. It does not matter to a spectral tarsier that a grasshopper only weighs 300 mg, because the tarsier (Tarsius) itself is so small that one grasshopper is a good-size meal (Figure 7.4a). That same grasshopper is not even a snack for an adult male mountain gorilla (Gorilla beringei beringei), who may weigh up to 200 kg. Fortunately for gorillas (Gorilla), their large body size means they have a slow metabolism, converting food into energy much more slowly, so they can eat lower quality food that takes longer to digest, provided there is a lot of it. For gorillas, leaves, which are hard to digest but plentiful, fit the bill (Figure 7.4b). Most medium-sized primates are highly frugivorous, and supplement their fruit based diet in ways that correspond with their size: Smaller frugivores tend to supplement with insects, while larger frugivores tend to supplement with leaves.
Food Abundance and Distribution
Nutrients are not the only dietary considerations primates must make. They must also ensure that they consume more calories than they use. The abundance and distribution of food affect energy expenditure and calorie intake because they determine how far animals must travel in search of food and how much they must compete to obtain it. Abundance refers to how much food is available in a given area while distribution refers to how food is spread out. In terms of abundance, food is either plentiful or scarce (Figure 7.5a–b). Food is distributed in one of three ways: uniformly (Figure 7.6a), in clumps (Figure 7.6b), or randomly (Figure 7.6c). In general, higher-quality foods, like fruit and insects, are less abundant and have patchier distributions than lower-quality foods, like leaves. Primates who eat fruit or insects usually have to travel farther to find food and burn more calories in the process. Abundance and distribution of food is another reason why larger primates tend to rely more heavily on leaves than either fruit or insects.
Community Ecology
Primates are members of broader ecological communities composed of other species, including other primates, predators, parasites, and even humans. Community ecology deals with the relationships and interactions between different organisms that occupy the same habitat. Interactions with conspecifics (members of the same species) and heterospecifics (members of different species) are critical aspects of ecological communities. Some habitats support highly diverse primate communities consisting of 10 or more primate species. How can so many primate species occupy the same area and avoid competition? In most cases, the primate species that live together occupy different niches, which means they do not meet their needs for food and shelter in the exact same way. Two species can avoid competition by eating different kinds of food, living at different levels of a forest, or even searching for food at different times of day. Because tropical rainforests, like Manu National Park in Peru, are highly variable, with many habitats and many sources of food and shelter, there are many different niches for multiple species to exploit, and large primate communities can result (Figure 7.7).

Competitive Interactions
Although species living in the same location often occupy different niches to avoid competition, when a resource that is important for survival or reproduction is scarce, individuals will compete to obtain that resource. This is a central tenet of Charles Darwin’s theory of evolution by natural selection (see Chapter 2). Competition between primates takes two forms: Individuals engage in direct competition, which involves physical interaction between individuals (such as fighting), over resources that are large and worth defending (fruit is a good example of a food resource over which primates will fight). Individuals engage in indirect competition, in which there is no physical interaction between individuals, when a resource is small. Primates often engage in indirect competition for insects, like grasshoppers, that are eaten quickly, often before another individual arrives on the scene. Primates may engage in direct and/or indirect competition with members of their own group, with members of other groups of conspecifics, or with heterospecifics.
Predator-Prey Interactions
The plants and animals that primates eat are an important part of their ecological community. In addition to insects, many primates incorporate some vertebrate (animals with an internal spinal column or backbone) prey into their diet. Often, predation by primates is opportunistic, occurring because the prey happens to be in the right place at the right time. I’ve observed vervets (Chlorocebus pygerythrus) opportunistically killing lizards by smashing them against a rock or tree trunk and eating them. More rarely, hunting is deliberate and cooperative. In some chimpanzee (Pan troglodytes) populations, hunts involve multiple individuals, each of whom plays a specific role and is rewarded afterward with a share of the prey that has been captured (Samuni et al. 2018).
All primates are susceptible to predation by mammalian carnivores (animals whose diet consists primarily of animal tissue (e.g., Figure 7.8a), reptiles (e.g., Figure 7.8b), or birds of prey (e.g., Figure 7.8c). Although the specific predators found in an ecological community differ based on geography, smaller primates always fall prey to a wider range of predators. Because predators are diverse in their hunting tactics, primates have evolved a wide range of tactics to avoid or escape them. We will discuss some of these behavioral adaptations later in this chapter in the section titled “Why Do Primates Live in Groups?.”

Mutualistic Interactions
So far, we've discussed competitive and predator-prey interactions in primate communities. But there are some interactions (between different primate species and between primates and other species) that are mutualistic, which is when organisms of different species work together, each benefiting from the interaction or relationship. One example is seed dispersal, which is the process by which seeds move away from the plant that produced them in preparation for germination and becoming a new plant. When seeds are dispersed by animals, like primates, it is an example of mutualism. The primate eats the fruit of a plant, which provides nutrients for its body, and in the process ingests the plant’s seeds. Later, it deposits the seeds at another location as a pile of fertilizer.
Another example of mutualism is polyspecific associations, which are associations between two or more different species that are maintained by behavioral changes by at least one of the species. While some associations are short in duration, others are semi-permanent. The mutualistic benefits of polyspecific associations include one species gaining access to food that would otherwise have been inaccessible or being alerted to the presence of predators that they would not have not have known were present otherwise. In some cases, individuals seem to recognize and seek out specific members of another species. Twenty years of observations on chimpanzees and Western lowland gorillas (Gorilla gorilla gorilla) in the Republic of Congo has revealed social ties (some might call them friendships) between individual chimpanzees and gorillas that last for years and occur in a variety of social contexts, including play (Sanz et al. 2022).
Parasite-Host Interactions
Primates are hosts for a variety of parasites, which are organisms that live in or on another organism (the host). Parasites come in many forms and pose varying levels of danger to the host. Blood parasites cause diseases like yellow fever and malaria. Skin parasites include fleas and ticks, which feed on the host’s blood, and botflies, which lay eggs in the host’s flesh. Bot fly larvae feed on the host’s flesh as they develop and eventually (if not removed) break through the skin at maturity. Gut parasites, like tapeworms, get into the intestines and feed off of the food that is being digested by the host. Because most primates live in groups (see the “Primate Societies” section of this chapter), the tendency for social transmission of parasites, or the transfer of parasites from one individual to another, is high. Primates have evolved mechanisms to avoid parasite infection, including switching sleeping and feeding sites so as to avoid parasites. Mandrills (Mandrillus sphinx) have been shown to avoid grooming infected conspecifics as well as to avoid their feces, which smell different than the feces of individuals who are not infected with parasites (Poirotte et al. 2017). Other primates, including chimpanzees, appear to self-medicate when infected with parasites by ingesting plants that have antiparasitic properties (Krief et al. 2005).
Human-Primate Interactions
Humans are part of many primate communities and our relationship with our closest relatives is often complicated. In some areas, humans hunt primates for their meat or as trophies, or so they can sell the infants as pets. As the human population increases in size, our demand for natural resources, like wood to build houses or land on which to grow food, also increases, often at the expense of pristine primate (and other animal) habitat. As their natural habitat shrinks, primates search for food in areas occupied by humans and may be shot as crop-raiding pests. While deforestation, hunting, and the pet trade are examples of ways in which humans negatively affect the lives of primates, some human-primate interactions are beneficial. In some parts of the world primates are central to ecotourism, which focuses on nature-based attractions to educate tourists and uses economically and ecologically sustainable practices. Perhaps one of the greatest success stories of ecotourism involves the mountain gorillas of Rwanda (see Figure 7.4b). After internal conflict plagued Rwanda during the 1990s, the Virunga Mountains area developed gorilla-based tourism to aid in socioeconomic development and to bring stability to the region. This process not only helped to increase mountain gorilla populations but was also able to generate enough income to cover the operation costs of three national parks and provide income and other benefits to people living in the area (Maekawa et al. 2013). You can learn more about human-primate interactions in Appendix B: Primate Conservation.
Primate Societies
Unlike many other animals, primates are highly social and many live in stable groups consisting of adult males and females, even outside the breeding season, when females are receptive and available for mating because they are not pregnant or nursing. Indeed, sociality, or the tendency to form social groups, is a key behavioral adaptation of the order primates (see Chapter 5). This has led primatologists to ask two questions: “Why do primates live in groups?” and “What types of groups do primates live in?”
Why Do Primates Live in Groups?
Primates live in groups when the benefits of doing so exceed the costs. Although there are many potential benefits to group living, enhanced feeding competition and predator avoidance are important benefits for many group living primates. When primates feed on high-quality, scarce food (like fruit), larger groups are more successful in competition with other groups. For example, in a long-term study of vervets in Kenya’s Amboseli National Park, larger vervet groups had larger and better home ranges, which is the area in which a group regularly moves around as it performs its daily activities, including searching for food and water. Females in larger groups had higher average infant and female survival rates than the smallest group. Because pregnancy and nursing are energetically expensive for females, female reproductive success, or genetic contribution to future generations (measured by the number of offspring produced), is limited by access to food. Although living in a group means females compete with members of their own group for food, the benefits of being a member of a larger vervet group outweigh the costs (Cheney and Seyfarth 1987).
However, because they contain more individuals, larger groups are more likely to attract the attention of predators compared to smaller groups. This is one of the reasons that primates who rely on crypsis, or the ability to avoid detection by others, including predators, are often solitary (the term used to describe individuals who do not live together with other members of their species) and nocturnal, or active at night. If an animal is already hard to see because it is active at night, then moving quietly in small groups is a good strategy to avoid detection by predators. The slow loris (Nycticebus coucang) of Southeast Asia is a good example of this strategy (Figure 7.9a). Nocturnal and solitary, the slow loris moves slowly and quietly as its primary strategy to avoid detection (Wiens and Zitzmann 2003). In contrast, primates who live in large groups and are diurnal, or active during the day (like gelada baboons [Theropithecus gelada]; Figure 7.9b) cannot avoid detection by predators. Instead, group-living primates rely on behaviors that alert others to the presence of danger and/or deter predators, including shared vigilance (watchful behavior to detect potential danger), mobbing (the act of cooperatively attacking or harassing a predator), and alarm calling (vocalizations emitted by social animals in response to danger). We will discuss alarm calls in the Communication section.

What Types of Groups Do Primates Live In?
Primates vary with regard to the types of groups in which they live. A social system describes a set of social interactions and behaviors that is typical for a species. The components that make up a species’ social system include:
- Group size, which refers to the number of individuals that typically live together. Primate group size can be highly variable, ranging from one or a few individuals, to a few dozen, upward to several hundred individuals.
- Group composition describes group membership in terms of age class (e.g., adult, juvenile, infant) and sex. In some primates, groups consist of a mother and her dependent offspring while in others, one adult male lives long-term with one adult female and their dependent offspring. In other species, one or more adult males live with multiple females and their offspring.
- A species’ mating system refers to which male(s) and female(s) mate. The terms that describe a mating system (e.g., polygyny, in which one male mates with multiple females) are sometimes used to describe a primate species’ social system, but a mating system is one component of the species’ social system. For example, two species might both have polygynous mating systems, but in one species, the group is composed of one male and multiple females, while members of the other species live as solitary individuals.
- Ranging behavior refers to the way in which animals move about their environment. Most primate species have a home range, where they perform their daily activities. Some primates defend a territory which is the part of the home range that the group actively guards in an attempt to keep out conspecifics.
- Dispersal patterns describe which sex moves to a new group to reproduce. In most primate species, males disperse because the benefits of dispersal, including increased access to mates and reduced competition from other males, outweigh the costs of migrating into a new group, which often comes with aggression from current group members. For many female primates, the opposite is true: females usually benefit from remaining philopatric, or in the group of their birth. This allows them to maintain strong alliances with female relatives, which helps them compete successfully against other groups for food. In solitary species, offspring of both sexes leave their mother’s home range and become solitary. If this did not happen, the species would not be solitary. Even though both sexes disperse in solitary species, males usually disperse farther than females.
- Social interactions describe the ways in which individuals interact with members of their own and other groups of conspecifics. Affiliative (i.e., friendly or nonaggressive) behaviors include grooming (picking through the fur of another individual), playing, or coalitions (temporary alliances between individuals). Agonistic (i.e., aggressive) behaviors include fighting over food or fighting over access to mates. In groups that contain multiple adult individuals of the same sex, it is common to have a dominance hierarchy, or a group of individuals that can be ranked according to their relative amount of power over others in the hierarchy. Initially, dominance hierarchies are established through the outcome of conflicts. Individuals who lose conflicts with others are subordinate (or low rank) to those who win them. Those who win conflicts are dominant (or high rank). Dominant individuals gain access to resources, like food or mates, before subordinates. Once a hierarchy is established, agonism decreases because everyone “knows their place.”
The main types of primate social systems are as follows: solitary; single-male, single-female; single-male, multi-female; multi-male, multi-female; fission-fusion; and multi-male, single-female. These types are discussed below.
Solitary
Recall that the term solitary is used to describe species in which individuals do not live or travel together with other members of the same species, except for mothers and unweaned offspring. Males typically occupy a large home range or territory that overlaps the home ranges of multiple females, with whom they mate (Figure 7.10a). Because one male mates with multiple females, the mating system of solitary primates is polygyny. Social interactions between adults are limited but because some males do not get to mate, competition between males is intense. When males compete physically, they benefit from large body size and weaponry. The result is sexual dimorphism, when males and females look different from one another. Both males and females disperse, although males move farther from their mother than females. The nocturnal West African potto (Perodicticus potto; Figure 7.10b) is solitary. Bornean orangutans, which are diurnal, are also solitary.
Single-Male, Single-Female
Primate species in which an adult male and adult female live together with their dependent offspring have a single-male, single-female social system, sometimes referred to as a “family,” with group sizes between two and five individuals. The adult male and adult female engage in behaviors that strengthen their social relationship, or pair bond, including mutual grooming and resting together. The pair defend a territory (Figure 7.11a) and keep same-sex individuals away from their mate. The adult male and adult female mate with each other, so the mating system is monogamy, although mating outside the pair bond may occur. Species with monogamous mating systems are usually sexually monomorphic (males and females look similar) because competition for mates is relaxed since most males are able to obtain a mate. Males are usually confident that they are the father of their mate’s infant, so they help with offspring care by carrying the infant when it is not nursing. Once offspring are sexually mature, both males and females disperse. As with solitary species, males disperse farther from their parents than females. Bolivian Gray titi monkeys (Plecturocebus donacophilus) are an example of a species that has a single-male, single-female social system. One of their signature behaviors is tail twining, when two individuals sit with their tails wrapped around each other (Figure 7.11b). This behavior reinforces the social bond among family members and is especially common between the adult male and female. Gibbons (Hylobates) and owl monkeys (Aotus) also live in single-male, single-female groups.
Single-Male, Multi-Female
Single-male, multi-female groups consist of one adult male living with multiple adult females and their dependent offspring (Figure 7.12a ) . These groups can range from as few as five or ten individuals to as many as 50. Female social relationships are governed by the female dominance hierarchy. Females are usually philopatric and males disperse. Males who are unable to join a group of females may join a bachelor group with other males. Because a single male mates with multiple females, the mating system is polygyny. Species that form single-male, multi-female groups may or may not defend a territory, but the resident male, who lives with a group of females, is aggressive toward other males, who may try to take over the group and become the new resident male. Competition between males to be the resident male of a group is intense, and these species usually display sexual dimorphism, with males being larger than females and possessing large canines. Hanuman langurs (Semnopithecus entellus) of India form single-male, multi-female groups (Figure 7.12b). When a new male takes over a group of females and ousts the former resident male, he may commit infanticide, or kill the unweaned infants. This is especially likely if the new resident male has not yet mated with any of the females and thus cannot be the infants’ father. This causes the females, who were nursing, to become sexually receptive sooner, increasing the new resident male’s chances of producing offspring (Sharma, Ram, and Raipurohit 2010). Gorillas, patas monkeys, and golden snub-nosed monkeys (Rhinopithecus roxellana) also live in single-male, multi-female groups.

Multi-Male, Multi-Female
Multi-male, multi-female groups consist of multiple adult males living with multiple adult females and their dependent offspring. Although there is more than one adult male, there are more adult females than adult males in the group (Figure 7.13a). Multi-male, multi-female groups can range in size from about ten to as many as 500 individuals. They occupy a home range but may or may not defend a territory. In groups that contain multiple males and multiple females, it is not possible for one male to monopolize all the matings, so the mating system is polygamy, in which multiple males mate with multiple females. However, this does not mean that all males have an equal opportunity to mate with all females. In multi-male, multi-female groups, both males and females form a dominance hierarchy. The male dominance hierarchy determines their access to females for mating in much the same way that a female dominance hierarchy determines a female’s access to food. Because their place in the hierarchy can affect their reproductive success, males compete with each other, but because it is rare for males to be excluded from mating altogether, the level of competition and degree of sexual dimorphism are less extreme than what we see in polygynous species. Usually, females are philopatric and males disperse. Vervet monkeys (Figure 7.13b), ring-tailed lemurs (Lemur catta), white-faced capuchins (Cebus capucinus), and black-capped squirrel monkeys (Saimiri boliviensis) live in multi-male, multi-female groups.

Fission-Fusion
Fission-fusion is a fluid social system in which the size and composition of the social group changes, with groups splitting (fission) or merging (fusion) depending on food availability (Pinacho-Guendulain and Ramos-Fernández 2017). When key resources are scarce, individuals spread out (fission) and move and feed individually or in small subgroups (Figure 7.14a). When key food resources are plentiful, individuals come together (fusion) and individuals travel and feed as a more cohesive group (Figure 7.14a). Fission-fusion social structure is believed to reduce feeding competition when resources are scarce. Because group composition changes over time, species with fission-fusion social systems are referred to as a community. Communities consist of multiple adult males, multiple adult females, and offspring, and group size varies but typically ranges from ten to a few dozen individuals. Females typically disperse and males are philopatric. Thus, community males are related and display unusual forms of cooperation. The mating system associated with fission-fusion is polygamy. Because males are not excluded from mating, competition for mates is relaxed and sexual dimorphism is moderate (males are slightly larger than females). Geoffroy’s spider monkeys (Ateles geoffroyi) (Figure 7.14b) and chimpanzees both have fission-fusion social system.
Multi-Male, Single-Female
In multi-male, single-female groups, two or more males live with one breeding female, her dependent offspring, and non-breeding females (Figure 7.15a). This type of social system is found in the callitrichids, the primate family that includes marmosets (Callithrix; Figure 7.15b) and tamarins (Saguinus) of Central and South America. Their groups rarely exceed 15 individuals, and each group actively defends their territory from conspecifics. Although more than one adult female may live in the group, the mating system is polyandry because there is only one breeding female who mates with all of the adult males. This is achieved through reproductive suppression, which involves the breeding female preventing other females from reproducing through physiological and/or behavioral means (Digby, Ferrari, and Salzman 2011). This limits the opportunities for other females in the group to become pregnant. Instead, these females, and the males in the group, help raise the breeding female’s offspring. This is referred to as cooperative breeding and usually takes the form of carrying infants, grooming them, and protecting them from danger (de Oliveira Terceiro and Burkart 2019). Because reproductive opportunities for female tamarins and marmosets are limited, they are very competitive, and females are slightly larger than males, which helps them compete for the breeding spot in a group.

Reproductive Strategies
Reproductive strategies have evolved to maximize individual reproductive success. These strategies can be divided into those that deal with offspring production and care (parental investment) and those that maximize mating success (sexual selection). Because the reproductive physiology of male and female primates differs, males and females differ with regard to parental investment and sexual selection strategies. Female strategies focus on obtaining the food necessary to sustain a pregnancy and choosing the best male(s) to father offspring. Male strategies focus on gaining access to receptive females.
Parental Investment

Biologically speaking, parental investment is any time or energy a parent devotes to the current offspring that enhances its survival (and eventual reproductive success) at the expense of the parent’s ability to invest in the next offspring (Trivers 1972). Female primates invest more heavily in offspring than males. Even before conception, females produce energy-containing eggs, and they will be responsible for sustaining a fertilized egg until it implants in the uterus. After that, they invest in pregnancy and lactation (Figure 7.16). Because all of this investment requires a lot of energy, female primates can only produce one offspring (or litter) at a time. A species’ interbirth interval, or the typical length of time between one birth and the next, is determined by the length of time necessary to maximize each offspring’s survival without jeopardizing the female’s ability to produce the greatest number of offspring possible. If a female invests too little (i.e., weans an offspring too early), she may give birth to many offspring, but very few (if any) of them will survive. If she invests too much (i.e., nurses an offspring even after it could be weaned), she ensures the survival of that individual offspring but will not be able to produce very many during her lifetime. To maximize her reproductive success, a female must invest just long enough to ensure the greatest number of offspring survive to reproduce. We often think of maternal care as an innate (or natural), instinctive behavior. Yet this is not the case. The “Special Topic: Is Maternal Behavior Innate?” dispels the myth that maternal behavior is solely instinctual and explains how female primates learn to be good mothers.
Sexual Selection
Sexual selection, or selection for traits that maximize mating success, comes in two forms. Intrasexual selection is selection for traits that enhance the ability of members of one sex to compete amongst themselves (“intrasexual” = within one sex). Intersexual selection is selection for traits that enhance the ability of one sex to attract the other (“intersexual” = between the sexes).
Intrasexual selection most often operates on males. In the wild, adult females are either pregnant or lactating for most of their adult lives. So, in a given population, there are usually more males available and willing to mate than there are females. The result? Females are a scarce resource over which males compete. Intrasexual selection favors traits that help a male win fights with other males. In primates, these traits include large body size (Figure 7.17a) and large canines (Figure 7.17b). Because females don’t possess these same traits, males and females of some species look different; that is, they are sexually dimorphic (Figure 7.17a).

Intersexual selection also tends to operate on males, selecting traits that make a male more attractive to females. Females, in turn, choose among potential fathers. Because female primates invest more in offspring production and care than males (see the “Parental Investment” section, above), it is more costly for them if the offspring dies before maturity or reaches maturity but does not reproduce. Thus, it benefits a female primate to be choosy and try to pick the healthiest male as a mate. Males must display traits that tell a female why she should choose him, and not another male, as her mate.
What traits are female primates looking for? In humans, women may look for a mate who can provide important resources, such as food, paternal care, or protection. This is rare in other primates, though, since most females do not need males to provide resources. More commonly, female primates obtain genetic benefits for their offspring from choosing one male over another. Often the specific criteria by which females select mates is unknown. However, if a female chooses a healthy (as indicated by traits like a plush coat, bright coloration, or large body size) or older male, she may obtain genes for her offspring that code for health or long life. If a male’s rank is determined by competitive ability that has a genetic component, females who choose males who win fights may acquire these genes (and qualities) for their offspring. Females in some species appear to prefer new immigrants, sometimes even “sneaking” copulations with males who are not established members of their groups. Such a preference may provide their offspring with novel genes and increase genetic variation (for more about the importance of genetic variation, see Chapter 4). Female choice is often more subtle than male-male competition, so it can be more difficult to study. However, as more research is conducted, we continue to improve our understanding of the ways that female primates exert their choice.
Special Topic: Is Maternal Behavior Innate?
Zoos almost always have nurseries where infants are cared for by zookeepers if their mothers will not care for them (Figure 7.18). These exhibits are among the most popular because the babies are so cute and so much fun to watch. And the caretaking positions in zoo nurseries are often among the most coveted by zoo personnel for the same reasons. But if maternal behavior is instinctive, why do zoo nurseries even exist? The answer is that in many species, including primates, maternal behavior is not purely instinctual; it is dependent on social learning (behavior learned by observing and imitating others), as well.

Captive female primates, including gorillas and chimpanzees, who have not had the opportunity to observe their mother or other females care for infants do not know how to care for their own offspring. Although it is preferred that the primate mother care for her own infant, there are cases when she will not and humans must step in to ensure the offspring survives. When hand-rearing by humans is necessary, the infant is returned to the group as soon as possible in the hopes that it will learn species-typical behavior from its mother and other conspecifics. Observations such as these indicate that maternal behavior is learned, not innate, and that maternal care is critically important to the social and psychological development of young primates.
Communication
In its most basic form, communication occurs when one individual (the sender) emits a signal that conveys information, which is detected by another individual (the receiver). We have discussed several aspects of primate sociality in this chapter, all of which require the communication of information between individuals. But exactly how does a female chimpanzee communicate her sexual availability? How does a vervet monkey communicate the approach of a leopard or that a python is nearby? How do solitary, nocturnal primates, like the slow loris, communicate information about themselves to conspecifics? Primate communication comes in four forms: vocal, visual, olfactory, and tactile. Species vary in their reliance on each.
Vocal Communication
Primates use sound to communicate danger or threats, to claim and maintain a territory, or make contact with other group members. Alarm calls are given in response to predators. In some cases, alarm calls are used to alert members of the group to the presence of a predator so they can take evasive action. In other cases, they are directed at the predator itself, signaling that it has been detected. You can learn more about alarm calls as forms of vocal communication in the highlight box in this chapter entitled “Dig Deeper: Alarm Calls: Signals to Friends or Foes?.”
Loud calls are designed to travel great distances and are used in territorial defense by many primate species including indris (Indri indri), orangutans, gibbons, and howler monkeys (Alouatta). In dense forest, where visual communication can be difficult, loud calls can be useful in signaling to conspecifics that a group or individual occupies a specific area. Howler monkeys are named for their loud calls, or “roars,” which can be heard one kilometer or more away (Schön Ybarra 1986). Howler monkey roars may act to maintain distance between neighboring groups or keep extragroup males from entering the home range (Schön Ybarra 1986).
Other vocalizations are intended to communicate with individuals in one’s own group. These include vocalizations given as part of threat displays or dominance interactions, as well as contact calls that provide information about one’s location to other group members. Chacma baboons (Papio ursinus) have a rich repertoire of vocalizations for communicating with other group members (Fischer et al. 2008). Adult males give specific vocalizations during threat displays and physical confrontations. Subordinates “screech” when retreating from a dominant individual, signaling submission. Since baboons rely on membership in their group for finding food and detecting predators, a baboon separated from his group will vocalize in an attempt to regain contact. Young baboons emit their own contact calls when separated from their mothers.
Visual Communication

Visual communication, which involves signals that can be seen, is an important component of nonhuman primate behavior, alone or in combination with other forms of communication. Piloerection, or raising one’s hair or fur, is used in aggressive interactions to make an individual appear larger than it actually is. Female macaques (Macaca), baboons (Papio), and chimpanzees, signal sexual receptivity through changes in the size, shape, and, often, color of their hindquarters, called a sexual swelling (Figure 7.19a). The sexual swelling reaches its maximum size at ovulation. When females are not receptive, either because they are pregnant or are nursing, they do not display a sexual swelling (Figure 7.19b). Thus, the presence or absence of a sexual swelling signals a female’s reproductive state.
Monkeys and apes use diverse facial expressions in visual communication. Showing your teeth in a “smile” sends a signal of friendship in humans. Displaying teeth in this way is a sign of anxiety or fear in primates. That male mandrill you see “yawning” at your local zoo is actually displaying his teeth to signal tension or to threaten a rival (Figure 7.20a). In addition to showing their canines, male gelada baboons use “lip flips,” in which the gums and teeth are exposed by flipping the upper lip up over the nostrils (Figure 7.20b), and “raised eyelids,” in which the pale eyelids are exposed by pulling the scalp back as threatening gestures (Aich, Moos-Heilen, and Zimmerman 1990). Submissive males respond by fleeing or presenting their hindquarters.



Primates also communicate through color. In female and male mandrills, facial coloration provides information about an individual’s health, competitive ability, and reproductive state to conspecifics (Figure 7.21; Setchell et al. 2008; Setchell, Wickings, and Knapp 2006). Variation in facial coloration among monkeys of Central and South America ranges from very simple (Figure 7.22a) to complex (Figure 7.22b). Species living with larger numbers of other primate species have evolved more complex facial coloration patterns, suggesting that this trait evolved as a form of species recognition, or the ability to differentiate conspecifics from members of other species (Santana, Lynch Alfaro, and Alfaro 2012).
Olfactory Communication
All primates use scent to communicate. Females secrete chemicals from their anogenital region (the area of the anus and genitals) that provide males with information about their reproductive state. In some species, like macaques and chimpanzees, this olfactory signal is enhanced by a sexual swelling, as discussed above. Olfactory communication, or communicating through scent, is particularly important for monkeys of Central and South America, lemurs, and lorises. Male and female common squirrel monkeys (Saimiri sciureus) (Figure 7.23a) engage in “urine washing,” in which an individual urinates on its hands and feet and then uses them to spread urine all over its body. Urine washing may be used to mark trails for others to follow, to control body temperature, as part of dominance displays, or to communicate reproductive state (Boinski 1992). During aggressive interactions with other males, male ring-tailed lemurs rub their tails with scent from glands on their wrists and chests. They use their “perfumed” tails in aggressive interactions with other males, who may respond by waiving their own scented tail, with physical aggression, or by fleeing (Jolly 1966). Males also waive their tails, saturated in scent, to attract females (Shirasu et al. 2020). Males use scent glands in their wrists to mark territorial boundaries (Figure 7.23b; Mertl-Millhollen 1988).

Tactile Communication
Tactile communication, or communicating through touch, is very important in all primate species. Physical contact is used to comfort and reassure, is part of courtship and mating, and is used to establish dominance and alliances. Grooming is an important and clearly enjoyable form of tactile communication for all primates (Figure 7.24). Not only does grooming serve to clean the skin and fur, removing parasites and debris, but it is an important affiliative behavior that helps reinforce social bonds, repair relationships, and cement alliances.

Dig Deeper: Alarm Calls: Signals to Friends or Foes?
Alarm calls are common among group-living primates. They often serve to notify conspecifics of potential danger, as is the case with vervet monkeys. Research has shown that: (1) vervets classify predators based on hunting style; (2) alarm calls convey information to other vervets about that hunting style; and (3) other vervets respond in ways appropriate for evading that type of predator (Seyfarth, Cheney, and Marler 1980a). When a vervet gives a “leopard” alarm call (directed at mammalian carnivores like leopards, Figure 7.25a), monkeys on the ground climb the nearest tree, while monkeys already in trees stay there or climb higher. Since most mammalian carnivores hunt on the ground, getting into, and staying in, a tree is the best option for escape. When the “snake” alarm call is given, vervets stand on their hind legs and look down at the ground (Figure 7.25b). Since snakes are not pursuit predators, locating them quickly so as to avoid them is the best strategy. Lastly, when an “eagle” alarm call is given, vervets look up or run into bushes, both of which are useful responses for avoiding hawks and eagles, which attack from above (Figure 7.25c). Vervets clearly understand the meaning of each type of alarm call, as they respond appropriately even when they do not see the actual predator (Seyfarth, Cheney, and Marler 1980b). Such semantic communication, which involves the systematic use of signals to refer to objects in the environment, was once believed to be unique to humans. It may be a precursor to the symbolic capacities of human language.
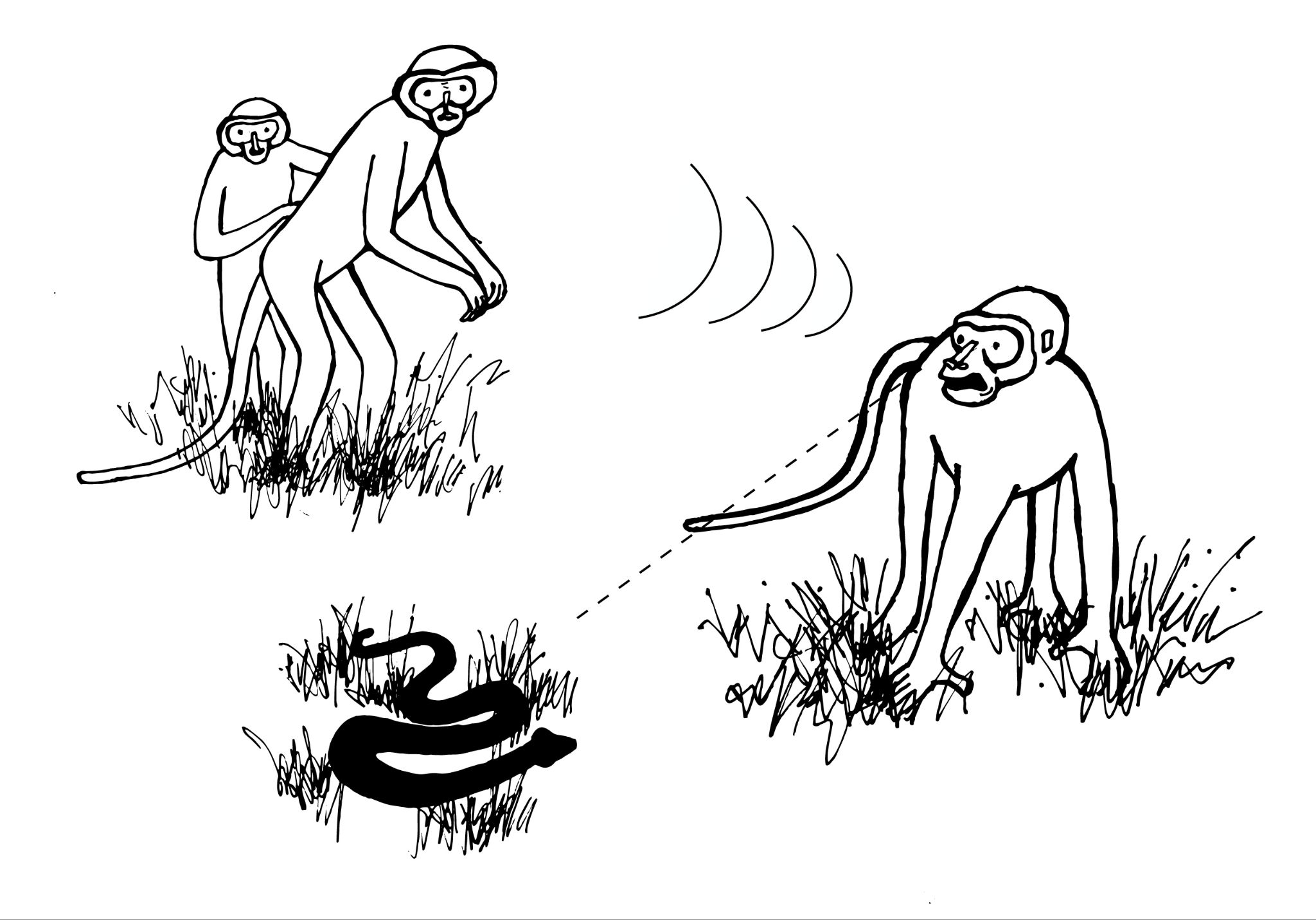
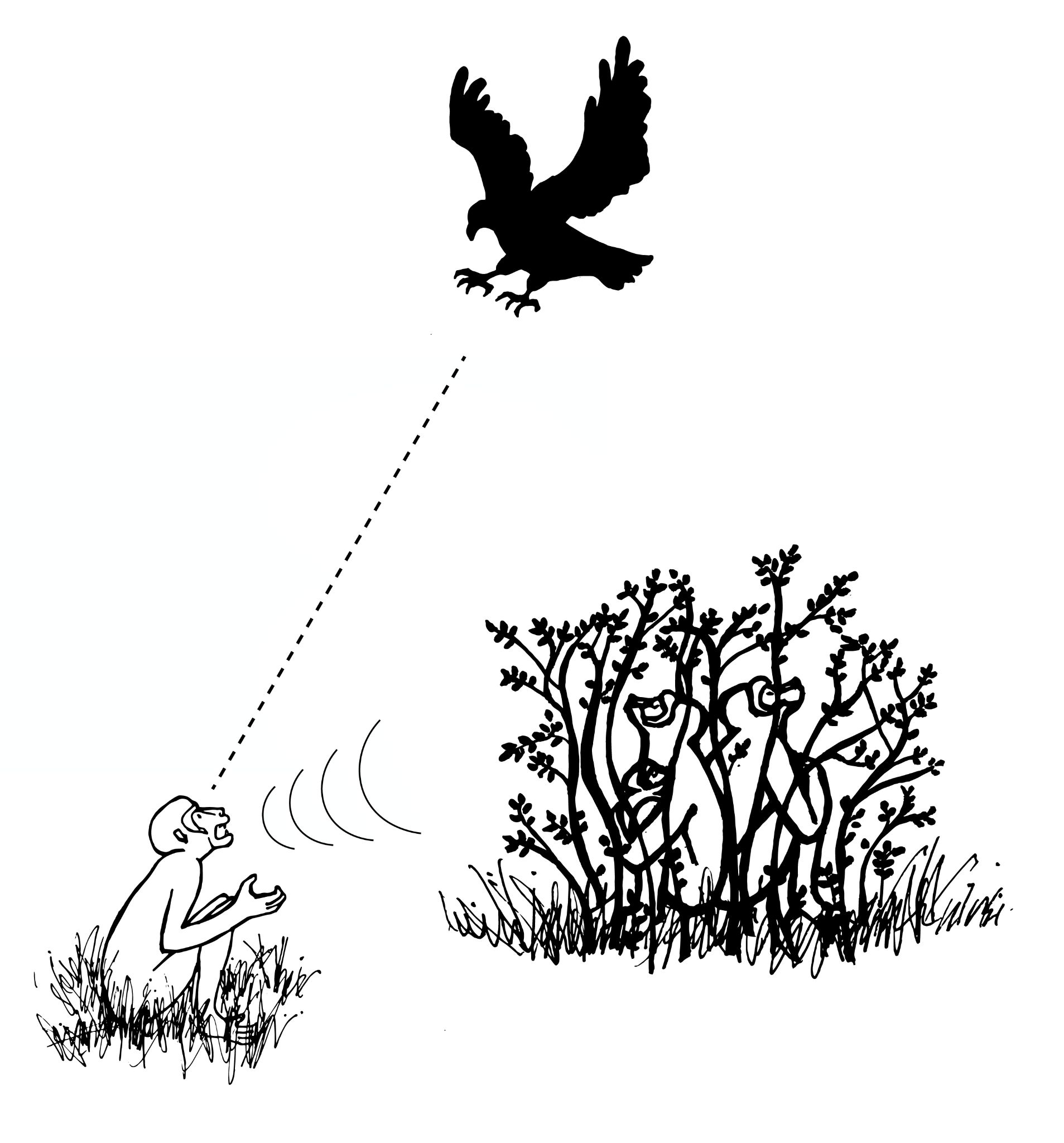
Research on other African monkeys indicates that some species use alarm calls to signal to the predator that it has been detected. Diana monkeys (Cercopithecus diana) give alarm calls to leopards (Panthera pardus) but not chimpanzees (Zuberbühler, Noë, and Seyfarth 1997). Because leopards are stealth predators, they rely on the element of surprise to sneak up on their prey (Figure 7.26a). Alarm calling at leopards appears to tell the leopard that it has been seen and therefore its chance of success will be low. Research shows leopards are more likely to stop hunting after an alarm call has been emitted. Unlike leopards, chimpanzees are pursuit predators and may even use alarm calls to locate potential prey (Figure 7.26b). With such a predator, prey are better off remaining as silent as possible so as not to alert the predator to their location (Zuberbühler et al. 1999).

The Question of Culture
It may be surprising in a chapter on nonhuman primates to see a discussion of culture. After all, culture is considered by many, including cultural anthropologists, to be a distinguishing characteristic of humans. Indeed, some anthropologists question claims of culture in primates and other animals. Definitions of animal culture focus on specific behaviors that are unique to one population. Anthropological definitions of human culture emphasize shared ideology (e.g., values, morals, beliefs) and symbols, not just behavior. Using this definition, some cultural anthropologists view primates as lacking culture because of the absence of symbolic life (e.g., religion). However, the longer we study primate groups and populations, the more insight we gain into primate behavioral variation. If we define culture as the transmission of behavior from one generation to the next through social learning, then we must view at least some of the behavioral variation we see in primates as forms of cultural tradition, or a distinctive pattern of behavior shared by multiple individuals in a social group that persists over time (Whiten 2001).
Chimpanzee Culture
Due to both their high level of intelligence and the large number of long-term studies on several different populations, chimpanzees provide the best example of cultural tradition in primates. Chimpanzees express cultural variation in multiple behavioral patterns, ranging from population-specific prey preferences and hunting strategies to tool-use techniques and social behaviors. For example, in Tanzania, chimpanzees fish for termites by stripping twigs and then poking the twigs into termite mounds. The termites react to the “invasion” by attacking the twig. The chimpanzee pulls the twig out, termites attached, and eats them. In Gambia, they use modified twigs to extract honey from holes in trees. In Fongoli, Sénégal, chimpanzees use sticks as “spears” that they stab into tree cavities to hunt for galagos (Figure 7.27). Multiple chimpanzee populations use a “hammer and anvil” to crack open nuts, but the specific techniques differ. Because the cultural traditions are so diverse and unique, if a researcher can observe enough of a chimpanzee’s behavior, it is possible to assign that individual to a specific community, much in the same way a human being can be associated with a specific culture based on his or her behavior (Whiten 2011).

How do chimpanzee cultures develop, and how does cultural transmission occur? Although we do not know for sure how chimpanzee cultural traditions develop initially, it is possible that different groups invent, either accidentally or deliberately, certain behaviors that other individuals copy. Immigration, or movement of an individual into a new group or community, is an important avenue of cultural transmission in chimpanzees, much as it is between human cultures. Immigrants (typically females) may bring cultural traditions to their new community, which residents observe and learn. Conversely, immigrants may observe and learn a cultural tradition practiced in their new community (Whiten 2011).
Cultural Transmission in Macaques
Two monkey species are well-known for behavioral variation that has been called “pre-cultural” by some primatologists: Japanese macaques and tufted capuchins (Sapajus apella). The transmission of unique foraging (the act of searching for food) behaviors through the members of a provisioned group of Japanese macaques on Koshima Island is well known (Matsuzawa 2015). In an effort to keep the monkeys nearby, researchers provided them with piles of sweet potatoes. A juvenile female named Imo spontaneously washed a muddy sweet potato in a stream. This new food-processing technique first spread among other juveniles and then gradually to older individuals. Within 30 years, it had spread across generations, and 46 of 57 monkeys in the group engaged in the behavior. Another example comes from a group living far to the north, in Shiga-Heights, Nagano Prefecture. Researchers used apples to entice Japanese macaques to the area. Within a few years, monkeys visited the area regularly and were observed playing with the water in the hot springs. Soon, they climbed into the hot springs and learned to immerse themselves to keep warm and reduce stress when not foraging (Figure 7.28; Matsuzawa 2018; Takeshita et al. 2018; recall also our discussion of hot spring use as an example of analogous traits at the beginning of this chapter). These examples share several characteristics with human culture, including invention or modification of behavior, transmission of behavior between individuals, and the persistence of the behavior across generations (McGrew 1998).

Summary
Primates are socially complex, extremely intelligent, and highly adaptable. In this chapter we discussed aspects of primate ecology, including how body size and characteristics of food affect what primates eat and how primates interact with other species in their environment. We examined why primates live in groups, the types of groups in which they are found, and the reproductive strategies used by males and females to maximize reproductive success. Like other aspects of their behavior, primate communication is varied and complex, and we discussed how primates communicate using vocal, visual, olfactory, and tactile signals. Finally, we explored the question of culture among nonhuman primates and learned that some species have cultural traditions, distinctive patterns of behavior shared by multiple individuals in a social group that persist over time. Humans and other primates are similar in many ways. Learning about principles of primate ecology and behavior can help us better understand our own behavior and the behaviors of our extinct relatives.
Review Questions
- If anthropology is the study of humans, why do some anthropologists study primates?
- How does a primate’s ecology affect their diet and interactions with other organisms?
- Why do primates live in groups and in what types of groups do they live?
- What is parental investment and sexual selection?
- What are some examples of primate communication?
- What is the evidence for cultural traditions in primates and how do primatologists think cultural transmission occurs in primates?
Key Terms
Abundance: How much food is available in a given area.
Adaptation: A trait with a function.
Affiliative: Nonaggressive social interactions and associations between individuals.
Agonistic: Conflict; aggressive interactions between individuals.
Alarm calling: Vocalizations emitted by social animals in response to danger.
Analogy: A similar trait found in different species that arose independently.
Anogenital: Relating to the anus and genitals.
Breeding season: The time of year when females are receptive to mating.
Callitrichids: The primate family that includes marmosets and tamarins.
Carnivores: Organisms whose diet consists primarily of animal tissue.
Coalition: A temporary alliance between individuals.
Community ecology: The branch of ecology that deals with the relationships and interactions between different organisms that occupy the same habitat.
Comparison: An examination of the similarities and differences between two things, such as two primate species.
Conspecifics: Members of the same species.
Cooperative breeding: When individuals other than the mother and father help raise the offspring.
Crypsis: The ability to avoid detection by other organisms, such as predators.
Cultural tradition: A distinctive pattern of behavior shared by multiple individuals in a social group, which persists over time and is acquired through social learning.
Culture: The transmission of behavior from one generation to the next through observation and imitation.
Decolonize: Understanding and highlighting the theory and research of non-Western individuals and perspectives.
Descendant: A species that comes after the ancestor species.
Direct competition: Competition that involves physical interaction between individuals, such as fighting.
Dispersal: To leave one’s group or area. This may or may not involve joining another group.
Distribution: How food is spread out.
Diurnal: Active during the day.
Dominance hierarchy: The ranked organization of individuals established by the outcome of aggressive-submissive interactions.
Dominant: Being of high rank.
Ecology: The relationship between organisms and their physical surroundings.
Ecotourism: A form of tourism that focuses on nature-based attractions to provide learning opportunities and that uses economically and ecologically sustainable practices.
Ethology: The study of animal behavior.
Fission-fusion: Societies in which group composition is flexible, such as chimpanzee and spider monkey societies. Individuals may break up into smaller feeding groups (fission) and combine into larger groups (fusion).
Fitness: An individual’s ability to survive and reproduce relative to other members of the same species.
Folivores: Organisms whose diet consists primarily of leaves.
Foraging: The act of searching for food.
Frugivores: Organisms whose diet consists primarily of fruit.
Grooming: Picking through the fur of another individual for cleaning or bonding purposes.
Heterospecifics: Members of different species.
Holism: The idea that the parts of a system interconnect and interact to make up the whole.
Home range: The area that a group or individual uses over a given period of time (often over a year).
Homology: A similar trait found in different species because it was inherited from a common ancestor.
Immigration: Movement of an individual into a new group or community.
Indirect competition: Competition that does not involve physical interaction between individuals, such as eating food before another individual arrives at the food site.
Infanticide: The killing of infants of one’s own species.
Innate: Natural; as in behavior that comes naturally.
Insectivores: Organisms whose diets consist primarily of insects.
Interbirth interval: The typical length of time between one birth and the next for a species.
Intersexual selection: The selection for traits that enhance the ability of the members of one sex to attract the attention of the other.
Intrasexual selection: Selection for traits that enhance the ability of members of one sex to compete amongst themselves.
Mating system: A way of describing which male(s) and female(s) mate.
Metabolism: The chemical changes that take place in an organism that turn nutrients into energy.
Mobbing: Cooperatively attacking or harassing a predator.
Monogamy: A mating system in which one male mates with one female.
Multi-male, multi-female: A group that consists of multiple adult males, multiple adult females, and their dependent offspring.
Multi-male, single-female: A group that consists of two or more adult males, one breeding female, their dependent offspring, and non-breeding females.
Mutualistic/mutualism: When different species work together, with each benefiting from the interaction.
Niche: The role of a species in its environment; how it meets its needs for food, shelter, etc.
Nocturnal: Active at night.
Olfactory communication: Conveying information through scent.
Omnivores: Organisms whose diet consists of plant and animal matter.
Pair bond: A strong, long-term relationship between two individuals.
Parasite: An organism that lives in or on another organism.
Parental investment: Any time or energy a parent devotes to the current offspring that enhances its survival (and eventual reproductive success) at the expense of the parent’s ability to invest in the next offspring.
Philopatric: Remaining in the group of one’s birth.
Piloerection: Raising one’s hair or fur in an effort to look bigger.
Polyandry: A mating system in which multiple males mate with a single breeding female.
Polygamy: A mating system in which multiple males mate with multiple females.
Polygyny: A mating system in which one male mates with multiple females.
Polyspecific association: An association between two or more different species that involves behavioral changes in at least one of them to maintain the association.
Primate community: All primate species that occur in an area.
Primatologist: A scientist who studies primate behavior and/or ecology.
Primatology: The scientific field that studies primate behavior and/or ecology.
Ranging behavior: Refers to the way in which animals move about their environment.
Receptive: A term used to describe females who are ready for sexual reproduction (i.e., not pregnant or nursing).
Reproductive success: An individual’s genetic contribution to future generations, often measured through the number of offspring produced.
Reproductive suppression: The prevention or inhibition of reproduction of healthy adults.
Resident male: Term that describes the male who lives with a group of females.
Seed dispersal: The process by which seeds move away from the plant that produced them in preparation for germination and becoming a new plant.
Semantic communication: The systematic use of signals to refer to objects in the environment.
Sexual dimorphism: When males and females of a species have different morphological traits.
Sexual selection: The selection for traits that increase mating success. This occurs via intersexual selection and intrasexual selection.
Sexual swelling: Area of the hindquarters that change in size, shape, and often color over the course of a female’s reproductive cycle, reaching maximum size at ovulation. Occurs in many primate species that live in Africa and Asia.
Sexually monomorphic: When males and females of a species have similar morphological traits.
Single-male, multi-female: A group that consists of one adult male, multiple adult female, and their dependent offspring.
Single-male, single-female: A group that consists of one adult male, one adult female, and their dependent offspring.
Social learning: The idea that new behaviors can be acquired by observing and imitating others.
Social system: A way of describing the typical number of males and females of all age classes that live together.
Social transmission: Transfer of something from one individual to another; this can include parasites, information, or cultural traditions.
Sociality: The tendency to form social groups.
Solitary: Living alone.
Species recognition: The ability to differentiate conspecifics from members of other species.
Subordinate: Being of low rank.
Tactile communication: Conveying information through touch.
Territory: A home range whose boundary is defended from intrusion by conspecifics.
Vertebrates: The group of animals characterized by an internal spinal column or backbone. This includes fish, amphibians, reptiles, birds, and mammals.
Vigilance: Watchful behavior used to detect potential danger, usually in the form of predators or potential competitors.
Visual communication: Conveying information through signals that can be seen.
Vocal communication: Conveying information through signals that can be heard.
For Further Exploration
Goodall, Jane. 1971. In the Shadow of Man. Boston: Houghton Mifflin.
Rowe, Noel, and Marc Myers, eds. 2016. All the World’s Primates. Charleston, RI: Pogonias Press.
Strier, Karen B. 2017. Primate Behavioral Ecology. 5th ed. New York: Routledge.
Primate Info Net is an information service of the National Primate Research Center at the University of Wisconsin, Madison. It includes Primate Factsheets, primate news and publications, a list of primate-related jobs, and an international directory of primatology, among other information.
Primate Specialist Group is a collection of scientists and conservationists who work in dozens of African, Asian, and Latin American nations to promote research on primate conservation.
Short videos of some primate behaviors discussed in this chapter:
- Watch vervet monkeys respond to different types of predators: BBC One. n.d. “Vervet Monkey’s Escape Plans - Talk to the Animals: Episode 2 Preview.” Accessed December 16, 2022. https://www.youtube.com/watch?v=q8ZG8Dpc8mM.
- Watch male gelada baboons use the lip flip in competition with other males: Smithsonian Channel, June 9, 2017. “Why These Vegetarian Monkeys Have Sharp Predator Teeth.” Accessed July 25, 2019. https://www.youtube.com/watch?time_continue=145&v=aC6iYj_EBjY.
- Watch (and listen to!) howler monkeys “roar”: Science News. N.d. “Hear a Male Howler Monkey Roar.” Accessed November 21, 2022. https://www.youtube.com/watch?v=PYar0dkZ6v8.
- Watch Japanese macaques using natural hot springs: National Geographic. N.d. “Meditative Snow Monkeys Hang Out in Hot Springs.” Accessed July 25, 2019. https://www.youtube.com/watch?v=Aat9O85ynsI.
- Watch chimpanzees make and use tools: National Geographic. n.d. “Chimps and Tools.” Accessed July 25, 2019. https://www.youtube.com/watch?v=o2TBicMRLtA.
References
Aich, H., R. Moos-Heilen, and E. Zimmermann. 1990. “Vocalizations of Adult Gelada Baboons (Theropithecus gelada): Acoustic Structure and Behavioural Context.” Folia Primatologica 55 (3–4): 109–132.
Bell, Sarah A. 2017. “Galdikas, Birute.” In The International Encyclopedia of Primatology, Volume A–G, edited by Agustín Fuentes, 445–446. Malden, MA: John Wiley & Sons.
Boinski, S. 1992. “Olfactory Communication among Costa Rican Squirrel Monkeys: A Field Study.” Folia Primatologica 59 (3): 127–136.
Cheney, D. L., and R. M. Seyfarth. 1987. “The Influence of Intergroup Competition on the Survival and Reproduction of Female Vervet Monkeys.” Behavioral Ecology and Sociobiology 21 (6): 375–386.
de Oliveira Terceiro, Francisco Edvaldo, and Judith M. Burkart. 2019. “Cooperative Breeding.” In Encyclopedia of Animal Cognition and Behavior, edited by Jennifer Vonk and Todd Shackelford, 1–6. Edinburg, Scotland: Springer Cham.
Digby, Leslie J., Stephen F. Ferrari, and Wendy Saltzman. 2011. “Callitrichines: The Role of Competition in Cooperatively Breeding Species.” In Primates in Perspective, edited by Christina J. Campbell, AugustÍn Fuentes, Katherine C. MacKinnon, Simon K. Bearder, and Rebecca M. Stumpf, 91–10. 2nd edition. New York: Oxford University Press.
Fischer, Julia, Kurt Hammerschmidt, Dorothy L. Cheney, and Robert M. Seyfarth. 2008. “Acoustic Features of Female Chacma Baboon Barks.” Ethology 107 (1): 33–54.
Jolly, Alison. 1966. Lemur Behavior: A Madagascar Field Study. Chicago: University of Chicago Press.
Krief, Sabrina, Claude Marcel Hladik, and Claudie Haxaire. 2005. “Ethnomedicinal and Bioactive Properties of Plants Ingested by Wild Chimpanzees in Uganda.” Journal of Ethnopharmacology 110 (1–3): 1–15.
Maekawa, Mkio, Annette Lanjouw, Eugène Rutagarama, and Doublas Sharp. 2013. “Mountain Gorilla Tourism Generating Wealth and Peace in Post-Conflict Rwanda.” Natural Resources Forum 37 (2): 127–137.
Matsuzawa, Tetsuro. 2015. “Sweet-Potato Washing Revisited: 50th Anniversary of the Primates Article.” Primates 56: 285–287.
Matsuzawa, Tetsuro. 2018. “Hot-Spring Bathing of Wild Monkeys in Shiga-Heights: Origin and Propagation of a Cultural Behavior.” Primates 59: 209–213.
McGrew, W. C. 1998. “Culture in Nonhuman Primates?” Annual Review of Anthropology 27: 301–328.
Mertl-Millhollen, Anne S. 1988. “Olfactory Demarcation of Territorial but Not Home Range Boundaries by Lemur catta.” Folia Primatologica 50 (3–4): 175–187.
Pinacho-Guendulain, B., and G. Ramos-Fernández. 2017. “Influence of Fruit Availability on the Fission-Fusion Dynamics of Spider Monkeys (Ateles geoffroyi).” International Journal of Primatology 38: 466–484.
Poirotte, Clémence, François Massol, Anaïs Herbert, Eric Willaume, Pacelle M. Bomo, Peter M. Kappeler, and Marie J. E. Charpentier. 2017. “Mandrills Use Olfaction to Socially Avoid Parasitized Conspicifics.” Science Advances 3 (4): e160172.
Rodrigues, Michelle. 2019. “It’s Time to Stop Lionizing Dian Fossey as a Conservation Hero.” Lady Science website, September 20. Accessed December 14, 2022. https://www.ladyscience.com/ideas/time-to-stop-lionizing-dian-fossey-conservation.
Samuni, Liran, Anna Preis, Tobias Deschner, Catherine Crockford, and Roman M. Wittig. 2018. “Reward of Labor Coordination and Hunting Success in Wild Chimpanzees.” Communications Biology 1: 138.
Santana, Sharlene E., Jessica Lynch Alfaro, and Michael E. Alfaro. 2012. “Adaptive Evolution of Facial Colour Patterns in Neotropical Primates.” Proceedings of the Royal Society B: Biological Sciences 279 (1736): 2204–2211.
Sanz, Crickette M., David Strait, Crepin Eyana Ayina, Jean Marie Massamba, Thierry Fabrice Ebombi, Severin Ndassoba Kialiema, Delon Ngoteni, et al. 2022. “Interspecific Interactions Between Sympatric Apes.” iScience 25 (10): 105059.
Schön Ybarra, M. A. 1986. “Loud Calls of Adult Male Red Howling Monkeys (Alouatta seniculus).” Folia Primatologica 47 (4): 204–216.
Setchell, Joanna M., Tessa Smith, E. Jean Wickings, and Leslie A. Knapp. 2008. “Social Correlates of Testosterone and Ornamentation in Male Mandrills.” Hormones and Behavior 54 (3): 365–372.
Setchell, Joanna M., E. Jean Wickings, and Leslie A. Knapp. 2006. “Signal Content of Red Facial Coloration in Female Mandrills (Mandrillus sphinx).” Proceedings of the Royal Society B: Biological Sciences 273 (1599): 2395–2400.
Seyfarth, R. M., D. L. Cheney, and P. Marler. 1980a. “Monkey Responses to Three Different Alarm Calls: Evidence of Predator Classification and Semantic Communication.” Science 210 (4471): 801–803.
Seyfarth, Robert M., Dorothy L. Cheney, and Peter Marler. 1980b. “Vervet Monkey Alarm Calls: Semantic Communication in a Free-Ranging Primate.” Animal Behaviour 28 (4): 1070–1094.
Sharma, Goutam, Chan Ram, and Lal Singh Rajpurohit. 2010. “A Case Study of Infantcide After Resident Male Replacement in Semnopithecus entellus around Jodhpur (India).” Proceeding of the Zoological Society 63 (2): 93–98.
Shirasu, Mika, Satomi Ito, Akihiro Itoigawa, Takashi Hayakawa, Kodzue Kinoshita, Isao Munechika, Hiroo Imai, and Kazushige Touhara. 2020. “Key Male Glandular Odorants Attracting Female Ring-Tailed Lemurs.” Current Biology 30 (11): 2131–2138.
Stanford, Craig B. 2017. “Goodall, Jane.” In The International Encyclopedia of Primatology, Volume A–G, edited by Agustín Fuentes, 471–472. Malden, MA: John Wiley & Sons.
Stewart, Kelly. 2017. “Fossey, Dian.” In The International Encyclopedia of Primatology, Volume A–G, edited by Agustín Fuentes, 432–433. Malden, MA: John Wiley & Sons.
Takeshita, Rafaela S.C., Fred B. Bercovitch, Kodzue Kinoshita, and Michael A. Huffman. 2018. “Beneficial Effect of Hot Spring Bathing on Stress Levels in Japanese Macaques.” Primates 59 (3): 215–225.
Trivers, Robert L. 1972. “Parental Investment and Sexual Selection.” In Sexual Selection and the Descent of Man, 1871–1971, edited by Bernard Campbell, 136–179. Chicago: Aldine.
Whiten, Andrew. 2011. “The Scope of Culture in Chimpanzees, Humans and Ancestral Apes.” Philosophical Transactions of the Royal Society of London B: Biological Sciences 366 (1567): 997–1007.
Wiens, Frank, and Annette Zitzmann. 2003. “Social Structure of the Solitary Slow Loris Nycticebus coucang (Lorisidae).” Journal of Zoology 261 (1): 35–46.
Zuberbühler, Klaus, David Jenny, and Redouan Bshary. 1999. “The Predator Deterrence Function of Primate Alarm Calls.” Ethology 105 (6): 477–490.
Zuberbühler, Klaus, Ronald Noë, and Robert M. Seyfarth. 1997. “Diana Monkey Long-Distance Calls: Messages for Conspecifics and Predators.” Animal Behaviour 53 (3): 589–604.
Acknowledgments
The author is grateful to the editors for the opportunity to contribute to this open-source textbook. She thanks Dr. Stephanie Etting for her encouragement and support during the revision of this chapter. Her suggestions, along with comments made by two anonymous reviewers on an earlier draft of this chapter, improved the final version considerably. Finally, she thanks all the primatologists who came before her, especially her advisor, Lynne A. Isbell, for their tireless efforts to understand the behavior and ecology of the living primates. Without their work, this chapter would not have been possible.
Jonathan M. G. Perry, Ph.D., Western University of Health Sciences
Stephanie L. Canington, Ph.D., University of Pennsylvania
This chapter is a revision from "Chapter 8: Primate Evolution” by Jonathan M. G. Perry and Stephanie L. Canington. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Understand the major trends in primate evolution from the origin of primates to the origin of our own species.
- Learn about primate adaptations and how they characterize major primate groups.
- Discuss the kinds of evidence that anthropologists use to find out how extinct primates are related to each other and to living primates.
- Recognize how the changing geography and climate of Earth have influenced where and when primates have thrived or gone extinct.
The first fifty million years of primate evolution was a series of adaptive radiations leading to the diversification of the earliest lemurs, monkeys, and apes. The primate story begins in the canopy and understory of conifer-dominated forests, with our small, furtive ancestors subsisting at night, beneath the notice of day-active dinosaurs.
From the ancient plesiadapiforms (archaic primates) to the earliest groups of true primates (euprimates) (Bloch and Boyer 2002), the origin of our own order is characterized by the struggle for new food sources and microhabitats in the arboreal setting. Climate change forced major extinctions as the northern continents became increasingly dry, cold, and seasonal and as tropical rainforests gave way to deciduous forests, woodlands, and eventually grasslands. Lemurs, lorises, and tarsiers—once diverse groups containing many species—became rare, except for lemurs in Madagascar, where there were no anthropoid competitors and perhaps few predators. Meanwhile, anthropoids (monkeys and apes) likely emerged in Asia and then dispersed across parts of the northern hemisphere, Africa, and ultimately South America. The movement of continents, shifting sea levels, and changing patterns of rainfall and vegetation contributed to the developing landscape of primate biogeography, morphology, and behavior. Today’s primates provide modest reminders of the past diversity and remarkable adaptations of their extinct relatives. This chapter explores the major trends in primate evolution from the origin of the Order Primates to the beginnings of our own lineage, providing a window into these stories from our ancient past.
Major Hypotheses About Primate Origins
For many groups of mammals, there is a key feature that led to their success. A good example is powered flight in bats. Primates lack a feature like this (see Chapter 5). Instead, if there is something unique about primates, it is probably a group of features rather than one single thing. Because of this, anthropologists and paleontologists struggle to describe an ecological scenario that could explain the rise and success of our own order. Three major hypotheses have been advanced to consider the origin of primates and to explain what makes our order distinct among mammals (Figure 9.1); these are described below.
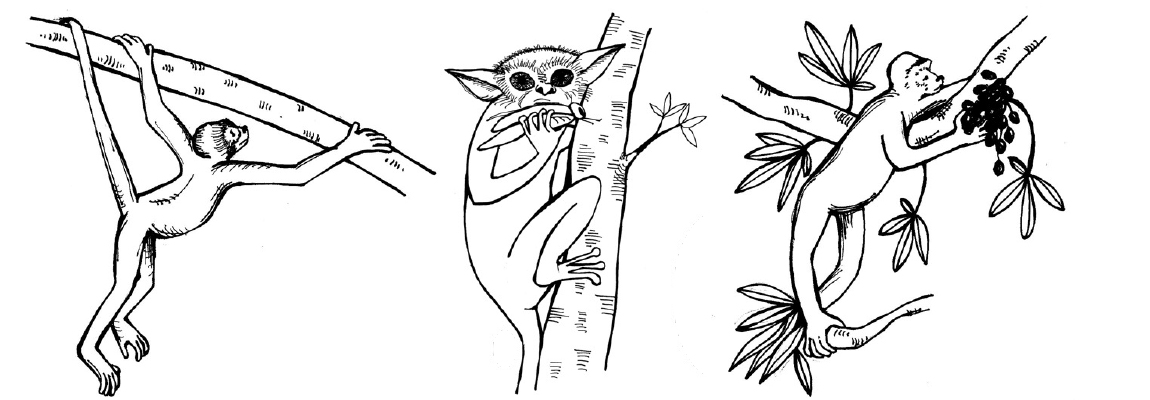
Arboreal Hypothesis
In the 1800s, many anthropologists viewed all animals in relation to humans. That is, animals that were more like humans were considered to be more “advanced” and those lacking humanlike features were considered more “primitive.” This way of thinking was particularly obvious in studies of primates. A more modern way of referring to members of a group that lack certain evolutionary innovations seen in other members is to call them plesiomorphic (literally “anciently shaped”). The state of their morphological features is sometimes referred to as ancestral traits.
Thus, when anthropologists sought features that separate primates from other mammals, they focused on features that were least developed in lemurs and lorises, more developed in monkeys, and most developed in apes (Figure 9.2). Frederic Wood Jones, one of the leading anatomist-anthropologists of the early 1900s, is usually credited with the Arboreal Hypothesis of primate origins (Jones 1916). This hypothesis holds that many of the features of primates evolved to improve locomotion in the trees; this way of getting around is referred to as arboreal. For example, the grasping hands and feet of primates are well suited to gripping tree branches of various sizes and our flexible joints are good for reorienting the extremities in many different ways. A mentor of Jones, Grafton Elliot Smith, had suggested that the reduced olfactory system, acute vision, and forward-facing eyes of primates are adaptations for making accurate leaps and bounds through a complex, three-dimensional canopy (Smith 1912). The forward orientation of the eyes in primates causes the visual fields to overlap, enhancing depth perception, especially at close range. Evidence to support this hypothesis includes the facts that many extant primates are arboreal, and the plesiomorphic members of most primate groups are dedicated arborealists. The Arboreal Hypothesis was well accepted by most anthropologists at the time and for decades afterward.
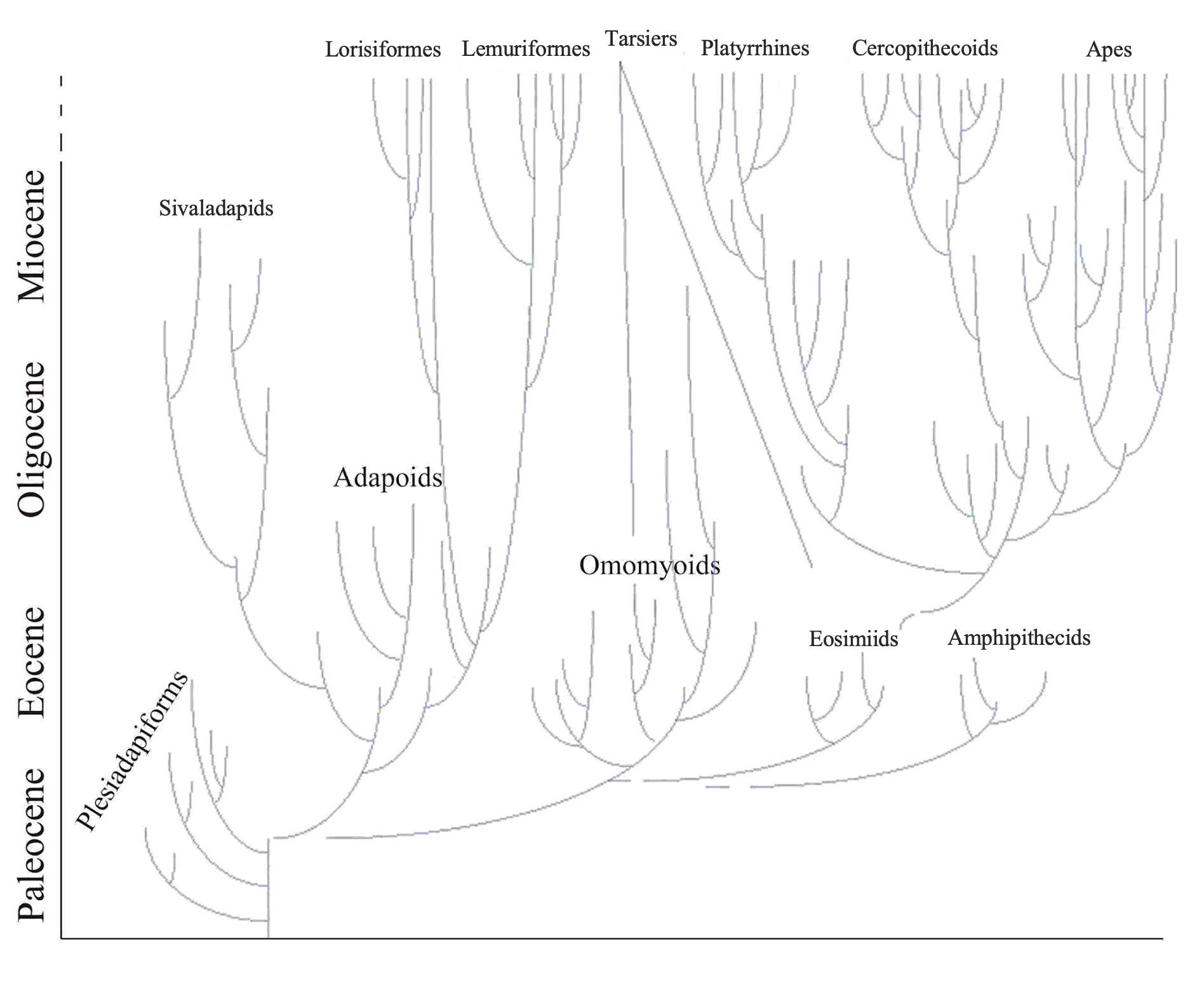
Visual Predation Hypothesis
In the late 1960s and early 1970s, Matt Cartmill studied and tested the idea that the characteristic features of primates evolved in the context of arboreal locomotion. Cartmill noted that squirrels climb trees (and even vertical walls) very effectively, even though they lack some of the key adaptations of primates. As members of the Order Rodentia, squirrels also lack the hand and foot anatomy of primates. They have claws instead of flattened nails and their eyes face more laterally than those of primates. Cartmill reasoned that there must be some other explanation for the unique traits of primates. He noted that some nonarboreal animals share at least some of these traits with primates; for example, cats and predatory birds have forward-facing eyes that enable visual field overlap. Cartmill suggested that the unique suite of features in primates is an adaptation to detecting insect prey and guiding the hands (or feet) to catch insects (Cartmill 1972). His hypothesis emphasizes the primary role of vision in prey detection and capture; it is explicitly comparative, relying on form-function relationships in other mammals and nonmammalian vertebrates. According to Cartmill, many of the key features of primates evolved for preying on insects in this special manner (Cartmill 1974).
Angiosperm-Primate Coevolution Hypothesis
The visual predation hypothesis was unpopular with some anthropologists. One reason for this is that many primates today are not especially predatory. Another is that, whereas primates do seem well adapted to moving around in the smallest, terminal branches of trees, insects are not necessarily easier to find there. A counterargument to the visual predation hypothesis is the angiosperm-primate coevolution hypothesis. Primate ecologist Robert Sussman (Sussman 1991) argued that the few primates that eat mostly insects often catch their prey on the ground rather than in tree branches. Furthermore, predatory primates often use their ears more than their eyes to detect prey. Finally, most early primate fossils show signs of having been omnivorous rather than insectivorous. Instead, he argued, the earliest primates were probably seeking fruit. Fruit (and flowers) of angiosperms (flowering plants) often develop in the terminal branches. Therefore, any mammal trying to access those fruits must possess anatomical traits that allow them to maintain their hold on thin branches and avoid falling while reaching for the fruits. Primates likely evolved their distinctive visual traits and extremities in the Paleocene (approximately 65 million to 54 million years ago) and Eocene (approximately 54 million to 34 million years ago) epochs, just when angiosperms were going through a revolution of their own—the evolution of large, fleshy fruit that would have been attractive to a small arboreal mammal. Sussman argued that, just as primates were evolving anatomical traits that made them more efficient fruit foragers, angiosperms were also evolving fruit that would be more attractive to primates to promote better seed dispersal. This mutually beneficial relationship between the angiosperms and the primates was termed coevolution or more specifically diffuse coevolution.
At about the same time, D. Tab Rasmussen noted several parallel traits in primates and the South American woolly opossum, Caluromys. He argued that early primates were probably foraging on both fruits and insects (Rasmussen 1990). As is true of Caluromys today, early primates probably foraged for fruits in the terminal branches of angiosperms, and they probably used their visual sense to aid in catching insects. Insects are also attracted to fruit (and flowers), so these insects represent a convenient opportunity for a primarily fruit-eating primate to gather protein. This solution is a compromise between the visual predation hypothesis and the angiosperm-primate coevolution hypothesis. It is worth noting that other models of primate origins have been proposed, and these include the possibility that no single ecological scenario can account for the origin of primates.
The Origins of Primates
Paleocene: Mammals in the Wake of Dinosaur Extinctions
Placental mammals, including primates, originated in the Mesozoic Era (approximately 251 million to 65.5 million years ago), the Age of Dinosaurs. During this time, most placental mammals were small, probably nocturnal, and probably avoided predators via camouflage and slow, quiet movement. It has been suggested that the success and diversity of the dinosaurs constituted a kind of ecological barrier to Mesozoic mammals. The extinction of the dinosaurs (and many other organisms) at the end of the Cretaceous Period (approximately 145.5–65.5 million years ago) might have opened up these ecological niches, leading to the increased diversity and disparity in mammals of the Tertiary Period (approximately 65.5–2.5 million years ago).
The Paleocene was the first epoch in the Age of Mammals. Soon after the Cretaceous-Tertiary (K-T) extinction event, new groups of placental mammals appear in the fossil record. Many of these groups achieved a broad range of sizes and lifestyles as well as a great number of species before declining sometime in the Eocene (or soon thereafter). These groups were ultimately replaced by the modern orders of placental mammals (Figure 9.3). It is unknown whether these replacements occurred gradually, for example by competitive exclusion, or rapidly, perhaps by sudden geographic dispersals with replacement. In some senses, the Paleocene might have been a time of recovery from the extinction event; it was cooler and more seasonal globally than the subsequent Eocene.

Plesiadapiforms, the Archaic Primates
The Paleocene epoch saw the emergence of several families of mammals that have been implicated in the origin of primates. These are the plesiadapiforms, which are archaic primates, meaning they possessed some primate features and lacked others. The word plesiadapiform means “almost adapiform,” a reference to some similarities between some plesiadapiforms and some adapiforms (or adapoids; later-appearing true primates)—mainly in the molar teeth. Because enamel fossilizes better than other parts of the body, the molar teeth are the parts most often found and first discovered for any new species. Thus, dental similarities were often the first to be noticed by early mammalian paleontologists, partly explaining why plesiadapiforms were thought to be primates. Major morphological differences between plesidapiforms and euprimates (true primates) were observed later when more parts of plesiadapiform skeletons were discovered. Many plesiadapiforms have unusual anterior teeth and most have digits possessing claws rather than nails. So far, no plesiadapiform ever discovered has a postorbital bar (seen in extant strepsirrhines) or septum (as seen in haplorhines), and whether or not the auditory bulla was formed by the petrosal bone remains unclear for many plesiadapiform specimens. Nevertheless, there are compelling reasons (partly from new skeletal material) for including plesiadapiforms within the Order Primates.
Geographic and Temporal Distribution
Purgatorius is generally considered to be the earliest primate. This Paleocene mammal is known from teeth that are very plesiomorphic for a primate. It has some characteristics that suggest it is a basal plesiadapiform, but there is very little to link it specifically with euprimates (see Clemens 2004). Its ankle bones suggest a high degree of mobility, signaling an arboreal lifestyle (Chester et al. 2015). Purgatorius is plesiomorphic enough to have given rise to all primates, including the plesiadapiforms. However, new finds suggest that this genus was more diverse and had more differing tooth morphologies than previously appreciated (Wilson Mantilla et al. 2021). Plesiadapiform families were numerous and diverse during parts of the Paleocene in western North America and western Europe, with some genera (e.g., Plesiadapis; see Figure 9.4) living on both continents (Figure 9.5). Thus, there were probably corridors for plesiadapiform dispersal between the two continents, and it stands to reason that these mammals were living all across North America, including in the eastern half of the continent and at high latitudes. A few plesiadapiforms have been described from Asia (e.g., Carpocristes), but the affinities of these remain uncertain.
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Paromomyidae |
Ignacius |
Long, dagger-like, lower incisor. |
North America and Europe |
Early Paleocene to Late Eocene |
|
Carpolestidae |
Carpolestes |
Plagiaulacoid dentition. Limb adaptations to terminal branch feeding. Grasping big toe. |
North America, Europe, and Asia |
Middle Paleocene to Early Eocene |
|
Plesiadapidae |
Plesiadapis |
Mitten-like upper incisor. Diastema. Large body size for group. |
North America and Europe |
Middle Paleocene to Early Eocene |
|
1 Derived from Fleagle 2013. |
|
|||
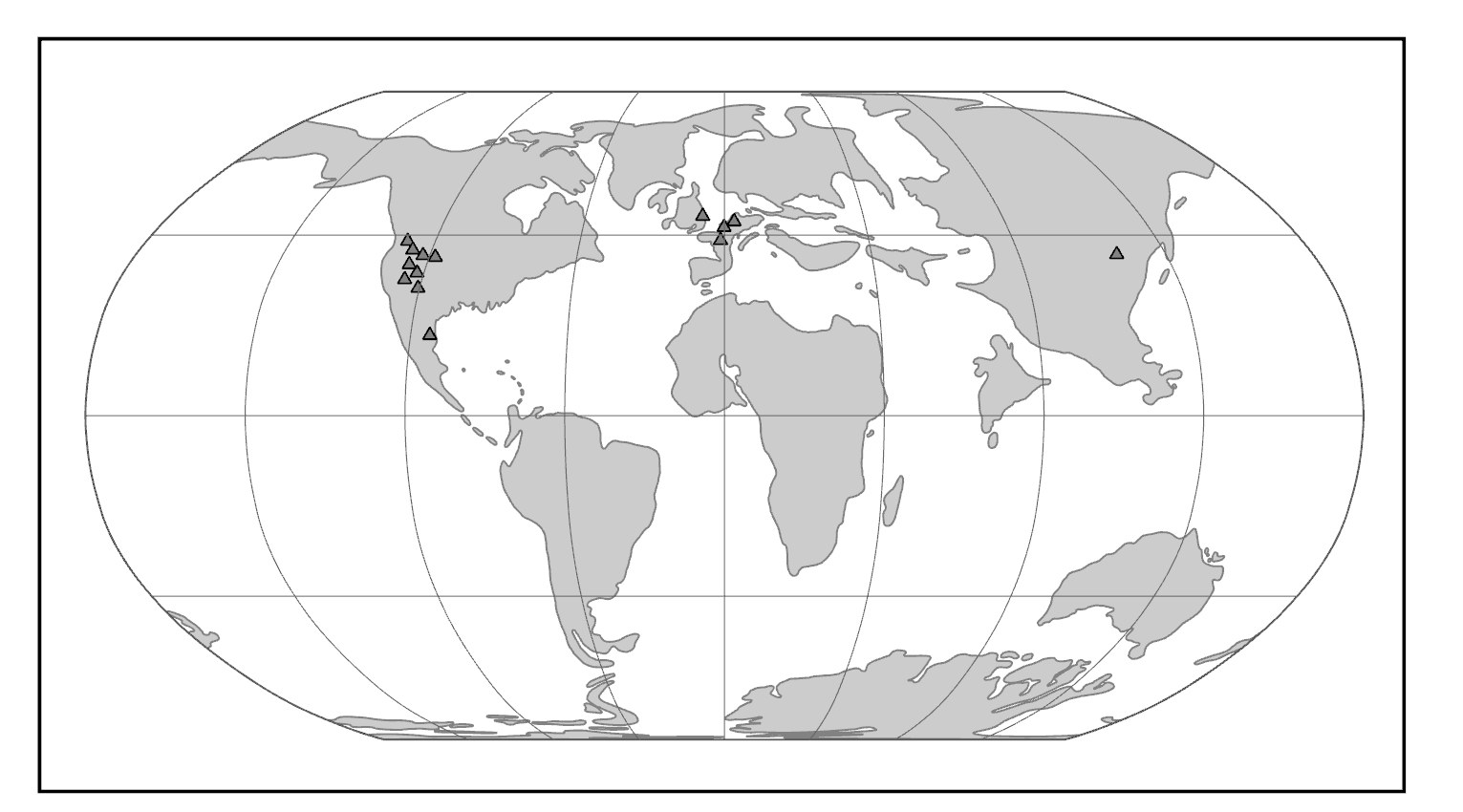
General Morphological Features
Although there is much morphological variation among the families of plesiadapiforms, some common features unite the group. Most plesiadapiforms were small, the largest being about three kilograms (approximately 7 lbs.; Plesiadapis cookei). They had small brains and fairly large snouts, with eyes that faced more laterally than in euprimates. Many species show reduction and/or loss of the canine and anterior premolars, with the resulting formation of a rodent-like diastema (a pronounced gap between the premolars and the incisors, with loss of at least the canine); this probably implies a herbivorous diet. Some families appear to have had very specialized diets, as suggested by unusual tooth and jaw shapes.
Arguably the most interesting and unusual family of plesiadapiforms is the Carpolestidae. They are almost exclusively from North America (with a couple of possible members from Asia), and mainly from the Middle and Late Paleocene. Their molars are not very remarkable, being quite similar to those of some other plesiadapiforms (e.g., Plesiadapidae). However, their lower posterior premolars (p4) are laterally compressed and blade-like with vertical serrations topped by tiny cuspules. This unusual dental morphology is termed plagiaulacoid (Simpson 1933). The upper premolar occlusal surfaces are broad and are covered with many small cuspules; the blade-like lower premolar might have cut across these cuspules, between them, or both.

Many plesiadapiforms have robust limb bones with hallmarks of arboreality. Instead of having nails, most taxa had sharp claws on most or all of the digits. The extremities show grasping abilities comparable to those of primates and some arboreal marsupials. Nearly complete skeletons have yielded a tremendous wealth of information on locomotor and foraging habits. Many plesiadapiforms appear to have been able to cling to vertical substrates (like a broad tree trunk) using their sharp claws, propelling themselves upward using powerful hindlimbs, bounding along horizontal supports, grasping smaller branches, and moving head-first down tree trunks. In carpolestids in particular, the skeleton appears to have been especially well adapted to moving slowly and carefully in small terminal branches (Figure 9.6).
Dig Deeper: Debate: Relationship of Plesiadapiforms to True Primates
In the middle of the twentieth century, treeshrews (Order Scandentia) were often considered part of the Order Primates, based on anatomical similarities between some treeshrews and primates. For many people, plesiadapiforms represented intermediates between primates and treeshrews, so plesiadapiforms were included in Primates as well.
Studies of reproduction and brain anatomy in treeshrews and lemurs suggested that treeshrews are not primates (e.g., Martin 1968). This was soon followed by the suggestion to also expel plesiadapiforms (Martin 1972) from the Order Primates. Like treeshrews, plesiadapiforms lack a postorbital bar, nails, and details of the ear region that characterize true primates. Many paleoanthropologists were reluctant to accept this move to banish plesiadapiforms (e.g., F. S. Szalay, P. D. Gingerich).
Later, K. Christopher Beard (1990) found that in some ways, the digits of paromomyid plesiadapiforms are actually more similar to those of dermopterans (Order Dermoptera), the closest living relatives of primates, than they are to those of primates themselves (but see Krause 1991). At the same time, Richard Kay and colleagues (1990) found that cranial circulation patterns and auditory bulla morphology in the paromomyid, Ignacius (see Figure 9.4), are more like those of dermopterans than of primates.
For many anthropologists, this one-two punch effectively removed plesiadapiforms from the Order Primates. In the last two decades, the tide of opinion has turned again, with many researchers reinstating plesiadapiforms as members of the Order Primates. New and more complete specimens demonstrate that the postcranial skeletons of plesiadapiforms, including the hands and feet, were primate-like, not dermorpteran-like (Bloch and Boyer 2002, 2007). New fine-grained CT scans of relatively complete plesiadapiform skulls revealed that they share some key traits with primates to the exclusion of other placental mammals (Bloch and Silcox 2006). Most significant was the suggestion that Carpolestes simpsoni possessed an auditory bulla formed by the petrosal bone, like in all living primates.
The debate about the status of plesiadapiforms continues, owing to a persistent lack of key bones in some species and owing to genuine complexity of the anatomical traits involved. Maybe plesiadapiforms were the ancestral stock from which all primates arose, with some plesiadapiforms (e.g., carpolestids) nearer to the primate stem than others.
Adapoids and Omomyoids, the First True Primates
Geographic and Temporal Distribution
The first universally accepted fossil primates are the adapoids (Superfamily Adapoidea) and the omomyoids (Superfamily Omomyoidea). These groups become quite distinct over evolutionary time, filling mutually exclusive niches for the most part. However, the earliest adapoids are very similar to the earliest omomyoids.
The adapoids were mainly diurnal and herbivorous, with some achieving larger sizes than any plesiadapiforms (10 kg; 22 lbs.). By contrast, the omomyoids were mainly nocturnal, insectivorous and frugivorous, and small.
Both groups appear suddenly at the start of the Eocene, where they are present in western North America, western Europe, and India (Figure 9.7). This wide dispersal of early primates was probably due to the presence of rainforest corridors extending far into northern latitudes.
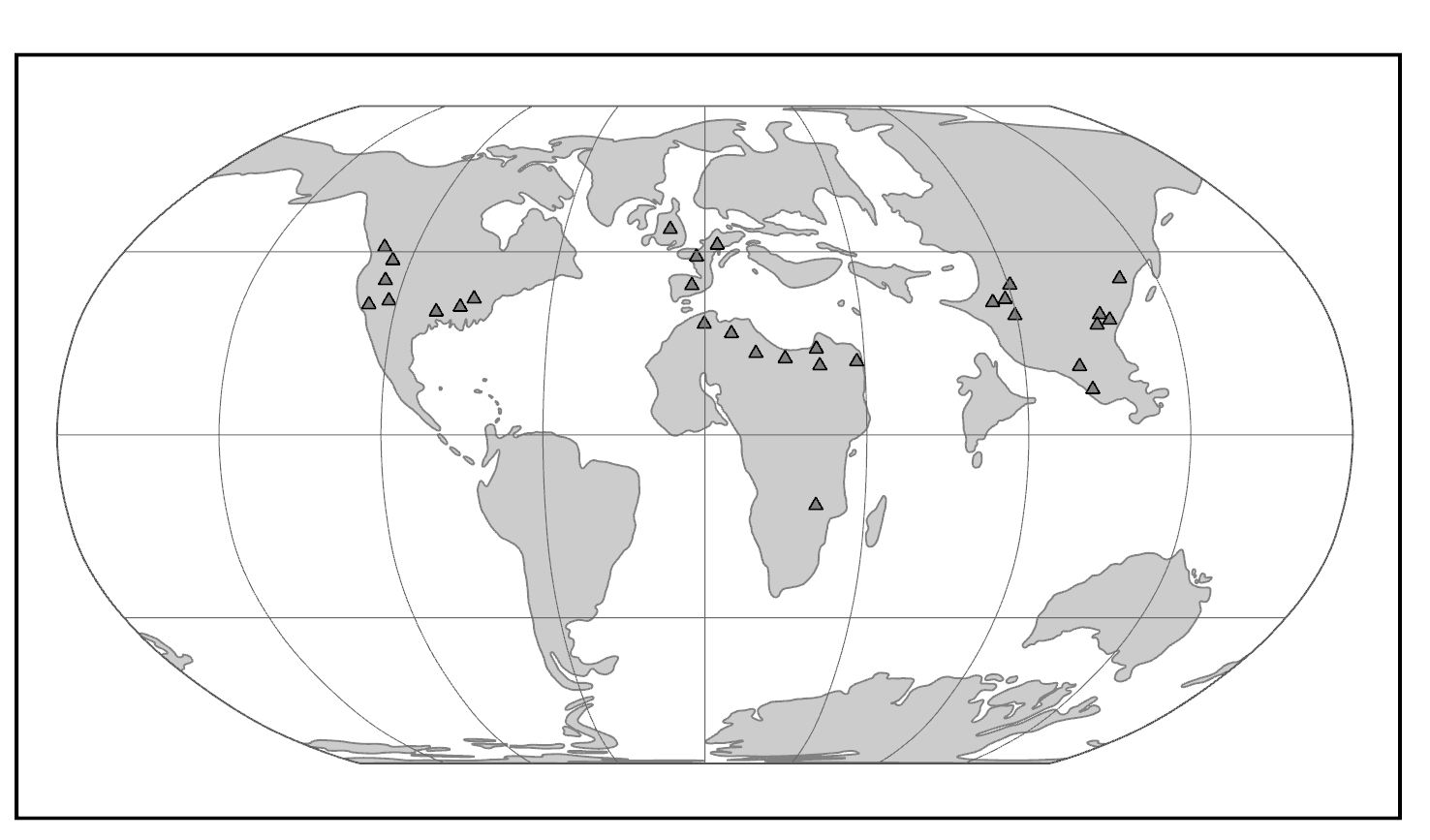
In North America and Europe, both groups achieved considerable diversity in the Middle Eocene, then mostly died out at the end of that epoch (Figure 9.8). In some Eocene rock formations in the western United States, adapoids and omomyoids make up a major part of the mammalian fauna. The Eocene of India has yielded a modest diversity of euprimates, some of which are so plesiomorphic that it is difficult to know whether they are adapoids or omomyoids (or even early anthropoids).
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Cercamoniidae |
Donrussellia |
Variable in tooth number and jaw shape. |
Europe and Asia |
Early to Late Eocene |
|
Asiadapidae2 |
Asiadapis |
Plesiomorphic teeth and jaw resemble early Omomyids. |
Asia |
Early Eocene |
|
Caenopithecidae3 |
Darwinius |
Robust jaws with crested molars. Fewer premolars. |
Europe, Africa, North America, and Asia |
Middle to Late Eocene |
|
Adapidae |
Adapis |
Fused mandible. Long molar crests. Large size and large chewing muscles. |
Europe |
Late Eocene to Early Oligocene |
|
Sivaladapidae |
Sivaladapis |
Some large with robust jaws. |
Asia |
Middle Eocene to Late Miocene |
|
Notharctidae4 |
Notharctus |
Canine sexual dimorphism. Lemur-like skull. Clinging and leaping adaptations. |
North America and Europe |
Early to Middle Eocene |
|
Omomyidae5 |
Teilhardina |
Small, nocturnal, frugivorous or insectivorous. Tarsier-like skull in some. |
North America, Europe, and Asia |
Early Eocene to Early Miocene |
|
Microchoeridae6 |
Necrolemur |
Long bony ear tubes. Tarsier-like lower limb adaptations for leaping. |
Europe and Asia |
Early Eocene to Early Oligocene |
|
1 Derived from Fleagle 2013. 2 See Dunn et al. 2016 and Rose et al. 2018. 3 See Kirk and Williams 2011 and Seiffert et al. 2009. 4 See Gregory 1920. 5 See Beard and MacPhee 1994 and Strait 2001. 6 See Schmid 1979. |
|
|||
Adapoids and omomyoids barely survived the Eocene-Oligocene extinctions, when colder temperatures, increased seasonality, and the retreat of rainforests to lower latitudes led to changes in mammalian biogeography. In North America, one genus (originally considered an omomyoid but recently reclassified as Adapoidea) persisted until the Miocene: Ekgmowechashala (Rose and Rensberger 1983). This taxon has highly unusual teeth and might have been a late immigrant to North America from Asia. In Asia, one family of adapoids, the Sivaladapidae, retained considerable diversity as late as the Late Miocene.
Adapoid Diversity
Adapoids were very diverse, particularly in the Eocene of North America and Europe. They can be divided into six families, with a few species of uncertain familial relationship. As a group, adapoids have some features in common, although much of what they share is plesiomorphic. Important features include the hallmarks of euprimates: postorbital bar, flattened nails, grasping extremities, and a petrosal bulla (Figures 9.9 and 9.10). In addition, some adapoids retain the ancestral dental formula of 2.1.4.3; that is, in each quadrant of the mouth, there are two incisors, one canine, four premolars, and three molars. In general, the incisors are small compared to the molars, but the canines are relatively large, with sexual dimorphism in some species. Cutting crests on the molars are well developed in some species, and the two halves of the mandible were fused at the midline in some species. Some adapoids were quite small (Anchomomys at a little over 100 g), and some were quite large (Magnadapis at 10 kg; 22 lbs.). Furthermore, the spaces and attachment features for the chewing muscles were truly enormous in some species, suggesting that these muscles were very large and powerful. Taken together, this suggests an overall adaptive profile of diurnal herbivory. The canine sexual dimorphism in some species suggests a possible mating pattern of polygyny, as males in polygynous primate species often compete with each other for mates and have especially large canine teeth.


Omomyoid Diversity
Like adapoids, omomyoids appeared suddenly at the start of the Eocene and then became very diverse with most species dying out before the Oligocene. Omomyoids are known from thousands of jaws with teeth, relatively complete skulls for about a half-dozen species, and very little postcranial material. Omomyoids were relatively small primates, with the largest being less than three kilograms (approximately 7 lbs.; Macrotarsius montanus). All known crania possess a postorbital bar, which in some has been described as “incipient closure.” Some—but not all—known crania have an elongated bony ear tube extending lateral to the location of the eardrum, a feature seen in living tarsiers and catarrhines. The anterior teeth tend to be large, with canines that are usually not much larger than the incisors. Often it is difficult to distinguish closely related species using molar morphology, but the premolars tend to be distinct from one species to another. The postcranial skeleton of most omomyoids shows hallmarks of leaping behavior reminiscent of that of tarsiers. In North America, omomyoids became very diverse and abundant. In fact, omomyoids from Wyoming are sufficiently abundant and from such stratigraphically controlled conditions that they have served as strong evidence for the gradual evolution of anatomical traits over time (Rose and Bown 1984).
Teilhardina (Figure 9.11; see Figure 9.2) is one of the earliest and arguably the most plesiomorphic of omomyoids. Teilhardina has several species, most of which are from North America, with one from Europe (T. belgica) and one from Asia (T. asiatica). The species of this genus are anatomically similar and the deposits from which they are derived are roughly contemporaneous. Thus, this small primate likely dispersed across the northern continents very rapidly (Smith et al. 2006).
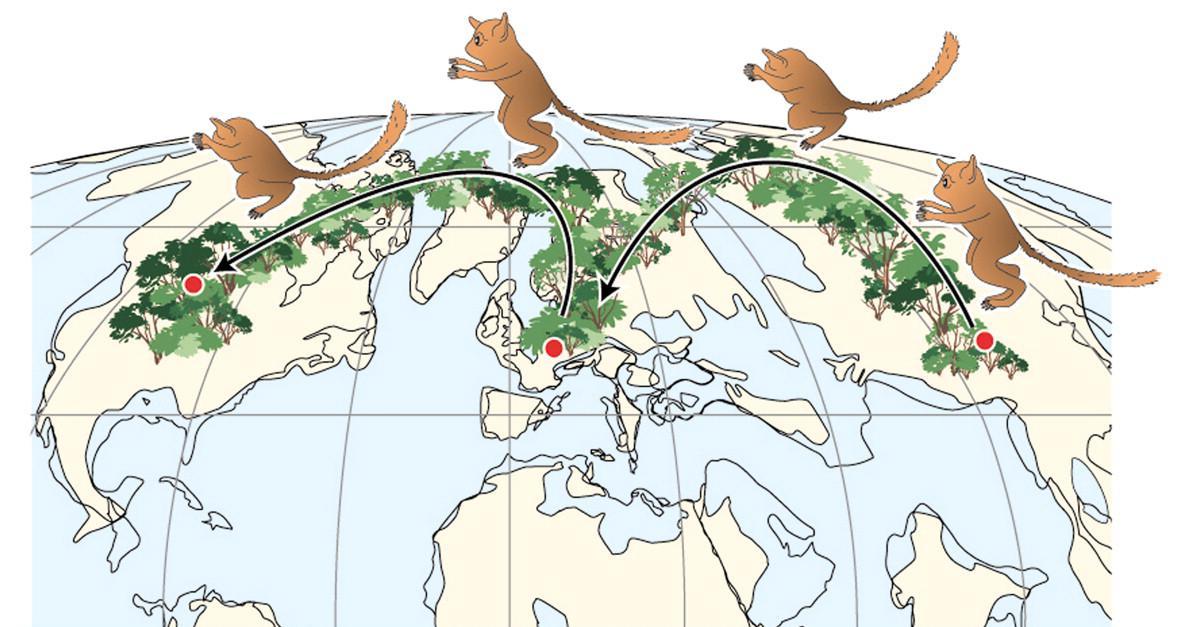
The Emergence of Modern Primate Groups
Origins of Crown Strepsirrhines
Until the turn of this century, very little was known about the origins of the crown (living) strepsirrhines. The Quaternary record of Madagascar contains many amazing forms of lemurs, including giant sloth-like lemurs, lemurs with perhaps monkey-like habits, lemurs with koala-like habits, and even a giant aye-aye (Godfrey and Jungers 2002). However, in Madagascar, early Tertiary continental sediments are lacking, and there is no record of lemur fossils before the Pleistocene.
The fossil record of galagos is slightly more informative. Namely, there are Miocene African fossils that are very likely progenitors of lorisids (Simpson 1967). However, these are much like modern galagos and do not reveal anything about the relationship between crown strepsirrhines and Eocene fossil primates (but see below regarding Propotto). A similar situation exists for lorises in Asia: there are Miocene representatives, but these are substantially like modern lorises. The discovery of the first definite toothcomb canine (a hallmark of stresirrhines) in 2003 provided the “smoking gun” for the origin of crown strepsirrhines (Seiffert et al. 2003). Recently, several other African primates have been recognized as having strepsirrhine affinities (Marivaux et al. 2013; Seiffert 2012). The enigmatic Fayum primate Plesiopithecus is known from a skull that has been compared to aye-ayes and to lorises (Godinot 2006; Simons and Rasmussen 1994a).
The now-recognized diversity of stem strepsirrhines from the Eocene and Oligocene of Afro-Arabia is strong evidence to suggest that strepsirrhines originated in that geographic area. This implies that lorises dispersed to Asia subsequent to an African origin. It is unknown what the first strepsirrhines in Madagascar were like. However, it seems likely that the lemuriform-lorisiform split occurred in continental Africa, followed by dispersal of lemuriform stock to Madagascar. Recent evidence suggests that Propotto, a Miocene primate from Kenya originally described as a potto antecedent, actually forms a clade with Plesiopithecus and the aye-aye; this might suggest that strepsirrhines dispersed to Madagascar from continental Africa more than once (Gunnell et al. 2018).
The Fossil Record of Tarsiers
Tarsiers are so unusual that they fuel major debates about primate taxonomy. Tarsiers today are moderately diverse but geographically limited and not very different in their ecological habits—especially considering that the split between them and their nearest living relative probably occurred over 50 million years ago. If omomyoids are excluded, then the fossil record of tarsiers is very limited. Two fossil species from the Miocene of Thailand have been placed in the genus Tarsius, as has an Eocene fossil from China (Beard et al. 1994). These, and Xanthorhysis from the Eocene of China, are all very tarsier-like. In fact, it is striking that Tarsius eocaenus from China was already so tarsier-like as early as the Eocene. This suggests that tarsiers achieved their current morphology very early in their evolution and have remained more or less the same while other primates changed dramatically. Two additional genera, Afrotarsius from the Oligocene of Egypt and Libya and Afrasia from the Eocene of Myanmar, have also been implicated in tarsier origins, though the relationship between them and tarsiers is unclear (Chaimanee et al. 2012). More recently, a partial skeleton of a small Eocene primate from China, Archicebus achilles (dated to approximately 55.8 million to 54.8 million years ago), was described as the most basal tarsiiform (Ni et al. 2013). This primate is reconstructed as a diurnal insectivore and an arboreal quadruped that did some leaping—but not to the specialized degree seen in living tarsiers. The anatomy of the eye in living tarsiers suggests that their lineage passed through a diurnal stage, so Archicebus (and diurnal omomyoids) might represent such a stage.
Climate Change and the Paleogeography of Modern Primate Origins
Changing global climate has had profound effects on primate dispersal patterns and ecological habits over evolutionary time. Primates today are strongly tied to patches of trees and particular plant parts such as fruits, seeds, and immature leaves. It is no surprise, then, that the distribution of primates mirrors the distribution of forests. Today, primates are most diverse in the tropics, especially in tropical rainforests. Global temperature trends across the Tertiary have affected primate ranges. Following the Cretaceous-Tertiary extinction event, cooler temperatures and greater seasonality characterized the Paleocene. In the Eocene, temperatures (and probably rainfall) increased globally and rainforests likely extended to very high latitudes. During this time, euprimates became diverse. With cooling and increased aridity at the end of the Eocene, many primate extinctions occurred in the northern continents and the surviving primates were confined to lower latitudes in South America, Afro-Arabia, Asia, and southern Europe. Among these survivors are the progenitors of the living groups of primates: lemurs and lorises, tarsiers, platyrrhines (monkeys of the Americas), and catarrhines (monkeys and apes of Africa and Asia) (Figure 9.12).
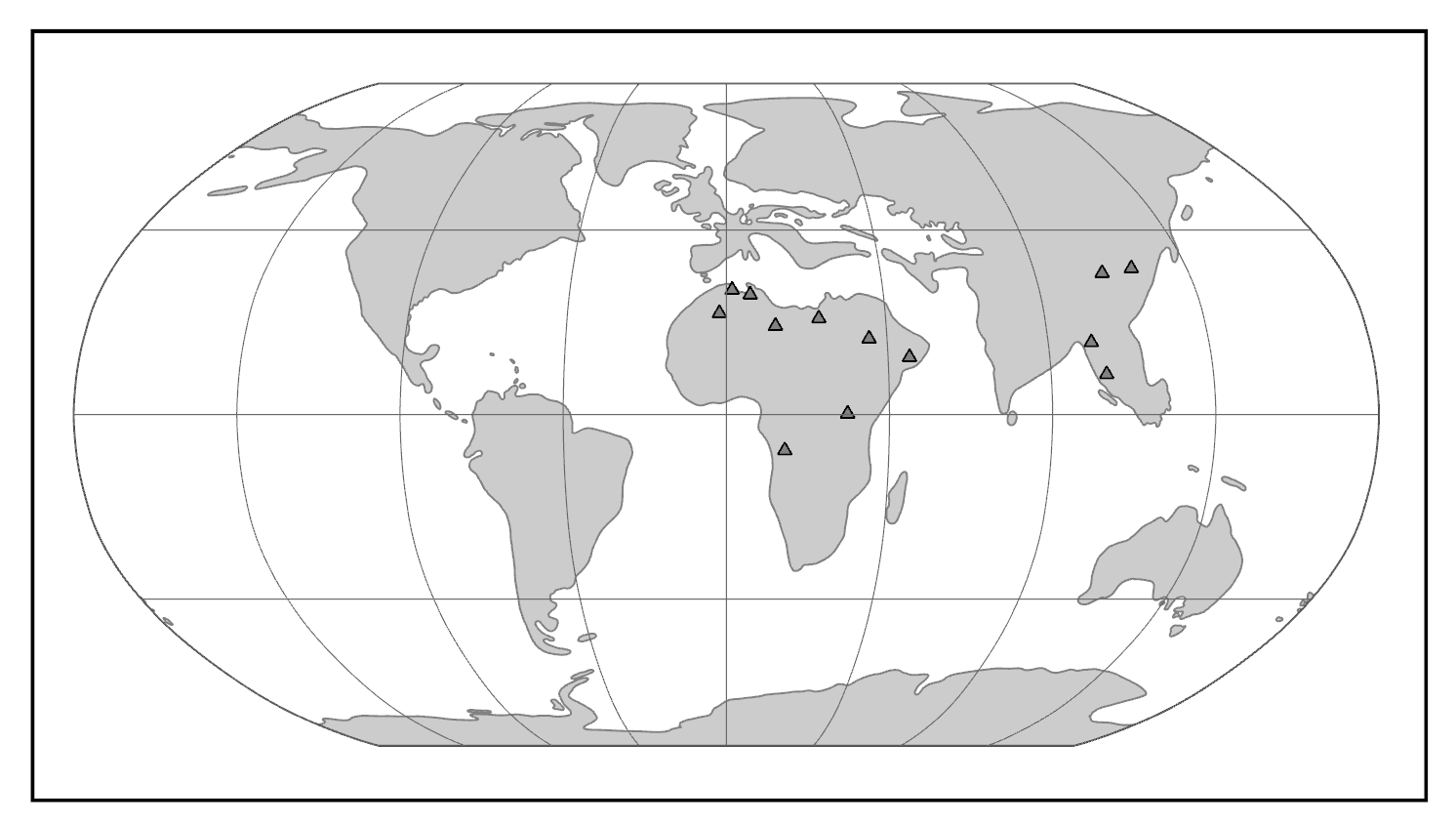
Competing Hypotheses for the Origin of Anthropoids
There is considerable debate among paleoanthropologists as to the geographic origins of anthropoids. In addition, there is debate regarding the source group for anthropoids. Three different hypotheses have been articulated in the literature. These are the adapoid origin hypothesis, the omomyoid origin hypothesis, and the tarsier origin hypothesis (Figure 9.13).
Adapoid Origin Hypothesis
Resemblances between some adapoids and some extant anthropoids include fusion of the mandibular symphysis, overall robusticity of the chewing system, overall large body size, features that signal a diurnal lifestyle (like relatively small eye sockets), and ankle bone morphology. Another feature in common is canine sexual dimorphism, which is present in some species of adapoids (probably) and in several species of anthropoids.
These features led some paleoanthropologists in the last half of the 20th century to suggest that anthropoids came from adapoid stock (Gingerich 1980; Simons and Rasmussen 1994b). One of the earliest supporters of the link between adapoids and anthropoids was Hans Georg Stehlin, who described much of the best material of adapoids and compared these Eocene primates to South American monkeys (Stehlin 1912). In more recent times, the adapoid origin hypothesis was reinforced by resemblances between these European adapoids (especially Adapis and Leptadapis) and some early anthropoids from the Fayum Basin (e.g., Aegyptopithecus, see below; Figure 9.14).
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Propliopithecidae2 |
Aegyptopithecus |
Large size. Cranial sexual dimorphism, large canines. Robust jaws and rounded molars. Partially ossified ear tube (in some). Robust skeleton; quadruped. |
Africa |
Late Eocene to Early Oligocene |
|
Parapithecidae3 |
Apidium |
Medium size. Retention of three premolars per quadrant. Rounded molars and premolars with large central cusps. Adaptations for leaping in the lower limb. |
Africa |
Late Eocene to Late Oligocene |
|
Proteopithecidae4 |
Proteopithecus |
Small size. Retention of three premolars per quadrant. Arboreal quadrupeds that ate fruit. |
Africa |
Late Eocene |
|
Oligopithecidae5 |
Catopithecus |
Small size. Skull has postorbital septum and unfused mandible. Deep jaws. Diet of fruits. Generalized quadruped. |
Africa |
Late Eocene |
|
Eosimiidae |
Eosimias |
Deep jaw with vertical unfused symphysis. Pointed incisors and canines. Crowded premolars. |
Asia |
Middle Eocene |
|
Amphipithecidae6 |
Pondaungia |
Deep jaws. Molars generally rounded with wide basins. |
Asia |
Middle Eocene to Early Oligocene |
|
1 Derived from Fleagle 2013. 2 See Gebo and Simons 1987 and Simons et al. 2007. 3 See Feagle and Simons 1995 and Simons 2001. 4 See Simons and Seiffert 1999. 5 See Simons and Rasmussen 1996. 6 See Kay et al. 2004. |
|
|||
Unfortunately for the adapoid hypothesis, most of the shared features listed above probably emerged independently in the two groups as adaptations to a diet of hard and/or tough foods. For example, fusion of the mandibular symphysis likely evolved as a means to strengthen the jaw against forces that would pull the two halves away from each other, in the context of active chewing muscles on both sides of the head generating great bite forces. This context would also favor the development of robust jaws, large chewing muscles, shorter faces, and some other features shared by some adapoids and some anthropoids.
As older and more plesiomorphic anthropoids were found in the Fayum Basin, it became clear that the earliest anthropoids from Africa did not possess these features of jaw robusticity (Seiffert et al. 2009). Furthermore, many adapoids never evolved these features. Fusion of the mandibular symphysis in adapoids is actually quite different from that in anthropoids and probably occurred during juvenile development in the former (Beecher 1983; Ravosa 1996). Eventually, the adapoid origin hypothesis fell out of favor among most paleoanthropologists, although the description of Darwinius is a recent revival of that idea (Franzen et al. 2009; but see Seiffert et al. 2009, Williams et al. 2010b).
Omomyoid Origin Hypothesis
Similarities in cranial and hindlimb morphology between some omomyoids and extant tarsiers have led to the suggestion that tarsiers arose from some kind of omomyoid. In particular, Necrolemur has many features in common with tarsiers, as does the North American Shoshonius, which is known from a few beautifully preserved (although distorted) crania. Tarsiers and Shoshonius share exclusively some features of the base of the cranium; however, Shoshonius does not have any sign of postorbital closure, and it lacks the bony ear tube of tarsiers. Nevertheless, some of the resemblances between some omomyoids and tarsiers suggest that tarsiers might have originated from within the Omomyoidea (Beard 2002; Beard and MacPhee 1994). In this scenario, although living tarsiers and living anthropoids might be sister taxa, they might have evolved from different omomyoids, possibly separated from each other by more than 50 million years of evolution, or from anthropoids evolved from some non-omomyoid fossil group. The arguments against the omomyoid origin hypothesis are essentially the arguments for the tarsier origin hypothesis (see below). Namely, tarsiers and anthropoids share many features (especially of the soft tissues) that must have been retained for many millions of years or must have evolved convergently in the two groups. Furthermore, a key hard-tissue feature shared between the two extant groups, the postorbital septum, was not present in any omomyoid. Therefore, that feature must have arisen convergently in the two extant groups or must have been lost in omomyoids. Neither scenario is very appealing, although recent arguments for convergent evolution of the postorbital septum in tarsiers and anthropoids have arisen from embryology and histology of the structure (DeLeon et al. 2016).
Tarsier Origin Hypothesis
Several paleoanthropologists have suggested that there is a relationship between tarsiers and anthropoids to the exclusion of omomyoids and adapoids (e.g., Cartmill and Kay 1978; Ross 2000; Williams and Kay 1995). Tarsiers and anthropoids today share several traits, including many soft-tissue features related to the olfactory system (e.g., the loss of a hairless external nose and loss of the median cleft running from the nose to the mouth, as possessed by strepsirrhines), and aspects of the visual system (e.g., the loss of a reflective layer at the back of the eye, similarities in carotid circulation to the brain, and mode of placentation). Unfortunately, none of these can be assessed directly in fossils. Some bony similarities between tarsiers and anthropoids include an extra air-filled chamber below the middle ear cavity, reduced bones within the nasal cavity, and substantial postorbital closure; these can be assessed in fossils, but the distribution of these traits in omomyoids does not yield clear answers. Furthermore, several similarities between tarsiers and anthropoids are probably due to similarities in sensory systems, which might have evolved in parallel for ecological reasons. Although early attempts to resolve the crown primates with molecular data were sometimes equivocal or in disagreement with one another, more recent analyses (including those of short interspersed elements) suggest that tarsiers and anthropoids are sister groups to the exclusion of lemurs and lorises (Williams et al. 2010a). However, this does not address omomyoids, all of which are far too ancient for DNA extraction.
The above three hypotheses are not the only possibilities for anthropoid origins. It may be that anthropoids are neither the closest sister group of tarsiers, nor evolved from adapoids or omomyoids. In recent years, two new groups of Eocene Asian primates have been implicated in the origin of anthropoids: the eosimiids and the amphipithecids. It is possible that one or the other of these two groups gave rise to anthropoids. Regardless of the true configuration of the tree for crown primates, the three major extant groups probably diverged from each other quite long ago (Seiffert et al. 2004).
Early Anthropoid Fossils in Africa


The classic localities yielding the greatest wealth of early anthropoid fossils are those from the Fayum Basin in Egypt (Simons 2008; Figure 9.15). The Fayum is a veritable oasis of fossil primates in an otherwise spotty early Tertiary African record. Since the 1960s, teams led by E. L. Simons have discovered several new species of early anthropoids, some of which are known from many parts of the skeleton and several individuals (Figure 9.16).
The Fayum Jebel Qatrani Formation and Birket Qarun Formation between them have yielded a remarkable array of terrestrial, arboreal, and aquatic mammals. These include ungulates, bats, sea cows, elephants, hyraces, rodents, whales, and primates. Also, many other vertebrates, like water birds, were present. The area at the time of deposition (Late Eocene through Early Oligocene) was probably very wet, with slow-moving rivers, standing water, swampy conditions, and lots of trees (see Bown and Kraus 1988). In short, it was an excellent place for primates.
General Morphology of Anthropoids
The anthropoids known from the Fayum (and their close relatives from elsewhere in East Africa and Afro-Arabia) bear many of the anatomical hallmarks of extant anthropoids; however, there are plesiomorphic forms in several families that lack one or more anthropoid traits. All Fayum anthropoids known from skulls possess postorbital closure, most had fused mandibular symphyses, and most had ring-like ectotympanic bones. Tooth formulae were generally either 2.1.3.3 or 2.1.2.3. Fayum anthropoids ranged in size from the very small Qatrania and Biretia (less than 500 g) to the much-larger Aegyptopithecus (approximately 7 kg; 15 lbs.). Fruit was probably the main component of the diet for most or all of the anthropoids, with some of them supplementing with leaves (Kay and Simons 1980; Kirk and Simons 2001; Teaford et al. 1996). Most Fayum anthropoids were probably diurnal above-branch quadrupeds. Some of them (e.g., Apidium; see Figure 9.14) were probably very good leapers (Gebo and Simons 1987), but none show specializations for gibbon-style suspensory locomotion. Some of the Fayum anthropoids are known from hundreds of individuals, permitting the assessment of individual variation, sexual dimorphism, and in some cases growth and development. The description that follows provides greater detail for the two best known Fayum anthropoid families, the Propliopithecidae and the Parapithecidae; the additional families are summarized briefly.
Fayum Anthropoid Families
The Propliopithecidae (see Figure 9.14) include the largest anthropoids from the fauna, and they are known from several crania and some postcranial elements. They have been suggested to be stem catarrhines, although perhaps near the split between catarrhines and platyrrhines. The best known propliopithecid is Aegyptopithecus, known from many teeth, crania, and postcranial elements (Figure 9.17) .
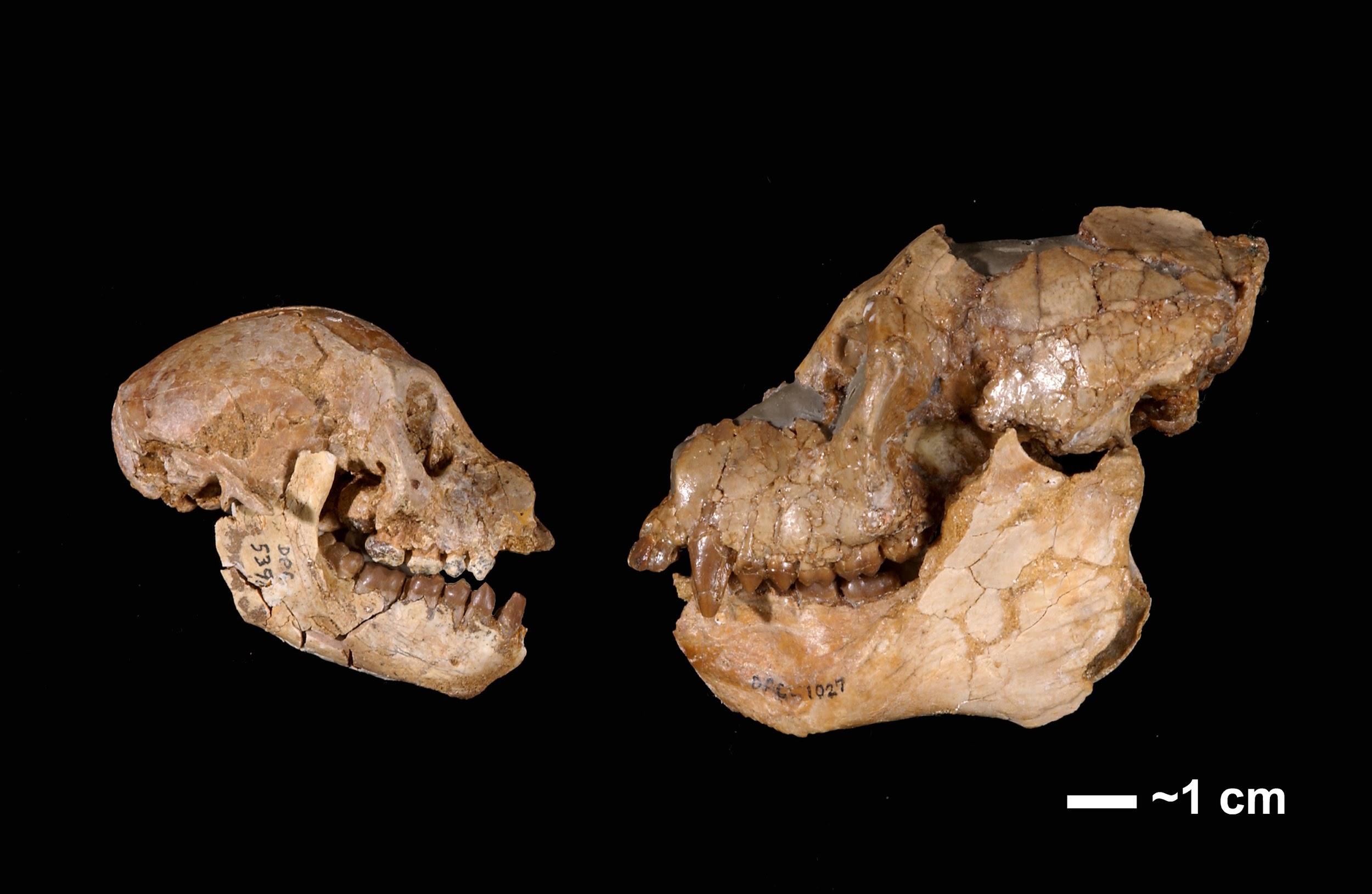
Parapithecidae are an extremely abundant and unusual family of anthropoids from the Fayum. The parapithecid Apidium is known from many jaws with teeth, crushed and distorted crania, and several skeletal elements. Parapithecus is known from cranial material including a beautiful, undistorted cranium. This genus shows extreme reduction of the incisors, including complete absence of the lower incisors in P. grangeri (Simons 2001). This trait is unique among primates. Parapithecids were once thought to be the ancestral stock of platyrrhines; however, their platyrrhine-like features are probably ancestral retentions, so the most conservative approach is to consider them stem anthropoids.
The Proteopithecidae were small frugivores that probably mainly walked along horizontal branches on all fours. They are considered stem anthropoids. The best known genus, Proteopithecus, is represented by dentitions, crania, and postcranial elements.
The Oligopithecidae share a mixture of traits that makes them difficult to classify more specifically within anthropoids. The best known member, Catopithecus, is known from crania that demonstrate a postorbital septum and from mandibles that lack symphyseal fusion. They share the catarrhine tooth formula of 2.1.2.3 and have a canine honing complex that involves the anterior lower premolar. The postcranial elements known for the group suggest generalized arboreal quadrupedalism. The best known member, Catopithecus, is known from crania that demonstrate a postorbital septum and from mandibles that lack symphyseal fusion (Simons and Rasmussen 1996). The jaws are deep, with broad muscle attachment areas and crested teeth. Catopithecus was probably a little less than a kilogram in weight.
Other genera of putative anthropoids from the Fayum include the very poorly known Arsinoea, the contentious Afrotarsius, and the enigmatic Nosmips. The last of these possesses traits of several major primate clades and defies classification (Seiffert et al. 2010).
Early Anthropoid Fossils in Asia
For the last half of the 1900s, researchers believed that Africa was the unquestioned homeland of early anthropoids (see Fleagle and Kay 1994). However, two very different groups of primates from Asia soon began to change that. One was an entirely new discovery (Eosimiidae), and the other was a poorly known group discovered decades prior (Amphipithecidae). Soon, attention on anthropoid origins began to shift eastward (see Ross and Kay 2004; Simons 2004). If anthropoids arose in Asia instead of Africa, then this implies that the African early anthropoids either emigrated from Asia or evolved their anthropoid traits in parallel with living anthropoids.
Eosimiids
First described in the 1990s, the eosimiids are best represented by Eosimias (see Figure 9.14; Figure 9.18). This tiny “dawn monkey” is known from relatively complete jaws with teeth, a few small fragments of the face, and some postcranial elements (Beard et al. 1994; Beard et al. 1996; Gebo et al. 2000). Eosimias (along with the other less-well-known genera in its family) bears some resemblance to tarsiers as well as anthropoids. Unfortunately, no good crania are known for this family, and the anatomy of, for example, the posterior orbital margin could be very revealing as to higher-level relationships.
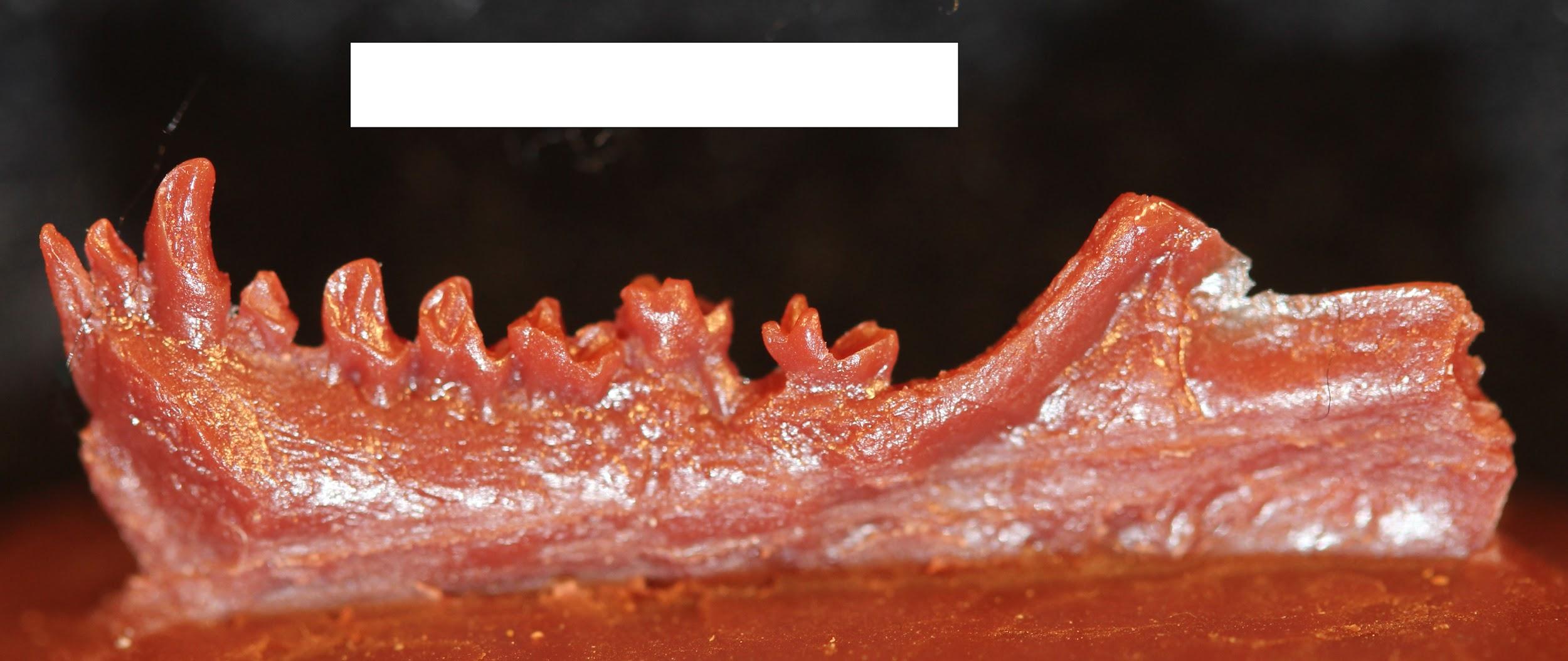
Amphipithecids
Amphipithecids are small- to medium-size primates (up to 10 kg; 22 lbs.). Most are from the Eocene Pondaung Formation in Myanmar (Early–Middle Eocene), but one genus is known from Thailand. Some dental similarities with anthropoids were noted early on, such as deep jaws and wide basins that separate low molar cusps. The best known genera were Pondaungia and Amphipithecus (Ciochon and Gunnell 2002; see Figure 9.14). Another amphipithecid, Siamopithecus from Thailand, has very rounded molars and was probably a seed-eater (Figure 9.19). In addition to teeth and jaws, some cranial fragments, ankle material, and ends of postcranial bones have been found for Pondaungia. There are important resemblances between the postcranial bones of Pondaungia and those of adapoids, suggesting adapoid affinities for the amphipithecidae. This would imply that the resemblances with anthropoids in the teeth are convergent, based on similarities in diet (see Ciochon and Gunnell 2002). Unfortunately, the association between postcranial bones and teeth is not definite. With other primates in these faunas (including eosimiids), one cannot be certain that the postcranial bones belong with the teeth. Some researchers suggest that some bones belong to a sivaladapid (or asiadapid) and others to an early anthropoid (Beard et al. 2007; Marivaux et al. 2003). Additional well-associated material of amphipithecids would help to clear up this uncertainty.
Platyrrhine Dispersal to South America
Today there is an impressive diversity of primates in South and Central America. These are considered to be part of a single clade, the Platyrrhini. Primates colonized South America sometime in the Eocene from an African source. In the first half of the 20th century, the source of platyrrhines was a matter of major debate among paleontologists, with some favoring a North American origin (e.g., Simpson 1940).
Part of the reason for this debate is that South America was an island in the Eocene. Primates needed to cross open ocean to get there from either North America or Africa, although the distance from the former was shorter. Morphology yields clues to platyrrhine origins. The first known primates in South America have more in common morphologically with African primates than with North American ones. At the time, anthropoids were popping up in North Africa, whereas the only euprimates in North America were adapoids and omomyoids. Despite lacking a bony ear tube, early platyrrhines shared a great deal with other anthropoids, including full postorbital closure and fusion of the mandibular symphysis.
The means by which a population of small North African primates managed to disperse across the Atlantic and survive to colonize South America remains a mystery. The most plausible scenario is one of rafting. That is, primates must have been trapped on vegetation that was blown out to sea by a storm. The vegetation then became a sort of life raft, which eventually landed ashore, dumping its passengers in South America. Rodents probably arrived in South America in the same way (Antoine et al. 2012).
Once ashore, platyrrhines must have crossed South America fairly rapidly because the earliest-known primates from that continent are from Peru (Bond et al. 2015). Soon after that, platyrrhines were in Bolivia, namely Branisella. By the Miocene, platyrrhines were living in extreme southern Argentina and were exploiting a variety of feeding niches. The Early Miocene platyrrhines were all somewhat plesiomorphic in their morphology, but some features that likely arose by ecological convergence suggest (to some) relationships with extant platyrrhine families. This has led to a lively debate about the pattern of primate evolution in South America (Kay 2015; Kay and Fleagle 2010; Rosenberger 2010). By the Middle Miocene, clear representatives of modern families were present in a diverse fauna from La Venta, Colombia (Wheeler 2010). The Plio-Pleistocene saw the emergence of giant platyrrhines as well as several taxa of platyrrhines living on Caribbean islands (Cooke et al. 2016).
The story of platyrrhines seems to be one of amazing sweepstakes dispersal, followed by rapid diversification and widespread geographic colonization of much of South America. After that, dramatic extinctions resulted in the current, much-smaller geographic distribution of platyrrhines. These extinctions were probably caused by changing climates, leading to the contraction of forests. Platyrrhines dispersed to the Caribbean and to Central America, with subsequent extinctions in those regions that might have been related to interactions with humans. Unlike anthropoids of Africa and Asia, platyrrhines do not seem to have evolved any primarily terrestrial forms and so have always been highly dependent on forests.
Special Topic: Jonathan Perry and Primates of the Extreme South
Many primates are very vulnerable to ecological disturbance because they are heavily dependent on fruit to eat and trees to live in. This is one reason why so many primates are endangered today and why many of them went extinct due to climatic and vegetational changes in the past. I (Jonathan Perry) have conducted paleontological research focusing on primates that lived on the edge of their geographic distribution. This research has taken me to extreme environments in the Americas: southern Patagonia, the Canadian prairies, western Wyoming, and the badlands of eastern Oregon.
Santa Cruz Province in Argentina is as far south as primates have ever lived. The Santa Cruz fauna of the Miocene has yielded a moderate diversity of platyrrhines, each with slightly different dietary adaptations. These include Homunculus, first described by Florentino Ameghino in 1891 (Figure 9.20). Recent fieldwork by my colleagues and I in Argentina has revealed several skulls of Homunculus as well as many parts of the skeleton (Kay et al. 2012). The emerging profile of this extinct primate is one of a dedicated arboreal quadruped that fed on fruits and leaves. Many of the foods eaten by Homunculus must have been very tough and were probably covered and impregnated with grit; we suspect this because the cheek teeth are very worn down, even in young individuals, and because the molar tooth roots were very large, presumably to resist strong bite forces (Perry et al. 2010, 2014).
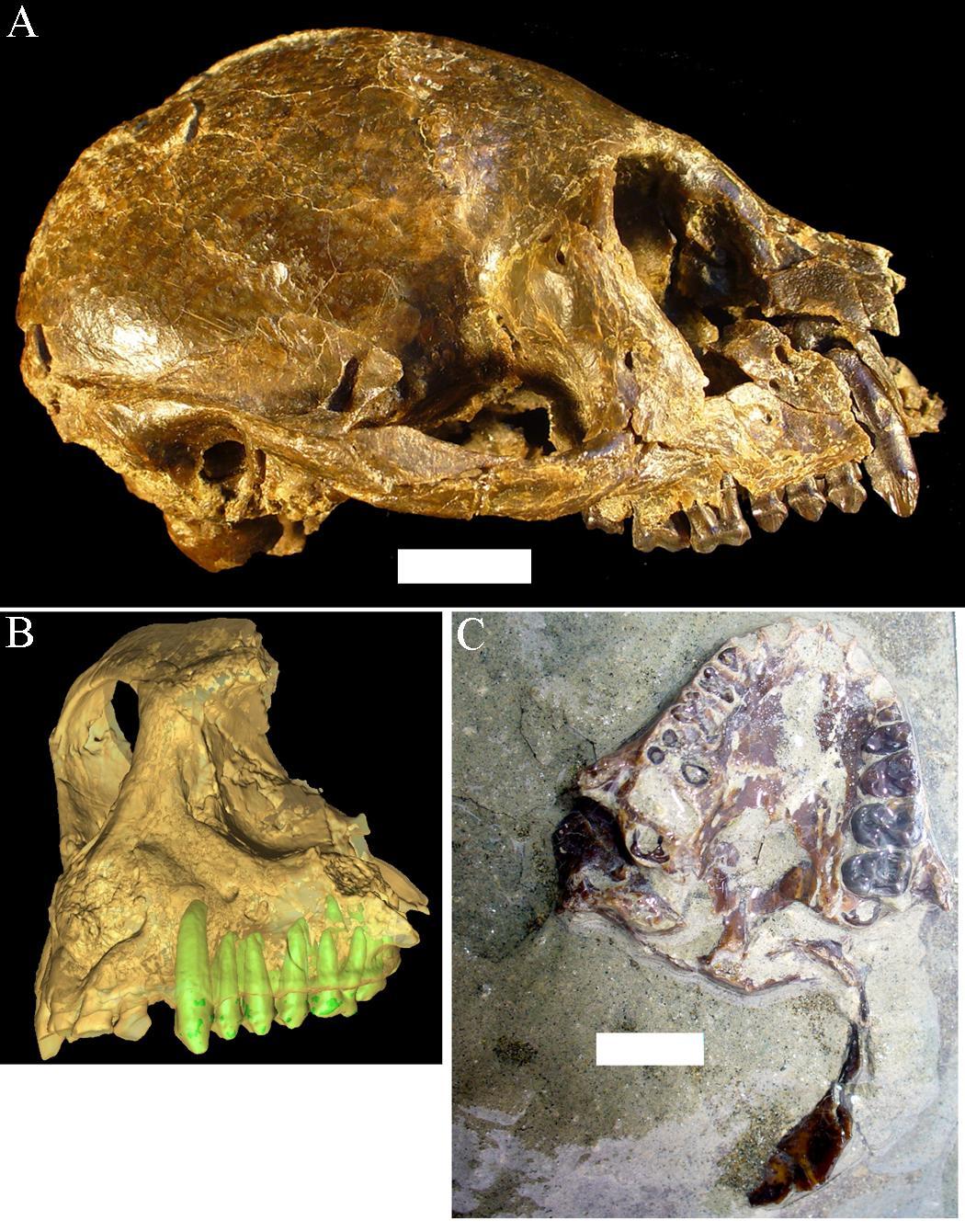
I began working in Argentina while a graduate student at Duke University. I participated as a field assistant in a team led by my Ph.D. advisor, Richard F. Kay, and Argentine colleagues Sergio F. Vizcaíno and M. Susana Bargo. Most of the localities examined belong to a suite of beach sites known since the 1800s and visited by many field parties from various museums in the early 1900s. Since 2003, our international team of paleontologists from the U.S. and Argentina has visited these localities every single year (Figure 9.21). Over time, new fossils and new students have led to new projects and new approaches, including the use of microcomputed tomography (microCT) to visualize and analyze internal structures of the skeleton.

Planet of Apes
Geologic Activity and Climate Change in the Miocene
The Miocene Epoch was a time of mammalian diversification and extinction, global climate change, and ecological turnover. In the Miocene, there was an initial warming trend across the globe with the expansion of subtropical forests, followed by widespread cooling and drying with the retreat of tropical forests and replacement with more open woodlands and eventually grasslands. It was also a time of major geologic activity. On one side of the globe, South America experienced the rise of the Andes Mountains. On the other side, the Indian subcontinent collided with mainland Asia, resulting in the rise of the Himalayan Mountains. In Africa, volcanic activity promoted the development of the East African Rift System. Critical to the story of ape evolution was the exposure of an intercontinental landbridge between East Africa and Eurasia, permitting a true planet of apes (Figure 9.22).
Geographic Distribution: Africa, Asia, Europe
The world of the Miocene had tremendous ape diversity compared to today. The earliest records of fossil apes are from Early Miocene deposits in Africa. However, something dramatic happened around 16 million years ago. With the closure of the ancient Tethys Sea, the subsequent exposure of the Gomphotherium Landbridge, and a period of global warming, the Middle–Late Miocene saw waves of emigration of mammals (including primates) out of Africa and into Eurasia, with evidence of later African re-entry for some (Harrison 2010). Some of the mammals that dispersed from Africa to Eurasia and back were apes. Though most of these early apes left no modern descendants, some of them gave rise to the ancestors of modern apes—including hominins (Figure 9.23).
Where Are the Monkeys? Diversity in the Miocene
Whereas the Oligocene deposits in the Fayum of Egypt have yielded the earliest-known catarrhine fossils, the Miocene demonstrates some diversification of Cercopithecoidea. However, compared to the numerous and diverse Miocene apes (see below), monkeys of the Miocene are very rare and restricted to a single extinct family, the Victoriapithecidae (Figure 9.24). This family contains the earliest definite cercopithecoids. These monkeys are found from northern and eastern Africa between 20 million and 12.5 million years ago (Miller et al. 2009). The best known early African monkey is Victoriapithecus (Figure 9.25), a small-bodied (approximately 7 kg; 15 lbs.), small-brained monkey. Bilophodonty, known to be a hallmark of molar teeth of modern cercopithecoid, was present to some extent in Victoriapithecids. Victoriapithecus has been reconstructed as being more frugivorous and perhaps spent more time on the ground (terrestrial locomotion) than in the trees (arboreal locomotion; Blue et al. 2006). The two major groups of cercopithecoids today are cercopithecines and colobines. The earliest records demonstrating clear members of each of these two groups are at the end of the Miocene. Examples include the early colobine Microcolobus from Kenya and the early cercopithecine Pliopapio from Ethiopia.
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Victoriapithecidae2 |
Victoriapithecus |
Long, sloping face. Round, narrowly spaced orbits. Deep cheek bones. Well-developed sagittal crest. |
Africa |
Early to Middle Miocene |
|
Proconsulidae3 |
Proconsul |
Short face. Generalized dentition. Arboreal quadruped. Probably tailless. |
Africa and Arabia |
Early to Middle Miocene |
|
Pongidae |
Gigantopithecus |
Largest primate ever. Deep jaws and low rounded molars. |
Asia |
Miocene to Present |
|
1 Derived from Fleagle 2013. 2 See Benefit and McCrossin 1997 and Fleagle 2013. 3 See Begun 2007. |
|
|||
The Story of Us, the Apes
African Ape Diversity
The Early Miocene of Africa has yielded around 14 genera of early apes (Begun 2003). Many of these taxa have been reconstructed as frugivorous arboreal quadrupeds (Kay 1977). One of the best studied of these genera is the East African Proconsul (Family Proconsulidae; see Figure 9.24). Several species have been described, with body mass reconstructions ranging from 17 to 50 kg (approximately 37–110 lbs.). A paleoenvironmental study reconstructed the habitat of Proconsul to be a dense, closed-canopy tropical forest (Michel et al. 2014). No caudal vertebrae (tail bones) have been found in direct association with Proconsul postcrania, and the morphology of the sacrum is consistent with Proconsul lacking a tail (Russo 2016; Ward et al. 1991).
Overall, the African ape fossil record in the Late Miocene is sparse, with seven fossil localities dating between eleven and five million years ago (Pickford et al. 2009). Nevertheless, most species of great apes live in Africa today. Where did the progenitors of modern African apes arise? Did they evolve in Africa or somewhere else? The paucity of apes in the Late Miocene of Africa stands in contrast to the situation in Eurasia. There, ape diversity was high. Furthermore, several Eurasian ape fossils show morphological affinities with modern hominoids (apes). Because of this, some paleoanthropologists suggest that the ancestors of modern African great apes recolonized Africa from Eurasia toward the end of the Miocene (Begun 2002). However, discoveries of Late Miocene hominoids like the Kenyan Nakalipithecus (9.9 million to 9.8 million years ago), the Ethiopian Chororapithecus (10.7 million to 10.1 million years ago), and the late-Middle Miocene Namibian Otavipithecus (13 million to 12 million years ago) fuel an alternative hypothesis—namely that African hominoid diversity was maintained throughout the Miocene and that one of these taxa might, in fact, be the last common ancestor of extant African apes (Kunimatsu et al. 2007; Mocke et al. 2002). The previously underappreciated diversity of Late Miocene apes in Africa might be due to poor sampling of the fossil record in Africa.
Eurasian Ape Diversity
With the establishment of the Gomphotherium Landbridge (a result of the closure of the Eastern Mediterranean seaway; Rögl 1999), the Middle Miocene was an exciting time for hominoid radiations outside of Africa (see Figure 9.23). Eurasian hominoid species exploited their environments in many different ways in the Miocene. Food exploitation ranged from soft-fruit feeding in some taxa to hard-object feeding in others, in part owing to seasonal fluctuations and the necessary adoptions of fallback foods (DeMiguel et al. 2014). For example, the molars of Oreopithecus bambolii (Family Hominidae) have relatively long lower-molar shearing crests, suggesting that this hominoid was very folivorous (Ungar and Kay 1995). Associated with variation in diet, there is great variation in the degree to which cranial features (e.g., zygomatic bone or supraorbital tori) are developed across the many taxa (Cameron 1997); however, Middle Miocene fossils tend to exhibit relatively thick molar enamel and relatively robust jaws (Andrews and Martin 1991).
In Spain, the cranium with upper dentition, part of a mandible, and partial skeleton of Pliobates (Family Pliobatidae), a small-bodied ape (4–5 kg; 9–11 lbs.), was discovered in deposits dating to 11.6 million years ago (Alba et al. 2015). It is believed to be a frugivore with a relative brain size that overlaps with modern cercopithecoids. The fossilized postcrania of Pliobates suggest that this ape might have had a unique style of locomotion, including the tendency to walk across the branches of trees with its palms facing downward and flexible wrists that permitted rotation of the forearm during climbing. However, the anatomy of the distal humerus differs from those of living apes in ways that suggest that Pliobates was less efficient at stabilizing its elbow while suspended (Benefit and McCrossin 2015). Two other recently described apes from Spain, Pierolapithecus and Anoiapithecus, are known from relatively complete skeletons. Pierolapithecus had a very projecting face and thick molar enamel as well as some skeletal features that suggest (albeit controversially) a less suspensory locomotor style than in extant apes (Moyà-Solà et al. 2004). In contrast to Pierolapithecus, the slightly younger Anoiapithecus has a very flat face (Moyà-Solà et al. 2009).
Postcranial evidence for suspensory or well-developed orthograde behaviors in apes does not appear until the Late Miocene of Europe. Primary evidence supporting these specialized locomotor modes includes the relatively short lumbar vertebrae of Oreopithecus (Figure 9.26) and Dryopithecus (Maclatchy 2004). Further, fossil material of the lower torso of O. bambolii (which dates to the Pan-hominin divergence) conveys a higher degree of flexion-extension abilities in the lumbar region (lower back) than what is possible in extant apes. Additionally, the hindlimb of O. bambolii is suggested to have supported powerful hip adduction during climbing (Hammond et al. 2020). The Late Miocene saw the extinction of most of the Eurasian hominoids in an event referred to as the Vallesian Crisis (Agustí et al. 2003). Among the latest surviving hominoid taxa in Eurasia were Oreopithecus and Gigantopithecus, the latter of which held out until the Pleistocene in Asia and was probably even sympatric with Homo erectus (Cachel 2015).

The Origins of Extant Apes
The fossil record of the extant apes is somewhat underwhelming: it ranges from being practically nonexistent for some taxa (e.g., chimpanzees) to being a little better for others (e.g., humans). There are many possible reasons for these differences in fossil abundance, and many are associated with the environmental conditions necessary for the fossilization of bones. One way to understand the evolution of extant apes that is not so dependent on the fossil record is via molecular evolutionary analyses. This can include counting up the differences in the genetic sequence between two closely related species to estimate the amount of time since these species shared a common ancestor. This is called a molecular clock, and it is often calibrated using fossils of known absolute age that stand in for the last common ancestor of a particular clade. Molecular clock estimates have placed the Hylobatidae and Hominidae split between 19.7 million and 24.1 million years ago, the African ape and Asian ape split between 15.7 million and 19.3 million years ago, and the split of Hylobatidae into its current genera between 6.4 million and 8 million years ago (Israfil et al. 2011).
Small Ape Origins and Fossils
Unfortunately, the fossil record for the small (formerly “lesser”) apes is meager, particularly in Miocene deposits. One possible early hylobatid is Laccopithecus robustus, a Late Miocene catarrhine from China (Harrison 2016). Although it does share some characteristics with modern gibbons and siamangs (including an overall small body size and a short face), Laccopithecus most likely represents a plesiomorphic stem catarrhine and is therefore distantly related to extant apes (Jablonski and Chaplin 2009). A more likely candidate for the hylobatid stem is another Late Miocene taxon from China, Yuanmoupithecus xiaoyuan. Interpretation of its phylogenetic standing, however, is complicated by contradicting dental features—some of them quite plesiomorphic—which some believe best place Yuanmoupithecus as a stem hylobatid (Harrison 2016). Recently, a Middle Miocene Indian fossil ape, Kapi ramnagarensis, has extended the fossil record of small apes by approximately five million years. Its teeth are suggestive of a shift to a more frugivorous diet and it is likely a stem hylobatid (Gilbert et al. 2020). The history of Hylobatidae becomes clearer in the Pleistocene, with fossils representing extant genera.
Great Ape Origins and Fossils
The most extensive fossil record of a modern great ape is that of our own genus, Homo. However, the evolutionary history of the Asian great ape, the orangutan (Pongo), is becoming clearer. Today, orangutans are found only on the islands of Borneo and Sumatra. However, Pleistocene-aged teeth, attributed to Pongo, have been found in Cambodia, China, Laos, Peninsular Malaysia, and Vietnam—demonstrating the vastness of the orangutan’s previous range (Ibrahim et al. 2013; Wang et al. 2014). Sivapithecus from the Miocene of India and Pakistan is represented by many specimens, including parts of the face. Sivapithecus is very similar to Pongo, especially in the face, and it probably is closely related to ancestral orangutans (Pilbeam 1982). Originally, jaws and teeth belonging to the former genus Ramapithecus were thought to be important in the origin of humans (Simons 1961), but now these are recognized as specimens of Sivapithecus (Kelley 2002). Postcranial bones of Sivapithecus, however, suggest a more generalized locomotor mode—including terrestrial locomotion—than seen in Pongo (Pilbeam et al. 1990). Stable carbon and oxygen isotope data from dental enamel have reconstructed the paleoecological space of Sivapithecus (as well as the contemporaneous Late Miocene pongine Khoratpithecus) within the canopies of forested habitats (Habinger et al. 2022).
A probable close relative of Sivapithecus is the amazing Gigantopithecus (see Figure 9.24). Known only from teeth and jaws from China and India (e.g., Figure 9.27), this ape probably weighed as much as 270 kg (595 lbs.) and was likely the largest primate ever (Bocherens et al. 2017). Because of unique features of its teeth (including molarized premolars and patterns of wear) and its massive size, it has been reconstructed as a bamboo specialist, somewhat like the modern panda. Small silica particles (phytoliths) from grasses have been found stuck to the molars of Gigantopithecus (Ciochon et al. 1990). Recent studies evaluating the carbon isotope composition of the enamel sampled from Gigantopithecus teeth suggest that this ape exploited a wide range of vegetation, including fruits, leaves, roots, and bamboo (Bocherens et al. 2017). Its face is reminiscent of that of modern orangutans and it might belong in the same family, Pongidae (Kelley 2002).
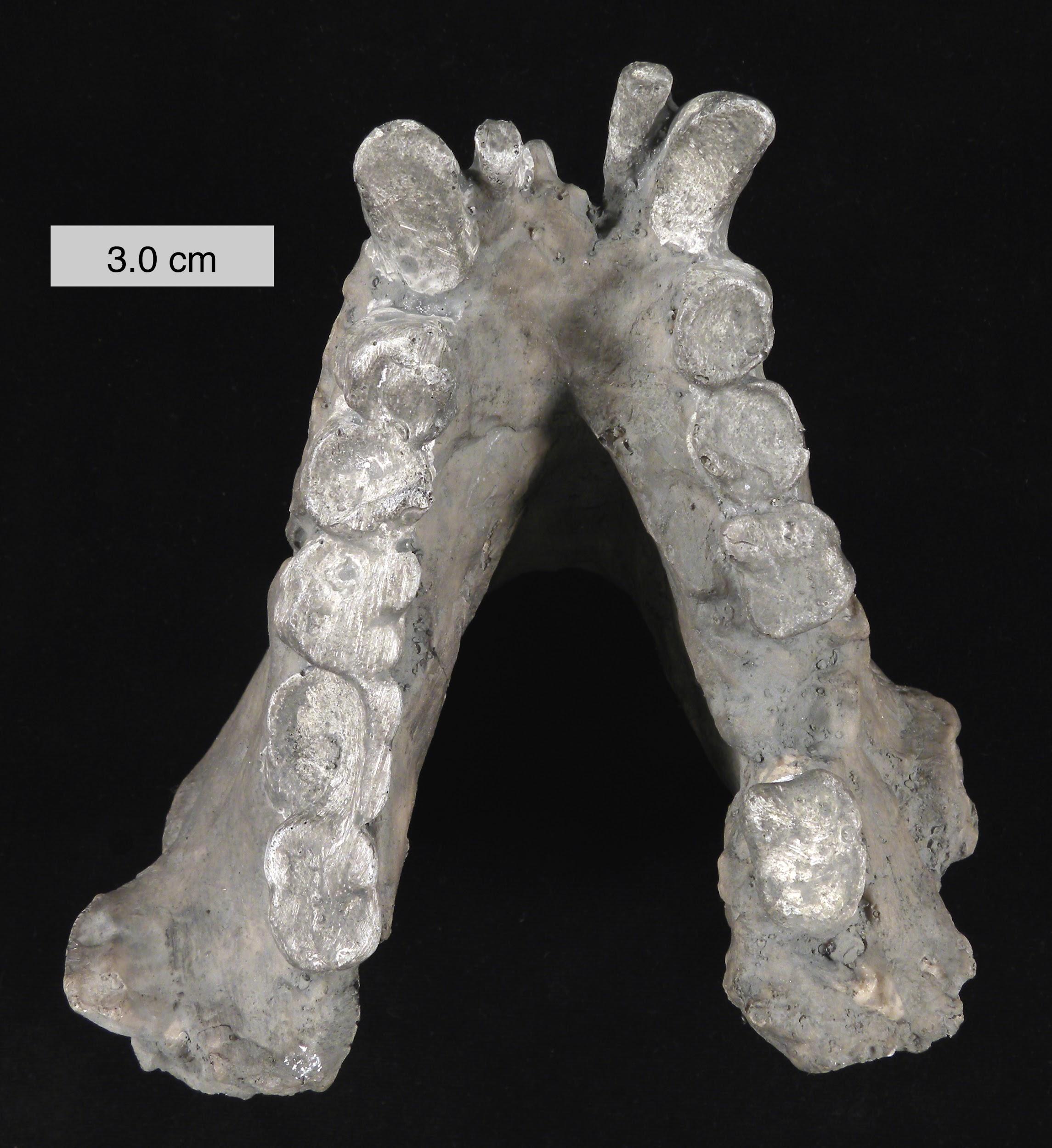
In Africa, the first fossil to be confidently attributed to Pan, and known to be the earliest evidence of a chimpanzee, was described based on teeth found in Middle Pleistocene deposits in the Eastern Rift Valley of Kenya (McBrearty and Jablonski 2005). Paleoenvironmental reconstructions of this locality suggest that this early chimpanzee was living in close proximity to early Homo in a closed-canopy wooded habitat. Similarly, fossil teeth and mandibular remains attributed to two species of Middle-Late Miocene apes—Chororapithecus abyssinicus (from Ethiopia; Suwa et al. 2007) and Nakalipithecus nakayamai (from Kenya; Kunimatsu et al. 2007)—have been suggested as basal members of the gorilla clade.
While the deposits of Eastern Africa have yielded a profound record of our fossil hominin ancestors, the continent’s rainforests remain a “palaeontological desert” (Rosas et al. 2022). Clearly, more work is needed to fill in the large gaps in the fossil record of the nonhuman great apes. The twentieth century witnessed the discovery of many hominin fossils in East Africa, which have been critical for improving our understanding of human evolution. While twenty-first-century conservationists fight to prevent the extinction of the living great apes, perhaps efforts by twenty-first-century paleoanthropologists will yield the evolutionary story of these, our closest relatives.
Summary
While there are large gaps in the fossil records linking primates to early hominins, evolutionary trends make it clear that humans are one branch of the broader primate family tree. In this chapter we go over the major development that characterize primate evolution: enhanced vision, grasping hands and feet, greater reliance on social behavior, and increased brain complexity. It is these traits which distinguish primates from other mammals and furthermore, help define major subsections within the primate class, such as strepsirrhines, haplorhines, monkeys, apes, and ultimately hominins.
Within this chapter, we also examine how anthropologists reconstruct these evolutionary relationships. Fossil evidence has provided key information about when and where different primates lived, while genetic data such as skeletal features allow researchers to understand how extinct species moved, ate, and interacted with their environments. Just as crucial are the influences of Earth’s changing environments: continental drift, glacial cycles, and long-term climate shifts have repeatedly reshaped habitats, driving both extinctions and the emergence of new adaptive forms, including the emergence of our own human lineage.
Review Questions
- Compare three major hypotheses about primate origins, making reference to each one’s key ecological reason for primate uniqueness.
- Explain how changes in temperature, rainfall, and vegetation led to major changes in primate biogeography over the Early Tertiary.
- List some euprimate features that plesiadapiforms have and some that they lack.
- Contrast adapoids and omomyoids in terms of life habits.
- Describe one piece of evidence for each of the adapoid, omomyoid, and tarsier origin hypotheses for anthropoids.
- Discuss the biogeography of the origins of African great apes and orangutans using examples from the Miocene ape fossil record.
Key Terms
Adapoidea: Order: Primates. One of the earliest groups of euprimates (true primates; earliest records from the early Eocene).
Adaptive radiations: Rapid diversifications of single lineages into many species which may present unique morphological features in response to different ecological settings.
Ancestral traits: Features that were inherited from a common ancestor and which remain (largely) unchanged.
Anthropoids:Group containing monkeys and apes, including humans.
Auditory bulla: The rounded bony floor of the middle ear cavity.
Bilophodonty: Dental condition in which the cusps of molar teeth form ridges (or lophs) separated from each other by valleys (seen, e.g., in modern catarrhine monkeys).
Catarrhines: Order: Primates; Suborder: Anthropoidea; Infraorder: Catarrhini. Group, with origins in Africa and Asia, that contains monkeys and apes, including humans.
Clade:Group containing all of the descendants of a single ancestor. A portion of a phylogenetic tree represented as a bifurcation (node) in a lineage and all of the branches leading forward in time from that bifurcation.
Convergent evolution: The independent evolution of a morphological feature in animals not closely related (e.g., wings in birds and bats).
Crown: Smallest monophyletic group (clade) containing a specified set of extant taxa and all descendants of their last common ancestor.
Diastema: Space between adjacent teeth.
Diffuse coevolution: The ecological interaction between whole groups of species (e.g., primates) with whole groups of other species (e.g., fruiting trees).
Ectotympanic: Bony ring or tube that holds the tympanic membrane (eardrum).
Euprimates: Order: Primates. True primates or primates of modern aspect.
Haplorhines: Group containing catarrhines, platyrrhines, and tarsiers.
Hominins: Modern humans and any extinct relatives more closely related to us than to chimpanzees.
Mandibular symphysis: Fibrocartilaginous joint between the left and right mandibular segments, located in the midline of the body.
Omomyoidea: Order: Primates; Superfamily: Omomyoidea. One of the earliest groups of euprimates (true primates; earliest record in the early Eocene).
Petrosal bone: The portion of the temporal bone that houses the inner ear apparatus.
Plagiaulacoid: Dental condition where at least one of the lower cheek-teeth (molars or premolars) is a laterally compressed blade.
Platyrrhines: Order: Primates; Suborder: Anthropoidea; Infraorder: Platyrrhini. Group containing monkeys found in the Americas.
Plesiadapiforms: Order: Plesiadapiformes. Archaic primates or primate-like placental mammals (Early Paleocene–Late Eocene).
Plesiomorphic: Having features that are shared by different groups which arose from a common ancestor.
Stem: Taxa that are basal to a given crown group but are more closely related to the crown group than to the closest living sister taxon of the crown group.
Strepsirrhines: Order: Primates; Suborder: Stresirrhini. Group containing lemurs, lorises, and galagos (does not include tarsiers).
Toothcomb: Dental condition found in modern strepsirrhines in which the lower incisors and canines are laterally compressed and protrude forward at a nearly horizontal inclination. This structure is used in grooming.
For Further Exploration
Beard, Chris. 2004. The Hunt for the Dawn Monkey: Unearthing the Origins of Monkeys, Apes, and Humans. Berkeley: University of California Press.
Begun, David R. 2010. “Miocene Hominids and the Origins of the African Apes and Humans.” Annual Review of Anthropology 39: 67–84.
Fleagle, John G. 2013. Primate Adaptation and Evolution. Third edition. San Diego, CA: Academic Press.
Gebo, Daniel L., ed. 1993. Postcranial Adaptations in Nonhuman Primates. Dekalb: Northern Illinois University Press.
Godfrey, Laurie R., and William L. Jungers. 2002. “Quaternary Fossil Lemurs.” In The Primate Fossil Record, edited by Walter C. Hartwig, 97–121. Cambridge: Cambridge University Press.
Godinot, Marc. 2006. “Lemuriform Origins as Viewed from the Fossil Record.” Folia Primatologica 77 (6): 446–464.
Kay, Richard F. 2018. “100 Years of Primate Paleontology.” American Journal of Physical Anthropology 165 (4): 652–676.
Marivaux, Laurent. 2006. “The Eosimiid and Amphipithecid Primates (Anthropoidea) from the Oligocene of the Bugti Hills (Balochistan, Pakistan): New Insight into Early Higher Primate Evolution in South Asia.” Palaeovertebrata, Montpellier 34 (1–2): 29–109.
Martin, R. D. 1990. Primate Origins and Evolution: A Phylogenetic Reconstruction. Princeton: Princeton University Press.
Rose, Kenneth D., Marc Godinot, and Thomas M. Bown. 1994. “The Early Radiation of Euprimates and the Initial Diversification of Omomyidae.” In Anthropoid Origins: The Fossil Evidence, edited by John G. Fleagle and Richard F. Kay, 1–28. New York: Plenum Press.
Ross, Callum F. 1999. “How to Carry Out Functional Morphology.” Evolutionary Anthropology 7 (6): 217–222.
Seiffert, Erik R. 2012. “Early Primate Evolution in Afro-Arabia.” Evolutionary Anthropology: Issues, News, and Reviews 21(6): 239–253.
Szalay, Frederic S., and Eric Delson. 1979. Evolutionary History of the Primates. New York: Academic Press.
Ungar, Peter S. 2002. “Reconstructing the Diets of Fossil Primates.” In Reconstructing Behavior in the Primate Fossil Record, edited by Joseph Plavcan, Richard F. Kay, William Jungers, and Carel P. van Schaik, 261–296. New York: Kluwer Academic/Plenum Publishers.
References
Agustí, J., A. Sanz de Siria, and M. Garcés M. 2003. “Explaining the End of the Hominoid Experiment in Europe.” Journal of Human Evolution 45 (2): 145–153.
Alba, David M., Sergio Almécija, Daniel DeMiguel, Josep Fortuny, Miriam Pérez de los Ríos, Marta Pina, Josep M. Robles, and Salvador Moyà-Solà. 2015. “Miocene Small-Bodied Ape from Eurasia Sheds Light on Hominoid Evolution.” Science 350 (6260): aab2625.
Andrews, Peter, and Lawrence Martin. 1991. “Hominoid Dietary Evolution.” Philosophical Transactions of the Royal Society of London B: Biological Sciences 334 (1270): 199–209.
Antoine, Pierre-Oliver, Laurent Marivaux, Darren A. Croft, Guillaume Billet, Morgan Ganerød, Carlos Jaramillo, Thomas Martin, et al. 2012. “Middle Eocene Rodents from Peruvian Amazonia Reveal the Pattern and Timing of Caviomorph Origins and Biogeography.” Proceedings of the Royal Society B: Biological Sciences 279 (1732): 1319–1326.
Beard, K. Christopher. 1990. “Gliding Behaviour and Palaeoecology of the Alleged Primate Family Paromomyidae (Mammalia, Dermoptera).” Nature 345 (6273): 340–341.
Beard, K. Christopher. 2002. “Basal Anthropoids.” In The Primate Fossil Record, edited by William C. Hartwig, 133–150. Cambridge: Cambridge University Press.
Beard, K. Christopher, and R. D. E. MacPhee. 1994. “Cranial Anatomy of Shoshonius and the Antiquity of Anthropoidea.” In Anthropoid Origins: The Fossil Evidence, edited by John G. Fleagle and Richard F. Kay, 55–98. New York: Plenum Press.
Beard, K. Christopher, Laurent Marivaux, Soe Thura Tun, Aung Naing Soe, Yaowalak Chaimanee, Wanna Htoon, Bernard Marandat, Htun Htun Aung, and Jean-Jacques Jaeger. 2007. “New Sivaladapid Primates from the Eocene Pondaung Formation of Myanmar and the Anthropoid Status of Amphipithecidae.” Bulletin of Carnegie Museum of Natural History 39: 67–76.
Beard, K. Christopher, Tao Qi, Mary R. Dawson, Banyue Wang, and Chuankuei Li. 1994. “A Diverse New Primate Fauna from Middle Eocene Fissure-Fillings in Southeastern China.” Nature 368 (6472): 604–609.
Beard, K. Christopher, Yongsheng Tong, Mary R. Dawson, Jingwen Wang, and Xueshi Huang. 1996. “Earliest Complete Dentition of an Anthropoid Primate from the Late Middle Eocene of Shanxi Province, China.” Science 272 (5258): 82–85.
Beecher, Robert M. 1983. “Evolution of the Mandibular Symphysis in Notharctinae (Adapidae, Primates).” International Journal of Primatology 4 (1): 99–112.
Begun, David R. 2002. “European Hominoids.” In The Primate Fossil Record, edited by William C. Hartwig, 339–368. Cambridge: Cambridge University Press.
Begun, David R. 2003. “Planet of the Apes.” Scientific American 289 (2): 74–83.
Begun, David R. 2007. “Fossil Record of Miocene Hominoids.” In Handbook of Paleoanthropology, edited by Winfried Henke and Ian Tattersall, 921–977. New York: Springer.
Benefit, Brenda R., and Monte L. McCrossin. 1997. “Earliest Known Old World Monkey Skull.” Nature 388 (6640): 368–371.
Benefit, Brenda R., and Monte L. McCrossin. 2015. “A Window into Ape Evolution.” Science 350 (6260): 515–516.
Bloch, Jonathan I., and David M. Boyer. 2002. “Grasping Primate Origins.” Science 298 (5598): 1606–1610.
Bloch, Jonathan I., and David M. Boyer. 2007. “New Skeletons of Paleocene-Eocene Plesiadapiformes: A Diversity of Arboreal Positional Behaviors in Early Primates.” In Primate Origins: Adaptations and Evolution, edited by Matthew J. Ravosa and Marian Dagosto, 535–581. New York: Springer.
Bloch, Jonathan I., and Mary T. Silcox. 2006. “Cranial Anatomy of the Paleocene Plesiadapiform Carpolestes simpsoni (Mammalia, Primates) Using Ultra High-Resolution X-ray Computed Tomography, and the Relationships of Plesiadapiforms to Euprimates.” Journal of Human Evolution: 50 (1): 1–35.
Blue, Kathleen T., Monte L. McCrossin, and Brenda R. Benefit. 2006. “Terrestriality in a Middle Miocene Context: Victoriapithecus from Maboko, Kenya.” In Human Origins and Environmental Backgrounds, edited by Hidemi Ishida, Russell Tuttle, Martin Pickford, Naomichi Ogihara, and Masato Nakatsukasa, 45–58. New York: Springer.
Bocherens, Hervé, Friedemann Schrenk, Yaowalak Chaimanee, Ottmar Kullmer, Doris Mörike, Diana Pushkina, and Jean-Jacques Jaeger. 2017. “Flexibility of Diet and Habitat in Pleistocene South Asian Mammals: Implications for the Fate of the Giant Fossil Ape Gigantopithecus.” Quaternary International 434 (A): 148–155.
Bond, Mariano, Marcelo F. Tejedor, Kenneth E. Campbell Jr., Laura Chornogubsky, Nelson Novo, and Francisco Goin. 2015. “Eocene Primates of South America and the African Origins of New World Monkeys.” Nature 520 (7548): 539–541.
Bown, T. M., and M. J. Kraus. 1988. “Geology and Paleoenvironment of the Oligocene Jebel Qatrani Formation and Adjacent Rocks, Fayum Depression, Egypt.” Professional Paper, 1452. Washington, DC: U.S. Geological Survey Professional Papers.
Cachel, Susan. 2015. Fossil Primates. Vol. 69. Cambridge: Cambridge University Press.
Cameron, David W. 1997. “A Revised Systematic Scheme for the Eurasian Miocene Fossil Hominidae.” Journal of Human Evolution 33 (4): 449–477.
Cartmill, Matt. 1972. “Arboreal Adaptations and the Origin of the Order Primates.” In The Functional and Evolutionary Biology of Primates, edited by Russell Tuttle, 97–122. Chicago: Aldine-Atherton.
Cartmill, Matt. 1974. “Rethinking Primate Origins.” Science 184 (4135): 436–443.
Cartmill, Matt, and Richard F. Kay. 1978. “Craniodental Morphology, Tarsier Affinities, and Primate Suborders.” In Recent Advances in Primatology: Evolution, edited by D. J. Chivers and K. A. Joysey, 205–214. London: Academic Press.
Casanovas-Vilar, Isaac, David M. Alba, Miguel Garcés, Josep M. Robles, and Salvador Moyà-Solà. 2011. “Updated Chronology for the Miocene Hominoid Radiation in Western Eurasia.” Proceedings of the National Academy of Sciences 108 (14): 5554-5559. https://doi:10.1073/pnas.1018562108.
Chaimanee, Yaowalak, Olivier Chavasseau, K. Christopher Beard, Aung Aung Kyaw, Aung Naing Soe, Chit Sein, Vincent Lazzari, et al. 2012. “Late Middle Eocene Primate from Myanmar and the Initial Anthropoid Colonization of Africa.” Proceedings of the National Academy of Sciences of the United States of America 109 (26): 10293–10297.
Chester, Stephen G. B., Jonathan I. Bloch, Doug M. Boyer, and William A. Clemens. 2015. “Oldest Known Euarchontan Tarsals and Affinities of Paleocene Purgatorius to Primates.” Proceedings of the National Academy of Sciences of the United States of America 112 (5): 1487–1492.
Ciochon, Russell L., and Gregg F. Gunnell. 2002. “Chronology of Primate Discoveries in Myanmar: Influences on the Anthropoid Origins Debate.” Yearbook of Physical Anthropology 45(S35): 2–35.
Ciochon, R. L., D. R. Piperno, and R. G. Thompson. 1990. “Opal Phytoliths Found on the Teeth of the Extinct Ape Gigantopithecus blacki: Implications for Paleodietary Studies.” Proceedings of the National Academy of Sciences of the United States of America 87 (20): 8120–8124.
Clemens, William A. 2004. “Purgatorius (Plesiadapiformes, Primates?, Mammalia), a Paleocene Immigrant into Northeastern Montana: Stratigraphic Occurrences and Incisor Proportions.” Bulletin of Carnegie Museum of Natural History 36: 3–13.
Cooke, Siobhán B., Justin T. Gladman, Lauren B. Halenar, Zachary S. Klukkert, and Alfred L. Rosenberber. 2016. “The Paleobiology of the Recently Extinct Platyrrhines of Brazil and the Caribbean.” In Molecular Population Genetics, Evolutionary Biology and Biological Conservation of Neotropical Primates, edited by Manuel Ruiz-Garcia and Joseph Mark Shostell, 41–89. New York: Nova Publishers.
DeLeon, Valerie B., Timothy D. Smith, and Alfred L. Rosenberger. 2016. “Ontogeny of the Postorbital Region in Tarsiers and Other Primates.” Anatomical Record: Advances in Integrative Anatomy and Evolutionary Biology 299 (12): 1631–1645.
DeMiguel, Daniel, David M. Alba, and Salvador Moyà-Solà. 2014. “Dietary Specialization during the Evolution of Western Eurasian Hominoids and the Extinction of European Great Apes.” PLoS ONE 9 (5): e97442. https://doi.org/10.1371/journal.pone.0097442.
Dunn, Rachel H., Kenneth D. Rose, Rajendra Rana, Kishore Kumar, Ashok Sahni, and Thierry Smith. 2016. “New Euprimate Postcrania from the Early Eocene of Gujarat, India, and the Strepsirrhine–Haplorhine Divergence.” Journal of Human Evolution 99: 25–51.
Fleagle, John G. 2013. Primate Adaptation and Evolution, Third Edition. San Diego, CA: Academic Press.
Fleagle, John G., and Richard F. Kay. 1994. Anthropoid Origins. New York: Plenum Press.
Franzen, Jens Lorenz, Phillip D. Gingerich, Jörg Habersetzer, Jørn Hurum, von Wighart Koenigswald, and B. Holly Smith. 2009. “Complete Primate Skeleton from the Middle Eocene of Messel in Germany: Morphology and Paleobiology.” PLoS ONE 4 (5): e5723. doi:10.1371/journal.pone.0005723.
Gebo, Daniel L., Marian Dagosto, K. Christopher Beard, Tao Qi, and Jingwen Wang. 2000. “The Oldest Known Anthropoid Postcranial Fossils and the Early Evolution of Higher Primates.” Nature 404 (6775): 276–278.
Gebo, Daniel L., and Elwyn L. Simons. 1987. “Morphology and Locomotor Adaptations of the Foot in Early Oligocene Anthropoids.” American Journal of Physical Anthropology 74 (1): 83–101.
Gilbert, Christopher C., Alejandra Ortiz, Kelsey D. Pugh, Christopher J. Campisano, Biren A. Patel, Ningthoujam Premjit Singh, John G. Fleagle, and Rajeev Patnaik. 2020. “New Middle Miocene Ape (Primates: Hylobatidae) from Ramnagar, India, Fills Major Gaps in the Hominoid Fossil Record.” Proceedings of the Royal Society B 287(1934): 20201655.
Gingerich, P. D. 1980. “Eocene Adapidae, Paleobiogeography, and the Origin of South American Platyrrhini.” In Evolutionary Biology of the New World Monkeys and Continental Drift, edited by Russell L. Ciochon and A. Brunetto Chiarelli, 123–138. New York: Plenum Press.
Godfrey, Laurie R., and William L. Jungers. 2002. “Quaternary Fossil Lemurs.” In The Primate Fossil Record, edited by Walter C. Hartwig, 97–121. Cambridge: Cambridge University Press.
Godinot, Marc. 2006. “Lemuriform Origins as Viewed from the Fossil Record.” Folia Primatologica 77 (6): 446–464.
Gregory, William K. 1920. “On the Structure and Relations of Notharctus, an American Eocene Primate.” Memoirs of the American Museum of Natural History (N.S.) 3 (2).
Gunnell, Gregg F., Doug M. Boyer, Anthony R. Friscia, Steven Heritage, Frederik Kyalo Manthi, Ellen R. Miller, Hesham M. Sallam, Nancy B. Simmons, Nancy J. Stevens, and Erik R. Seiffert. 2018. “Fossil Lemurs from Egypt and Kenya Suggest an African Origin for Madagascar’s Aye-aye.” Nature Communications 9 (3193): 1–12.
Habinger, S. G., O. Chavasseau, J. J. Jaeger, Y. Chaimanee, A. N. Soe, C. Sein, and H. Bocherens. 2022. “Evolutionary Ecology of Miocene Hominoid Primates in Southeast Asia.” Scientific Reports 12 (1): 1–12.
Hammond, Ashley, Lorenzo Rook, Alisha D.Anaya, Elisabetta Cioppi, Loïc Costeur, Salvadore Moyà-Solà, and Sergio Almécija. 2020. “Insights into the Lower Torso in Late Miocene Hominoid Oreopithecus bambolii.” Proceedings of the National Academy of Sciences 117 (1): 278–284.
Harrison, Terry. 2010. “Apes among the Tangled Branches of Human Origins.” Science 327 (5965): 532–534.
Harrison, Terry. 2016. “The Fossil Record and Evolutionary History of Hylobatids.” In Evolution of Gibbons and Siamang, edited by Ullrich H. Reichard, Hirohisa Hirai, and Claudia Barelli, 91–110. New York: Springer.
Ibrahim, Yasamin Kh., Lim Tze Tshen, Kira E. Westaway, Earl of Cranbrook, Louise Humphrey, Ross Fatihah Muhammad, Jian-xin Zhao, and Lee Chai Peng. 2013. “First Discovery of Pleistocene Orangutan (Pongo sp.) Fossils in Peninsular Malaysia: Biogeographic and Paleoenvironmental Implications.” Journal of Human Evolution 65 (6): 770–797.
Israfil, Hulya, Sarah M. Zehr, Alan R. Mootnick, Maryellen Ruvolo, and Michael E. Steiper. 2011. “Unresolved Molecular Phylogenies of Gibbons and Siamangs (Family: Hylobatidae) Based on Mitochondrial, Y-linked, and X-linked Loci Indicate a Rapid Miocene Radiation or Sudden Vicariance Event.” Molecular Phylogenetics and Evolution 58 (3): 447–455.
Jablonski, Nina G., and George Chaplin. 2009. “The Fossil Record of Gibbons.” In The Gibbons, edited by Danielle Whittaker and Susan Lappan, 111–130. New York: Springer.
Jones, F. Wood. 1916. Arboreal Man. London: Edward Arnold.
Kay, Richard F. 1977. “Diets of Early Miocene African Hominoids.” Nature 268 (5621): 628–630.
Kay, Richard F. 2015. “Biogeography in Deep Time: What Do Phylogenetics, Geology, and Paleoclimate Tell Us about Early Platyrrhine Evolution?” Molecular Phylogenetics and Evolution 82 (B): 358–374.
Kay, Richard F., and John G. Fleagle. 2010. “Stem Taxa, Homoplasy, Long Lineages, and the Phylogenetic Position of Dolichocebus.” Journal of Human Evolution 59 (2): 218–222.
Kay, Richard F., Jonathan M. G. Perry, Michael Malinzak, Kari L. Allen, E. Christopher Kirk, J. Michael Plavcan, and John G. Fleagle. 2012. “Paleobiology of Santacrucian Primates.” In Early Miocene Paleobiology in Patagonia: High-Latitude Paleocommunities of the Santa Cruz Formation, edited by Sergio F. Vizcaíno, Richard F. Kay, and M. Susana Bargo, 306–330. Cambridge: Cambridge University Press.
Kay, Richard F., Daniel O Schmitt, Christopher J. Vinyard, Jonathan M. G. Perry, Nobuo Shigehara, Masanaru Takai, and Naoko Egi. 2004. “The Paleobiology of Amphipithecidae, South Asian Late Eocene Primates.” Journal of Human Evolution 46 (1): 3–25.
Kay, Richard F., and Elwyn L. Simons. 1980. “The Ecology of Oligocene African Anthropoidea.” International Journal of Primatology 1 (1): 21–37.
Kay, Richard F., Richard W. Thorington, and Peter Houde. 1990. “Eocene Plesiadapiform Shows Affinities with Flying Lemurs Not Primates.” Nature 345 (6273): 342–344.
Kelley, Jay. 2002. “The Hominoid Radiation in Asia.” In The Primate Fossil Record, edited by Walter C. Hartwig, 369–384. Cambridge: Cambridge University Press.
Kirk, E. Christopher, and Elwyn L. Simons. 2001. “Diets of Fossil Primates from the Fayum Depression of Egypt: A Quantitative Analysis of Molar Shearing.” Journal of Human Evolution 40 (3): 203–229.
Kirk, E. Christopher, and Blythe A. Williams. 2011. “New Adapiform Primate of Old World Affinities from the Devil’s Graveyard Formation of Texas.” Journal of Human Evolution 61 (2): 156–168.
Krause, David W. 1991. “Were Paromomyids Gliders? Maybe, Maybe Not.” Journal of Human Evolution 21 (3): 177–188.
Kunimatsu, Yutaka, Masato Nakatsukasa, Yoshihiro Sawada, Tetsuya Sakai, Masayuki Hyodo, Hironobu Hyodo, Tetsumaru Itaya, et al. 2007. “A New Late Miocene Great Ape from Kenya and Its Implications for the Origins of African Great Apes and Humans.” Proceedings of the National Academy of Sciences of the United States of America 104 (49): 19220–19225.
Maclatchy, Laura. 2004. “The Oldest Ape.” Evolutionary Anthropology: Issues, News, and Reviews 13 (3): 90–103.
Marivaux, Laurent, Yaowalak Chaimanee, Stéphane Ducrocq, Bernard Marandat, Jean Sudre, Aung Naing Soe, Soe Thura Tun, Wanna Htoon, and Jean-Jacques Jaeger. 2003. “The Anthropoid Status of a Primate from the Late Middle Eocene Pondaung Formation (Central Myanmar): Tarsal Evidence.” Proceedings of the National Academy of Sciences of the United States of America 100 (23): 13173–13178.
Marivaux, Laurent, Anusha Ramdarshan, El Mabrouk Essid, Wissem Marzougui, Hayet Khayati Ammar, Renaud Lebrun, Bernard Marandat, Gilles Merzeraud, Rodolphe Tabuce, and Monique Vianey-Liaud. 2013. “Djebelemur, a Tiny Pre-ToothCombed Primate from the Eocene of Tunisia: A Glimpse into the Origin of Crown Strepsirrhines.” PLoS ONE 8 (12): e80778. doi.org/10.1371/journal.pone.0080778.
Martin, R. D. 1968. “Towards a New Definition of Primates.” Man (N.S.) 3 (3): 377–401.
Martin, R. D. 1972. “Adaptive Radiation and Behaviour of the Malagasy Primates.” Philosophical Transactions of the Royal Society B: Biological Sciences 264 (862): 295–352.
Martin, R. D. 1990. Primate Origins and Evolution, a Phylogenetic Reconstruction. Princeton: Princeton University Press.
McBrearty, Sally, and Nina G. Jablonski. 2005. “First Fossil Chimpanzee.” Nature 437 (7055): 105–108.
Michel, Lauren A., Daniel J. Peppe, James A. Lutz, Stephen G. Driese, Holly M. Dunsworth, William E. H. Harcourt-Smith, William H. Horner, Thomas Lehmann, Sheila Nightingale, and Kieran P. McNulty. 2014. “Remnants of an Ancient Forest Provide Ecological Context for Early Miocene Fossil Apes.” Nature Communications 5: 1-9.
Miller, E. R., B. R. Benefit, M. L. McCrossin, J. M. Plavcan, M. G. Leakey, A. N. El-Barkooky, M. A. Hamdan, M. K. A. Gawad, S. M. Hassan, and E. L. Simons. 2009. “Systematics of Early and Middle Miocene Old World Monkeys.” Journal of Human Evolution 57 (3): 195–211.
Mocke, H., M. Pickford, B. Senut, and D. Gommery. 2022. “New Information about African Late Middle Miocene to Latest Miocene (13–5.5 Ma) Hominoidea. Communications of the Geological Survey of Namibia 24: 33–66.
Moyà-Solà, Salvadore, David M. Alba, Sergio Almécija, Isaac Casanovas-Vilar, Meike Köhler, Soledad De Esteban-Trivigno, Josep M. Robles, Jordi Galindo, and Josep Fortuny. 2009. “A Unique Middle Miocene European Hominoid and the Origins of the Great Ape and Human Clade.” Proceedings of the National Academy of Sciences of the United States of America 106 (24): 9601–9606.
Moyà-Solà, Salvador, Meike Köhler, David M. Alba, Isaac Casanovas-Vilar, and Jordi Galindo. 2004. “Pierolapithecus catalaunicus, a New Middle Miocene Great Ape from Spain.” Science 306 (5700): 1339–1344.
Ni, Xijun, Daniel L. Gebo, Marian Dagosto, Jin Meng, Paul Tafforeau, John J. Flynn, and K. Christopher Beard. 2013. “The Oldest Known Primate Skeleton and Early Haplorhine Evolution.” Nature 498 (7452): 60–64.
Perry, Jonathan M. G., Richard F. Kay, Sergio F. Vizcaíno, and M. Susana Bargo. 2010. “Tooth Root Size, Chewing Muscle Leverage, and the Biology of Homunculus patagonicus (Primates) from the Late Early Miocene of Patagonia.” Ameghiniana 47 (3): 355–371.
Perry, Jonathan M. G., Richard F. Kay, Sergio F. Vizcaíno, and M. Susana Bargo. 2014. “Oldest Known Cranium of a Juvenile New World Monkey (Early Miocene, Patagonia, Argentina): Implications for the Taxonomy and the Molar Eruption Pattern of Early Platyrrhines.” Journal of Human Evolution 74: 67–81.
Pickford, Martin, Yves Coppens, Brigitte Senut, Jorge Morales, and José Braga. 2009. “Late Miocene Hominoid from Niger.” Comptes Rendus Palevol 8 (4): 413–425.
Pilbeam, David. 1982. “New Hominoid Skull Material from the Miocene of Pakistan.” Nature 295 (5846): 232–234.
Pilbeam, David, Michael D. Rose, John C. Barry, and S. M. Ibrahim Shah. 1990. “New Sivapithecus Humeri from Pakistan and the Relationship of Sivapithecus and Pongo.” Nature 348 (6298): 237–239.
Rasmussen, D. Tab. 1990. “Primate Origins: Lessons from a Neotropical Marsupial.” American Journal of Primatology 22 (4): 263–277.
Ravosa, Matthew J. 1996. “Mandibular Form and Function in North American and European Adapidae and Omomyidae.” Journal of Morphology 229 (2): 171–190.
Rögl, Fred. 1999. “Mediterranean and Paratethys Palaeogeography during the Oligocene and Miocene.” In Hominoid Evolution and Climatic Change in Europe, edited by Jorge Agustí, Lorenzo Rook, and Peter Andrews, 8–22. Cambridge: Cambridge University Press.
Rosas, A., A. García-Tabernero, D. Fidalgo, M. Fero Meñe, C. Ebana Ebana, F. Esono Mba, and P. Saladie. 2022. “The Scarcity of Fossils in the African Rainforest: Archaeo-Paleontological Surveys and Actualistic Taphonomy in Equatorial Guinea.” Historical Biology 34 (8): 1–9.
Rose, Kenneth D., and Thomas M. Bown. 1984. “Gradual Phyletic Evolution at the Generic Level in Early Eocene Omomyoid Primates.” Nature 309 (5965): 250–252.
Rose, Kenneth D., Rachel H. Dunn, Kishor Kumar, Jonathan M. G. Perry, Kristen A. Prufrock, Rajendra S. Rana, and Thierry Smith. 2018. “New Fossils from Tadkeshwar Mine (Gujarat, India) Increase Primate Diversity from the Early Eocene Cambay Shale.” Journal of Human Evolution 122: 93–107.
Rose, Kenneth D., and John M. Rensberger. 1983. “Upper Dentition of Ekgmowechashala (Omomyoid Primate) from the John Day Formation, Oligo-Miocene of Oregon.” Folia Primatologica 41(1-2): 102–111.
Rosenberger, Alfred L. 2010. “Platyrrhines, PAUP, Parallelism, and the Long Lineage Hypothesis: A Reply to Kay et al. (2008).” Journal of Human Evolution 59 (2): 214–217.
Ross, Callum F. 2000. “Into the Light: The Origins of Anthropoidea.” Annual Review of Anthropology 29: 147–194.
Ross, Callum F., and Richard F. Kay, eds. 2004. Anthropoid Origins: New Visions. New York: Kluwer Academic/Plenum Publishers.
Russo, Gabrielle A. 2016. “Comparative Sacral Morphology and the Reconstructed Tail Lengths of Five Extinct Primates: Proconsul heseloni, Epipliopithecus vindobonensis, Archaeolemur edwardsi, Megaladapis grandidieri, and Palaeopropithecus kelyus.” Journal of Human Evolution 90: 135–162.
Schmid, Peter. 1979. “Evidence of Microchoerine Evolution from Dielsdorf (Zürich Region, Switzerland): A Preliminary Report.” Folia Primatologica 31 (4): 301–311.
Seiffert, Erik R. 2012. “Early Primate Evolution in Afro-Arabia.” Evolutionary Anthropology: Issues, News, and Reviews 21 (6): 239–253.
Seiffert, Erik R., Jonathan M. G. Perry, Elwyn L. Simons, and Doug M. Boyer. 2009. “Convergent Evolution of Anthropoid-like Adaptations in Eocene Adapiform Primates.” Nature 461 (7267): 1118–1121.
Seiffert, Erik R., Elwyn L. Simons, and Yousry Attia. 2003. “Fossil Evidence for an Ancient Divergence of Lorises and Galagos.” Nature 422 (6930): 421–424.
Seiffert, Erik R., Elwyn L. Simons, Doug M. Boyer, Jonathan M. G. Perry, Timothy M. Ryan, and Hesham M. Sallam. 2010. “A Fossil Primate of Uncertain Affinities from the Earliest Late Eocene of Egypt.” Proceedings of the National Academy of Sciences of the United States of America 107 (21): 9712–9717.
Seiffert, Erik R., Elwyn L. Simons, and Cornelia V. M. Simons. 2004. “Phylogenetic, Biogeographic, and Adaptive Implications of New Fossil Evidence Bearing on Crown Anthropoid Origins and Early Stem Catarrhine Evolution.” In Anthropoid Origins: New Visions, edited by Callum F. Ross and Richard F. Kay, 157–182. New York: Kluwer/Plenum Publishing.
Simons, Elwyn L. 1961. “The Phyletic Position of Ramapithecus.” Postilla 57: 1–9.
Simons, Elwyn L. 2001. “The Cranium of Parapithecus grangeri, an Egyptian Oligocene Anthropoidean Primate.” Proceedings of the National Academy of Sciences of the United States of America 98 (4): 7892–7897.
Simons, Elwyn L. 2004. “The Cranium and Adaptations of Parapithecus grangeri, a Stem Anthropoid From the Fayum Oligocene of Egypt.” In Anthropoid Origins: New Visions, edited by Callum F. Ross and Richard F. Kay, 183–204. New York: Kluwer/Plenum Publishing.
Simons, Elwyn L. 2008. “Eocene and Oligocene Mammals of the Fayum, Egypt.” In Elwyn Simons: A Search for Origins, edited by John G. Fleagle and Christopher C. Gilbert, 87–105. New York: Springer.
Simons, Elwyn L., and D. Tab Rasmussen. 1994a. “A Remarkable Cranium of Plesiopithecus teras (Primates, Prosimii) from the Eocene of Egypt.” Proceedings of the National Academy of Sciences of the United States of America 91(21): 9946–9950.
Simons, Elwyn L., and D. Tab Rasmussen. 1994b. “A Whole New World of Ancestors: Eocene Anthropoideans from Africa.” Evolutionary Anthropology 3 (4): 128–139.
Simons, Elwyn L., and D. Tab Rasmussen. 1996. “Skull of Catopithecus browni, an Early Tertiary Catarrhine.” American Journal of Physical Anthropology 100 (2): 261–292.
Simons, Elwyn L., and Erik R. Seiffert. 1999. “A Partial Skeleton of Proteopithecus sylviae (Primates Anthropoidea): First Associated Dental and Postcranial Remains of an Eocene Anthropoidean.” Comptes Rendus de l'Académie des Sciences, Paris 329 (12): 921–927.
Simons, Elwyn L., Erik R. Seiffert, Timothy M. Ryan, and Yousry Attia. 2007. “A Remarkable Female Cranium of the Early Oligocene Anthropoid Aegyptopithecus zeuxis (Catarrhini, Propliopithecidae).” Proceedings of the National Academy of Sciences of the United States of America 104 (21): 8731–8736.
Simpson, George Gaylord. 1933. “The ‘Plagiaulacoid’ Type of Mammalian Dentition: A Study of Convergence.” Journal of Mammalogy 14 (2): 97–107.
Simpson, George Gaylord. 1940. “Review of the Mammal-Bearing Tertiary of South America.” Proceedings of the American Philosophical Society 83 (5): 649–709.
Simpson, George Gaylord. 1967. “The Tertiary Lorisiform Primates of Africa.” Bulletin of the Museum of Comparative Zoology at Harvard University 136: 39–62.
Smith, G. Elliot. 1912. “The Evolution of Man.” Smithsonian Institute Annual Report 2012: 553–572.
Smith, Thierry, Kenneth D. Rose, and Philip D. Gingerich. 2006. “Rapid Asia–Europe–North America Geographic Dispersal of Earliest Eocene Primate Teilhardina during the Paleocene–Eocene Thermal Maximum.” Proceedings of the National Academy of Sciences of the United States of America 103 (30): 11223–11227.
Stehlin, Hans G. 1912. “Die säugetiere des schweizerischen Eocaens. Siebenter teil, erst hälfte: Adapis” [“The Mammals of the Swiss Eocene. Part Seven, First Half: Adapis”]. Abhandlungen der Schweizerischen Paläontologischen Gesellschaft 38: 1165–1298.
Strait, Suzanne G. 2001. “Dietary Reconstruction of Small-Bodied Omomyoid Primates.” Journal of Vertebrate Paleontology 21 (2): 322–334.
Sussman, Robert W. 1991. “Primate Origins and the Evolution of Angiosperms.” American Journal of Primatology 23 (4): 209–223.
Suwa, Gen, Reiko T. Kono, Shigehiro Katoh, Berhane Asfaw, and Yonas Beyene. 2007. “A New Species of Great Ape from the Late Miocene Epoch in Ethiopia.” Nature 448 (7156): 921–924.
Teaford, Mark F., Mary C. Maas, and Elwyn L. Simons. 1996. “Dental Microwear and Microstructure in Early Oligocene Primates from the Fayum, Egypt: Implications for Diet.” American Journal of Physical Anthropology 101 (4): 527–543.
Ungar, Peter S., and Richard F. Kay. 1995. “The Dietary Adaptations of European Miocene Catarrhines.” Proceedings of the National Academy of Sciences of the United States of America 92 (12): 5479–5481.
Wang, Cui-Bin, Ling-Xia Zhao, Chang-Zhu Jin, Yuan Wang, Da-Gong Qin, and Wen-Shi Pan. 2014. “New Discovery of Early Pleistocene Orangutan Fossils from Sanhe Cave in Chongzuo, Guangxi, Southern China.” Quaternary International 354: 68–74.
Ward, C. V., A. Walker, and M. F. Teaford. 1991. “Proconsul Did Not Have a Tail.” Journal of Human Evolution 21 (3): 215–220.
Wheeler, Brandon C. 2010. “Community Ecology of the Middle Miocene Primates of La Venta, Colombia: The Relationship between Ecological Diversity, Divergence Time, and Phylogenetic Richness.” Primates 51 (2): 131–138.
Williams, Blythe A., and Richard F. Kay. 1995. “The Taxon Anthropoidea and the Crown Clade Concept.” Evolutionary Anthropology 3 (6): 188–190.
Williams, Blythe A., Richard F. Kay, and E. Christopher Kirk. 2010a. “New Perspectives on Anthropoid Origins.” Proceedings of the National Academy of the United States of America 107 (11): 4797–4804.
Williams, Blythe A., Richard F. Kay, E. Christopher Kirk, and Callum F. Ross. 2010b. “Darwinius masillae Is a European Middle Eocene Stem Strepsirrhine—A Reply to Franzen et al.” Journal of Human Evolution 59(5): 567–573.
Wilson Mantilla, G. P., S. G. B. Chester, W. A. Clemens, J. R. Moore, C. J. Sprain, B. T. Hovatter, W. S. Mitchell, W. W. Mans, R. Mundil, and P. R. Renne. 2021. “Earliest Palaeocene Purgatoriids and the Initial Radiation of Stem Primates.” Royal Society Open Science 8(2):210050. doi:10.1098/rsos.210050.
Acknowledgments
We are immensely grateful to the editors of this book, Drs. Beth Shook, Lara Braff, Katie Nelson, and Kelsie Aguilera, for their time and commitment to making this knowledge freely accessible to all, and for giving us the opportunity to participate in this important project.
Bonnie Yoshida-Levine Ph.D., Grossmont College
This chapter is a revision from "Chapter 10: Early Members of the Genus Homo" by Bonnie Yoshida-Levine. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Describe how early Pleistocene climate change influenced the evolution of the genus Homo.
- Identify the characteristics that define the genus Homo.
- Describe the skeletal anatomy of Homo habilis and Homo erectus based on the fossil evidence.
- Assess opposing points of view about how early Homo should be classified.
The boy was no older than nine years when he perished by the swampy shores of the lake. After death, his slender, long-limbed body sank into the mud of the lake shallows. His bones fossilized and lay undisturbed for 1.5 million years. In the 1980s, fossil hunter Kamoya Kimeu, working on the western shore of Lake Turkana, Kenya, glimpsed a dark-colored piece of bone eroding in a hillside. This small skull fragment led to the discovery of what is arguably the world’s most complete early hominin fossil—a youth identified as a member of the species Homo erectus. Now known as Nariokotome Boy, after the nearby lake village, the skeleton has provided a wealth of information about the early evolution of our own genus, Homo (see Figure 11.1). Today, a stone monument with an inscription in three languages—English, Swahili, and the local Turkana language—marks the site of this momentous fossil discovery.
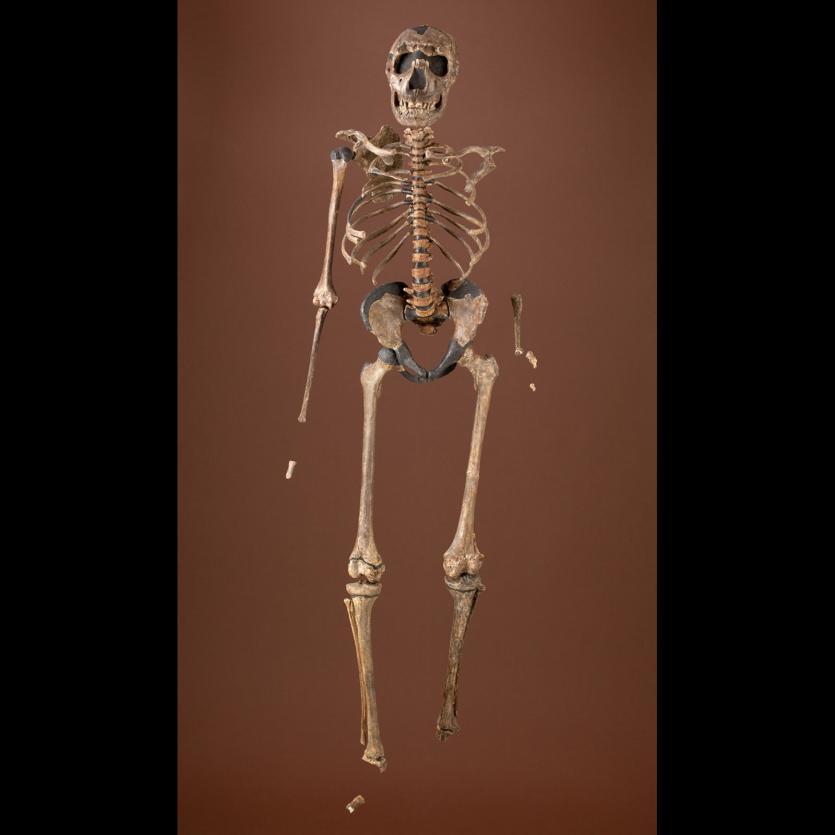

Chapter 9 described our oldest human ancestors, primarily members of the genus Australopithecus, who lived between 2 million and 4 million years ago. This chapter introduces the earliest members of the genus Homo, focusing on Homo habilis and Homo erectus.
Defining the Genus Homo
Because Anthropology is fundamentally concerned with what makes us human, defining our own genus takes on special significance for anthropologists. Ever since scientists acknowledged the existence of extinct species of humans, they have debated which of them display sufficient “humanness” to merit classification in the genus Homo. When grouping species into a common genus, biologists consider criteria such as physical characteristics (morphology), evidence of recent common ancestry, and adaptive strategy (use of the environment). However, there is disagreement about which of those criteria should be prioritized, as well as how specific fossils should be interpreted in light of the criteria.
Nevertheless, there is general agreement that species classified as Homo should share characteristics that are broadly similar within our species. These include the following:
- a relatively large brain size
- a smaller and flatter face
- smaller jaws and teeth
- increased reliance on culture, particularly the use of stone tools, to exploit a greater diversity of environments (adaptive zone).
Some researchers would include larger overall body size and limb proportions (longer legs/shorter arms) in this list. While these criteria seem relatively clear-cut, evaluating them in the fossil record has proved more difficult, particularly for the earliest members of the genus. There are several reasons for this. First, many fossil specimens dating to this time period are incomplete and poorly preserved. Second, early Homo fossils appear quite variable in brain size, facial features, and teeth and body size, and there is not yet consensus about how to best make sense of this diversity. Finally, there is growing evidence that the evolution of the genus Homo proceeded in a mosaic pattern: in other words, these characteristics did not appear all at once in a single species; rather, they were patchily distributed in different species from different regions and time periods. Consequently, different researchers have come up with conflicting classification schemes depending on which criteria they think are most important.
Climate Change and Human Evolution
A key goal in the study of human origins is to learn about the environmental pressures that may have shaped human evolution. As indicated in Chapter 7, scientists use a variety of techniques to reconstruct ancient environments. These include stable isotopes, core samples from oceans and lakes, windblown dust, analysis of geological formations and volcanoes, and fossils of ancient plant and animal communities. Such studies have provided valuable information about the environmental context of early Homo.
The early hominin species covered in Chapter 9, such as Ardipithecus ramidus and Australopithecus afarensis, evolved during the late Pliocene epoch. The Pliocene (5.3 million to 2.6 million years ago) was marked by cooler and drier conditions, with ice caps forming permanently at the poles. Still, Earth’s climate during the Pliocene was considerably warmer and wetter than at present.
The subsequent Pleistocene epoch (2.6 million years to 11,000 years ago) ushered in major environmental change. The Pleistocene is popularly referred to as the Ice Age. Since the term “Ice Age” tends to conjure up images of glaciers and woolly mammoths, one would naturally assume that this was a period of uniformly cold climate around the globe. But this is not actually the case. Instead, climate became much more variable, cycling abruptly between warm/wet (interglacial) and cold/dry (glacial) cycles. These patterns were influenced by changes in Earth’s elliptical orbit around the sun. As is shown in Figure 11.2, each cycle averaged about 41,000 years during the early Pleistocene; the cycles then lengthened to about 100,000 years starting around 1.25 million years ago. Since mountain ranges, wind patterns, ocean currents, and volcanic activity can all influence climate patterns, there were wide-ranging regional and local effects.
Data on ancient geography and climate help us understand how our ancestors moved and migrated to different parts of the world—as well as the constraints under which they operated. When periods of global cooling dominated, sea levels were lower as more water was captured as glacial ice. This exposed continental margins and opened pathways between land masses. During glacial periods, the large Indonesian islands of Sumatra, Java, and Borneo were connected to the Southeast Asian mainland, while New Guinea was part of the southern landmass of greater Australia. There was a land bridge connection between Britain and continental Europe, and an icy, treeless plain known as Beringia connected Northern Asia and Alaska. At the same time, glaciation made some northern areas inaccessible to human habitation. For example, there is evidence that hominin species were in Britain 950,000 years ago, but it does not appear that Britain was continuously occupied during this period. (It is speculated) These early humans may have died out or been forced to abandon the region during glacial periods.
In Africa, paleoclimate research has determined that grasslands (shown in Figure 11.3) expanded and shrank multiple times during this period, even as they expanded over the long term (deMenocal 2014). From studies of fossils, paleontologists have been able to reconstruct Pleistocene animal communities and to consider how they were affected by the changing climate. Among the African animal populations, the number of grazing animal species such as antelope increased. Although the African and Eurasian continents are connected by land, the Sahara desert and the mountainous topography of North Africa serve as natural barriers to crossing. But the fossil record shows that at different times animal species have moved back and forth between Africa and Eurasia. During the early Pleistocene, there is evidence of African mammal species such as baboons, hippos, antelope, and African buffalo migrating out of Africa into Eurasia during periods of aridity (Belmaker 2010).

This changing environment was undoubtedly challenging for our ancestors, but it offered new opportunities to make a living. It is theorized that one solution adopted by some hominins was to specialize in feeding on the new types of plants growing in this landscape. The robust australopithecines (described in Chapter 9) likely developed their large molar teeth with thick enamel in order to exploit this particular dietary niche.
Members of the genus Homo took a different route. Faced with the unstable African climate and shifting landscape, it is proposed that they evolved bigger brains that might have enabled them to rely on cultural solutions such as crafting stone tools that opened up new foraging opportunities. This strategy of behavioral flexibility might have served them well during this unpredictable time and may have led to new innovations such as increased meat-eating, cooperative hunting, and the exploitation of new environments outside Africa.
Homo habilis: The Earliest Members of Our Genus
Homo habilis has traditionally been considered the earliest species placed in the genus Homo. However, as we will see, there is substantial disagreement among paleoanthropologists about the fossils classified as Homo habilis, including whether they come from a single species or multiple, or even whether they should be part of the genus Homo at all.
Homo habilis has a somewhat larger brain size—an average of 650 cubic centimeters (cc)—compared to Australopithecus with less than 500 cc. Additionally, the skull is more rounded and the face less prognathic. However, the postcranial remains show a body size and proportions similar to Australopithecus.
Known dates for fossils identified as Homo habilis range from about 2.5 million years ago to 1.7 million years ago. Recently, a partial lower jaw dated to 2.8 million years from the site of Ledi-Gararu in Ethiopia has been tentatively identified as belonging to the genus Homo (Villmoare et al. 2015). If this classification holds up, it would push the origins of our genus back even further.
Discovery and Naming
The first fossils to be named Homo habilis were discovered at the site of Olduvai Gorge in Tanzania, East Africa, by members of a team led by Louis and Mary Leakey (Figure 11.4). The Leakey family had been conducting fieldwork in the area since the 1930s and had discovered other hominin fossils at the site, such as the robust Paranthropos boisei. The key specimen, a juvenile individual, was actually found by their 20-year-old son Jonathan Leakey. Louis Leakey invited South African paleoanthropologist Philip Tobias and British anatomist John Napier to reconstruct and analyze the remains. The fossil of the juvenile shown in Figure 11.5 (now known as OH-7) consisted of a lower jaw, parts of the parietal bones of the skull, and some hand and finger bones. The fossil was dated by potassium-argon dating to about 1.75 million years. In 1964, the team published their findings in the scientific journal Nature (Leakey et al. 1964). As described in the publication, the new fossils had smaller molar teeth that were less “bulgy” than australopithecine teeth. Although the primary specimen was not yet fully grown, an estimate of its anticipated adult brain size would make it somewhat larger-brained than australopithecines such as Austalopithecus africanus. The hand bones were capable of a precision grip like a human’s hand. This increased the likelihood that stone tools found earlier at Olduvai Gorge were made by this group of hominins. Based on these findings, the authors inferred that it was a new species that should be classified in the genus Homo. They gave it the name Homo habilis, meaning “handy” or “skilled.”
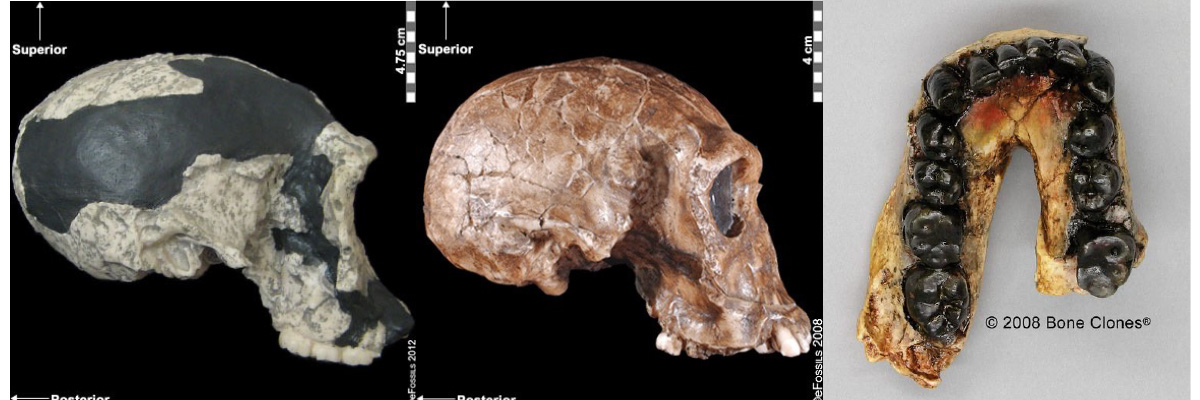
Controversies over the Classification of Homo habilis
Since its initial discovery, many more Homo habilis were discovered in East and South African sites during the 1970s and 1980s (Figure 11.6). As more fossils joined the ranks of Homo habilis, several trends became apparent. First, the fossils were quite variable. While some resembled the fossil specimen first published by Leakey and colleagues, others had larger cranial capacity and tooth size. A well-preserved fossil skull from East Lake Turkana labeled KNM-ER-1470 displayed a larger cranial size along with a strikingly wide face. The diversity of the Homo habilis fossils prompted some scientists to question whether they displayed too much variation to all belong to the same species. They proposed splitting the fossils into at least two groups. The first group resembling the original small-brained specimen would retain the species name Homo habilis; the second group consisting of the larger-brained fossils such as KNM-ER-1470 would be assigned the new name of Homo rudolfensis (see Figure 11.7). Researchers who favored keeping all fossils in Homo habilis argued that sexual dimorphism, adaptation to local environments, or developmental plasticity could be the cause of the differences. For example, modern human body size and body proportions are influenced by variations in climates and nutritional circumstances.
|
Location of Fossils |
Dates |
|
|
Ledi-Gararu, Ethiopia |
2.8 mya |
Partial lower jaw with evidence of both Australopithecus and Homo traits; tentatively considered oldest Early Homo fossil evidence. |
|
Olduvai Gorge, Tanzania |
1.7 mya to 1.8 mya |
Several different specimens classified as Homo habilis, including the type specimen found by Leakey, a relatively complete foot, and a skull with a cranial capacity of about 600 cc. |
|
Koobi Fora, Lake Turkana Basin, Kenya |
1.9 mya |
Several fossils from the Lake Turkana basin show considerable size differences, leading some anthropologists to classify the larger specimen (KNM-ER-1470) as a separate species, Homo rudolfensis. |
|
Sterkfontein and other possible South African cave sites |
about 1.7 mya |
South African caves have yielded fragmentary remains identified as Homo habilis, but secure dates and specifics about the fossils are lacking. |
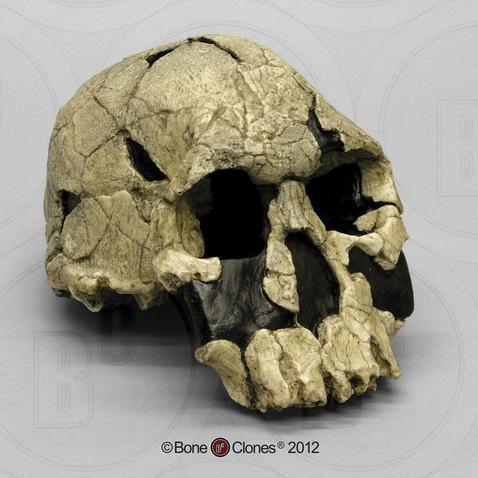
Given the incomplete and fragmentary fossil record from this time period, it is not surprising that classification has proved contentious. As a scholarly consensus has not yet emerged on the classification status of early Homo, this chapter makes use of the single (inclusive) Homo habilis species designation.
There is also disagreement on whether Homo habilis legitimately belongs in the genus Homo. Most of the fossils first classified as Homo habilis were skulls and teeth. When arm, leg, and foot bones were later found, making it possible to estimate body size, the specimens turned out to be quite small in stature with long arms and short legs. Analysis of the relative strength of limb bones suggested that the species, though bipedal, was much more adapted to arboreal climbing than Homo erectus and Homo sapiens (Ruff 2009). This has prompted some scientists to assert that Homo habilis behaved more like an australopithecine—with a shorter gait and the ability to move around in the trees (Wood and Collard 1999). They were also skeptical of the claim that the brain size of Homo habilis was much larger than that of Australopithecus. They have proposed reclassifying some or all of the Homo habilis fossils into the genus Australopithecus, or even placing them into a newly created genus (Wood 2014).
Other scholars have interpreted the fossil evidence differently. A recent reanalysis of Homo habilis/rudolfensis fossils concluded that they sort into the genus Homo rather than Australopithecus (see Hominin Species Summaries at chapter end). In particular, statistical analysis performed indicates that the Homo habilis fossils differ significantly in average cranial capacity from the australopithecines. They also note that some australopithecine species such as the recently discovered Australopithecus sediba have relatively long legs, so body size may not have been as significant as brain- and tooth-size differences (Anton et al. 2014).
Special Topic: Kamoya Kimeu
Kamoya Kimeu (1938–2022) is arguably the most prolific fossil hunter in the history of paleoanthropology (Figure 11.8). In addition to his many decades of work as a field excavator and project supervisor in East Africa, he also trained field workers and scholars and has served as curator for prehistoric sites for the National Museum of Kenya.

Kamoya Kimeu was born in 1938 in rural southeastern Kenya. Despite a formal education that did not go past the sixth grade, he had an aptitude for languages and familiarity with the plants and animals in the East African bush that led him to a job in Tanzania as a field excavator for Louis and Mary Leakey in 1960. In the years that followed, Kimeu found dozens of major hominin fossils. These included a Paranthropus boisei mandible at Olduvai Gorge, Homo habilis specimen KNM-ER-1813 from the Turkana Basin (shown in Figure 11.5), and a key early modern Homo sapiens fossil from the Omo Valley, Ethiopia. Kimeu’s most famous fossil discovery was the skeleton of a young Homo erectus by the Nariokotome river bed in 1984. This finding was highly significant because it was a nearly complete early hominin skeleton and provided insight into child development within this species. In recognition of his work, Kimeu was awarded the National Geographic Society La Gorce Medal by U.S. President Ronald Reagan in 1985.
Traditionally, there has been a divide between African field workers and foreign research scientists, who would typically conduct seasonal field work in Africa, then travel back to their home institutions to publish their findings. Although Kimeu received widespread acclaim for the Nariokotome discovery, as well as a personal acknowledgement in the publication of the find in the journal Nature, he was not credited as an author. More recently, Kimeu’s intellectual contributions to the field of paleoanthropology have been recognized. In 2021, he received an honorary doctorate degree from Case Western Reserve University in Ohio. Kimeu’s most lasting legacy may be his mentorship of countless field workers and students. Today, there are a small but growing number of Black African paleoanthropologists taking on principal roles in the science of human origins.
Homo habilis Culture and Lifeways
Early Stone Tools
It is theorized that the larger brains and smaller teeth of early Homo are linked to a different adaptive strategy than that of earlier hominins: one dependent on modifying rocks to make stone tools and exploit new food sources. As discussed in Chapter 9, the 3.3-million-year-old stone tools from the Lomekwi 3 site in Kenya were made by earlier hominin species than Homo. However, stone tools become more frequent at sites dating to about 2 million years ago, the time of Homo habilis (Roche et al. 2009). This suggests that these hominins were increasingly reliant on stone tools to make a living.
Stone tools are assigned a good deal of importance in the study of human origins. Examining the form of the tools, the raw materials selected, and how they were made and used can provide insight into the thought processes of early humans and how they modified their environment in order to survive. Paleoanthropologists have traditionally classified collections of stone tools into industries, based on their form and mode of manufacture. There is not an exact correspondence between a tool industry and a hominin species; however, some general associations can be made between tool industries and particular hominins, locations, and time periods.
The Oldowan tool industry is named after the site of Olduvai Gorge in Tanzania where the tools were first discovered. The time period of the Oldowan is generally estimated to be 2.5 mya to 1.6 mya. The tools of this industry are described as “flake and chopper” tools—the choppers consisting of stone cobbles with a few flakes struck off them (Figure 11.9). To a casual observer, these tools might not look much different from randomly broken rocks. However, they are harder to make than their crude appearance suggests. The rock selected as the core must be struck by the rock serving as a hammerstone at just the right angle so that one or more flat flakes are removed. This requires selecting rocks that will fracture predictably instead of chunking, as well as the ability to plan ahead and envision the steps needed to create the finished product. The process leaves both the core and the flakes with sharp cutting edges that can be used for a variety of purposes.
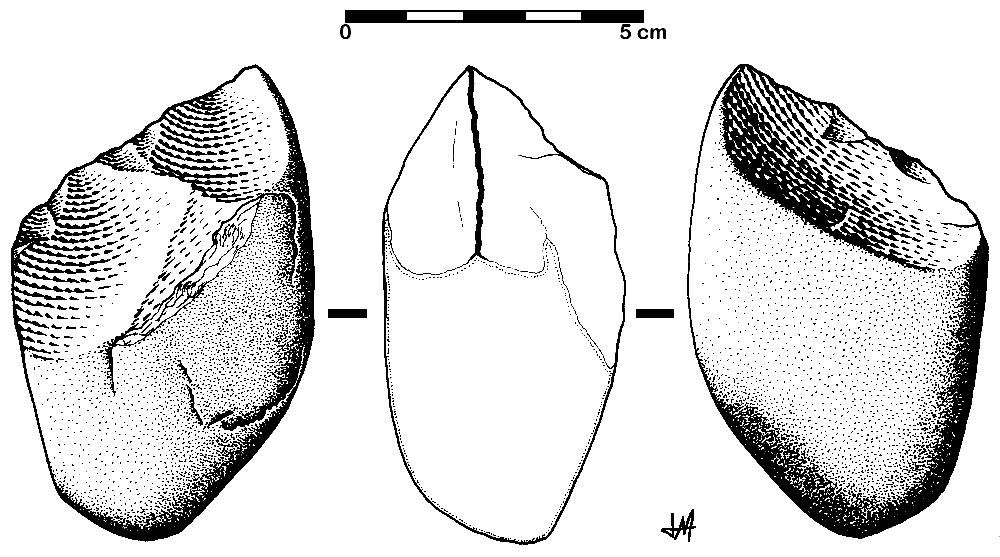
Stone Tool Use and the Diet of Early Homo
What were the hominins doing with the tools? One key activity seems to have been butchering animals. Studies of animal bones at the site show cut marks on bones, and leg bones are often cracked open, suggesting that they were extracting the marrow from the bone cavities. It is interesting to consider whether the hominins hunted these animals or acquired them through other means. The butchered bones come from a variety of African mammals, ranging from small antelope to animals as big as wildebeest and elephants! It is difficult to envision slow, small-bodied Homo habilis with their Oldowan tools bringing down such large animals. One possibility is that the hominins were scavenging carcasses from lions and other large cats. Paleoanthropologist Robert Blumenschine has investigated this hypothesis by observing the behavior of present-day animal carnivores and scavengers on the African savanna. When lions abandon a kill after eating their fill, scavenging animals arrive almost immediately to pick apart the carcass. By the time slow-footed hominins arrived on the scene, the carcass would be mostly stripped of meat. However, if hominins could use stone tools to break into the leg bone cavities, they could get to the marrow, a fatty, calorie-dense source of protein (Blumenschine et al. 1987). Reconstructing activities that happened millions of years ago is obviously a difficult undertaking, and paleoanthropologists continue to debate whether scavenging or hunting was more commonly practiced during this time.
Regardless of how they were acquiring the meat, these activities suggest an important dietary shift from the way that the australopithecines were eating. The Oldowan toolmakers were exploiting a new ecological niche that provided them with more protein and calories. And it was not just limited to meat-eating—stone tool use could have made available numerous other subsistence opportunities. A study of microscopic wear patterns on a sample of Oldowan tools indicates that they were used for processing plant materials such as wood, roots or tubers, and grass seeds and stems (Lemorini et al. 2014). In fact, it has been pointed out that the Oldowan toolmakers’ cutting ability (whether for the purposes of consuming meat and plants or for making tools, shelters, or clothing) represents a new and unique innovation, never seen before in the natural world (Roche et al. 2009).
Homo erectus: Biological and Cultural Innovations
Two million years ago, a new hominin appeared on the scene. Known as Homo erectus, the prevailing scientific view was that this species was much more like us. These hominins were equipped with bigger brains and large bodies with limb proportions similar to our own. Perhaps most importantly, their way of life is now one that is recognizably human, with more advanced tools, hunting, use of fire, and colonizing new environments outside of Africa.
As will be apparent below, new data suggests that the story is not quite as simple. The fossil record for Homo erectus is much more abundant than that of Homo habilis, but it is also more complex and varied—both with regard to the fossils as well as the geographic context in which they are found.
Homo erectus Anatomy
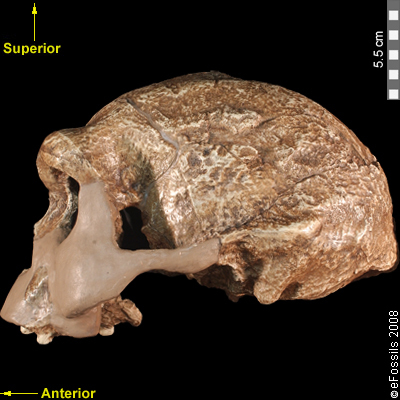
Compared to Homo habilis, Homo erectus showed increased brain size, smaller teeth, and a larger body. However, it also displayed key differences from later hominin species including our own. Although the head of Homo erectus was less ape-like in appearance than the australopithecines, it did not resemble modern humans (Figure 11.10). Compared to Homo habilis, Homo erectus had a larger brain size: an average of about 900 cc compared to 650 cc to 750 cc. Instead of a rounded shape like our skulls, the erectus skull was long and low like a football, with a receding forehead, and a horizontal ridge called an occipital torus that gave the back of the skull a squared-off appearance. The cranial bones are thicker than those of modern humans, and some Homo erectus skulls have a slight thickening along the sagittal suture called a sagittal keel. Large, shelf-like brow ridges hang over the eyes. The face shows less prognathism, and the back teeth are smaller than those of Homo habilis. Instead of a pointed chin, like ours, the mandible of Homo erectus recedes back.
Apart from these features, there is significant variation among Homo erectus fossils from different regions. Scientists have long noted differences between the fossils from Africa and those from Indonesia and China. For example, the Asian fossils tend to have a thicker skull and larger brow ridges than the African specimens, and the sagittal keel described above is more pronounced. Homo erectus fossils from the Republic of Georgia (described in the next section) also display distinctive characteristics. As with Homo habilis, this diversity has prompted a classification debate about whether or not Homo erectus should be split into multiple species. When African Homo erectus is characterized as a separate species, it is called Homo ergaster, while the Asian variant retains the erectus species name because it was discovered first. Here, the species name Homo erectus will be used for both variants.
Homo erectus was thought to have a body size and proportions more similar to modern humans. Unlike Homo habilis and the australopithecines, both of whom were small-statured with long arms and short legs, Homo erectus shows evidence of being fully committed to life on the ground. This meant long, powerfully muscled legs that enabled these hominins to cover more ground efficiently. Indeed, studies of the Homo erectus body form have linked several characteristics of the species to long-distance running in the more open savanna environment (Bramble and Lieberman 2004). Many experts think that hominins around this time had lost much of their body hair, were particularly efficient at sweating, and had darker-pigmented skin—all traits that would support the active lifestyle of such a large-bodied hominin (see Special Topic box, “How We Became Sweaty, Hairless Primates”).
Much of the information about the body form of Homo erectus comes from the Nariokotome fossil of the Homo erectus youth, described at the beginning of the chapter (see Figure 11.1). However, Homo erectus fossils are turning out to be more varied than previously thought. Homo erectus fossils from sites in Africa, as well as from Dmanisi, Georgia, show smaller body sizes than the Nariokotome boy. Even the Nariokotome skeleton itself has been reassessed: some now predict he would have been about 5 feet and 4 inches when fully grown rather than over 6 feet as initially hypothesized, although there is still disagreement about which measurement is more accurate. One explanation for the range of body sizes could be adaptation to a range of different local environments, just as humans today show reduced body size in poor nutritional environments (Anton and Snodgrass 2012).
Homo erectus in Africa
Although the earliest discoveries of Homo erectus fossils were from Asia, the greatest quantity and best-preserved fossils of the species come from East African sites. The earliest fossils in Africa identified as Homo erectus come from the East African site of Koobi Fora, around Lake Turkana in Kenya, and are dated to about 1.8 million years ago. Other fossil remains have been found in East African sites in Kenya, Tanzania, and Ethiopia. Other notable African Homo erectus finds are a female pelvis from the site of Gona, Ethiopia (Simpson et al. 2008), and a cranium with massive brow ridges from Olduvai Gorge known as Olduvai 9, thought to be about 1.4 million years old.
Until recently, Homo erectus’ presence in southern Africa has not been well documented. However, work at the Drimolen cave site in South Africa has yielded new fossils of Paranthropus robustus, and the cranium of a 2–3 year old child tentatively identified as Homo erectus, dated to about 2 million years (Herries et al. 2020). If substantiated, this would be the oldest discovery to date of Homo erectus anywhere.
Regional Discoveries Outside Africa
It is generally agreed that Homo erectus was the first hominin to migrate out of Africa and colonize Asia and later Europe (although recent discoveries in Asia may challenge this view). Key locations and discoveries of Homo erectus fossils, along with the fossils’ estimated ages, are summarized in Figures 11.11 and 11.12.
|
Region
|
Sites
|
Dates
|
Significance of Fossils
|
|
East Africa |
East and West Lake Turkana, Kenya; Olduvai Gorge, Tanzania |
1.8 to 1.4 mya |
Earliest evidence of H. erectus; significant variation in skull and facial features. |
|
South Africa |
Drimolen Cave, South Africa |
2 mya |
Recent find of a 2–3 year old child would be oldest H. erectus anywhere to date. |
|
Western Eurasia |
Dmanisi, Republic of Georgia |
1.75 mya |
Smaller brains and bodies than H. erectus from other regions. |
|
Western Europe |
Atapuerca, Spain (Sima del Elefante and Gran Dolina caves) |
1.2 mya– 400,000 ya |
Partial jaw from Atapuerca is oldest evidence of H. erectus in Western Europe. Fossils from Gran Dolina (dated to about 800,000 years) sometimes referred to as H. antecessor. |
|
Indonesia |
Ngandong, Java; Sangiran, Java |
1.6 mya |
Early dispersal of H. erectus to East Asia; Asian H. erectus features. |
|
China |
Zhoukoudian, China; Loess Plateau (Lantian) |
780,000– 400,000 ya; 2.1 mya |
Large sample of H. erectus fossils and artifacts. Recent evidence of stone tools from Loess Plateau suggests great antiquity of Homo in East Asia. |
Indonesia
The first discovery of Homo erectus was in the late 1800s in Java, Indonesia. A Dutch anatomist named Eugene Dubois searched for human fossils with the belief that since orangutans lived there, it might be a good place to look for remains of early humans. He discovered a portion of a skull, a femur, and other bone fragments on a riverbank. While the femur looked human, the top of the skull was smaller and thicker than that of a modern person. Dubois named the fossil Pithecanthropus erectus (“upright ape-man”), popularized in the media at the time as “Java Man.” After later discoveries of similar fossils in China and Africa, they were combined into a single species (retaining the erectus name) under the genus Homo.
Although Homo erectus has a long history in Indonesia, the region’s geology has complicated the dating of fossils and sites. Fossils from the Sangiran Dome, Java, had previously been estimated to be as old as 1.8 million years, but scientists using new dating methods have arrived at a later date of about 1.3 mya (Matsu’ura et al. 2020). On the recent end of the timeline, a cache of H. erectus fossils from the site of Ngandong in Java has yielded a surprisingly young date of 43,000 years, although a newer study with different dating methods concluded that they were between 117,000 to 108,000 years old (Rizal et al. 2020).
China
There is evidence of Homo erectus in China from several regions and time periods. Homo erectus fossils from northern China, collectively known as “Peking Man,” are some of the most famous human fossils in the world. Dated to about 400,000–700,000 years ago, they were excavated from the site of Zhoukoudian, near the outskirts of Beijing. Hundreds of bones and teeth, including six nearly complete skulls, were excavated from a cave in the 1920s and 1930s. Much of the fossils’ fame comes from the fact that they disappeared under mysterious circumstances. As Japan advanced into China during World War II, Chinese authorities, concerned for the security of the fossils, packed up the boxes and arranged for them to be transported to the United States. But in the chaos of the war, they vanished and were never heard about again. Fortunately, an anatomist named Frans Weidenreich had previously studied the bones and made casts and measurements of the skulls, so this valuable information was not lost. More recent excavations at Longgushan “Dragon Bone Cave” at Zhoukoudian—of tools, living sites, and food remains—have revealed much about the lifestyle of Homo erectus during this time.
Despite this long history of research, China, compared to Africa, was perceived as somewhat peripheral to the study of hominin evolution. Although Homo erectus fossils have been found at several sites in China, with dates that make them comparable to those of Indonesian Homo erectus, none seemed to approximate the antiquity of African sites. The notable finds at sites like Nariokotome and Olorgesaille took center stage during the 1970s and 1980s, as scientists focused on elucidating the species’ anatomy and adaptations in its African homeland. In contrast, fewer research projects were focused on East Asian sites (Dennell and Roebroeks 2005; Qiu 2016).
However, isolated claims of very ancient hominin occupation kept cropping up from different locations in Asia. While some were dismissed because of problems with dating methods or stratigraphic context, the 2018 publication of the discovery of 2.1-million-year-old stone tools from China caught everyone’s attention. Based on paleomagnetic techniques that date the associated soils and windblown dust, these tools indicate that hominins in Asia predated those from the Georgian site of Dmanisi by at least 300,000 years (Zhu et al. 2018). In fact, the tools are older than any Homo erectus fossils anywhere. Since no fossils were found with the tools, it isn’t known which species made them, but it opens up the intriguing possibility that hominins could have migrated out of Africa earlier than Homo erectus. These new discoveries are shaking up previously held views of the East Asian human fossil record.
Western Eurasia
An extraordinary collection of fossils from the site of Dmanisi in the Republic of Georgia has revealed the presence of Homo erectus in Western Eurasia between 1.75 million and 1.86 million years ago. Dmanisi is located in the Caucasus mountains in Georgia. When archaeologists began excavating a medieval settlement near the town in the 1980s and came across the bones of extinct animals, they shifted their focus from the historic to the prehistoric era, but they probably did not anticipate going back quite so far in time. The first hominin fossils were discovered in the early 1990s, and since that time, at least five relatively well-preserved crania have been excavated.
There are several surprising things about the Dmanisi fossils. Compared to African Homo erectus, they have smaller brains and bodies. However, despite the small brain size, they show clear signs of Homo erectus traits such as heavy brow ridges and reduced facial prognathism. Paleoanthropologists have pointed to some aspects of their anatomy (such as the shoulders) that appear rather primitive, although their body proportions seem fully committed to terrestrial bipedalism. One explanation for these differences could be that the Dmanisi hominins represent a very early form of Homo erectus that left Africa before increases in brain and body size evolved in the African population.
Second, although the fossils at this location are from the same geological context, they show a great deal of variation in brain size and in facial features. One skull (Skull 5) has a cranial capacity of only 550 cc, smaller than many Homo habilis fossils, along with larger teeth and a protruding face. Scientists disagree on what these differences mean. Some contend that the Dmanisi fossils cannot all belong to a single species because each one is so different. Others assert that the variability of the Dmanisi fossils proves that they, along with all early Homo fossils, including H. habilis and H.rudolfensis, could all be grouped into Homo erectus (Lordkipanidze et al. 2013). Regardless of which point of view ends up dominating, the Dmanisi hominins are clearly central to the question of how to define the early members of the genus Homo.
Europe
Until recently, there was scant evidence of any Homo erectus presence in Europe, and it was assumed that hominins did not colonize Europe until much later than East Asia or Eurasia. One explanation for this was that the harsh climate of Western Europe served as a barrier to settlement. However, recent fossil finds from Spain suggest that Homo erectus could have made it into Europe over a million years ago. In 2008 a mandible from the Atapuerca region in Spain was discovered, dating to about 1.2 million years ago. A more extensive assemblage of fossils from the site of Gran Dolina in Atapuerca have been dated to about 800,000 years ago. In England in 2013 fossilized hominin footprints of adults and children dated to 950,000 years ago were found at the site of Happisburgh, Norfolk, which would make them the oldest human footprints found outside Africa (Ashton et al. 2014).
At this time, researchers aren’t in agreement as to whether the first Europeans belonged to Homo erectus proper or to a later descendent species. Some scientists refer to the early fossils from Spain by the species name Homo antecessor.
Special Topic: How We Became Hairless, Sweaty Primates
As an anthropology instructor teaching human evolution, my students often ask me about human body hair: When did our ancestors lose it and why? It is assumed that our earliest ancestors were as hairy as modern-day apes. Yet, today, we lack thick hair on most parts of our bodies except in the armpits, pubic regions, and tops of our heads. Humans actually have about the same number of hair follicles per unit of skin as chimpanzees, but, the hairs on most of our body are so thin as to be practically invisible. When did we develop this peculiar pattern of hairlessness? Which selective pressures in our ancestral environment were responsible for this unusual characteristic?
Many experts believe that the driving force behind our loss of body hair was the need to effectively cool ourselves. Along with the lack of hair, humans are also distinguished by being exceptionally sweaty: we sweat larger quantities and more efficiently than any other primate. Humans have a larger amount of eccrine sweat glands than other primates and these glands generate an enormous volume of watery sweat. Sweating produces liquid on the skin that cools the body off as it evaporates. It seems likely that hairlessness and sweating evolved together, as a recent DNA analysis has identified a shared genetic pathway between hair follicles and eccrine sweat gland production (Kamberov et al. 2015).
Which particular environmental conditions led to such adaptations? In this chapter, we learned that the climate was a driving force behind many changes seen in the hominin lineage during the Pleistocene. At that time, the climate was increasingly arid and the forest canopy in parts of Africa was being replaced with a more open grassland environment, resulting in increased sun exposure for our ancestors. Compared to the earlier australopithecines, members of the genus Homo were also developing larger bodies and brains, starting to obtain meat by hunting or scavenging carcasses, and crafting sophisticated stone tools.
According to Nina Jablonski, an expert on the evolution of human skin, the loss of body hair and increased sweating capacity are part of the package of traits characterizing the genus Homo. While larger brains and long-legged bodies made it possible for humans to cover long distances while foraging, this new body form had to cool itself effectively to handle a more active lifestyle. Preventing the brain from overheating was especially critical. The ability to keep cool may have also enabled hominins to forage during the hottest part of the day, giving them an advantage over savanna predators, like lions, that typically rest during this time (Jablonski 2010).
When did these changes occur? Although hair and soft tissue do not typically fossilize, several indirect methods have been used to explore this question. One method tracks a human skin color gene. Since chimpanzees have light skin under their hair, it is probable that early hominins also had light skin color. Apes and other mammals with thick fur coats have protection against the sun’s rays. As our ancestors lost their fur, it is likely that increased melanin pigmentation was selected for as a way to shield our ancestors from harmful ultraviolet radiation. A recent genetic analysis determined that one of the genes responsible for melanin production originated about 1.2 million years ago (Rogers et al 2004).
Another line of evidence tracks the coevolution of a rather unpleasant human companion—the louse. A genetic study identified human body louse as the youngest of the three varieties of lice that infest humans, splitting off as a distinct variety around 70,000 years ago (Kittler et al. 2003). Because human body lice can only spread through clothing, this may have been about the time when humans started to regularly wear clothing. However, the split between human head and pubic lice is estimated to have occurred much earlier, about three million years ago (Bower 2003; Reed et al. 2007). When humans lost much of their body hair, lice that used to roam freely around the body were now confined to two areas: the head and pubic region. As a result of this separation, the lice population split into two distinct groups.
Other explanations have been suggested for the loss of human body hair. For example, being hairless makes it more difficult for skin parasites like lice, fleas, and ticks to live on us. Additionally, after bipedality evolved, hairless bodies would also make reproductive organs and female breasts more visible, suggesting that sexual selection may have played a role.
Homo erectus Lifeways
Tool Technology: Acheulean Tool Industry
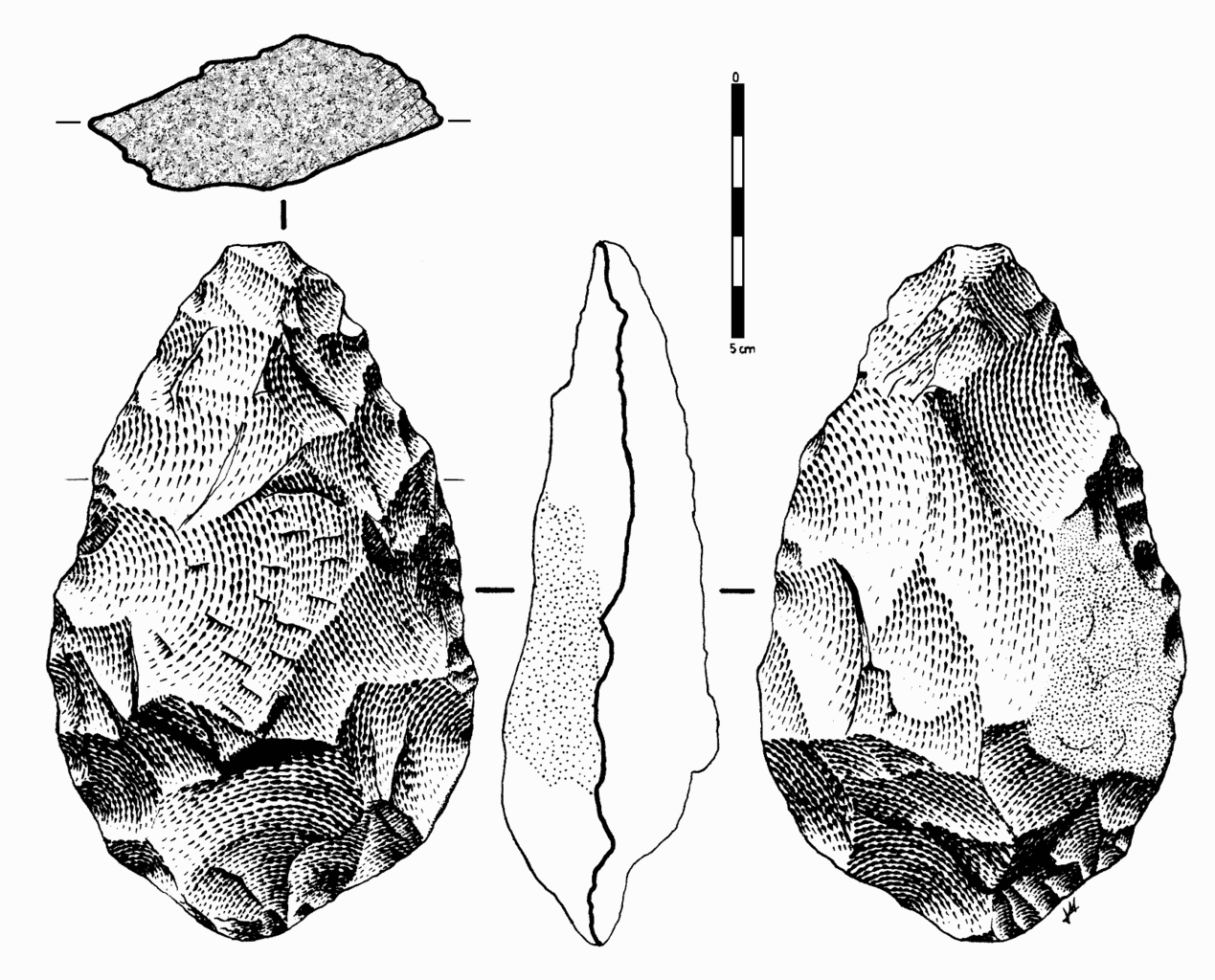
In early African sites associated with Homo erectus, stone tools such as flakes and choppers identified to the Oldowan Industry dominate. Starting at about 1.5 million years ago, some Homo erectus populations began making different forms of tools. These tools—classified together as constituting the Acheulean tool industry—are more complex in form and more consistent in their manufacture. Unlike the Oldowan tools, which were cobbles modified by striking off a few flakes, Acheulean toolmakers carefully shaped both sides of the tool. This type of technique, known as bifacial flaking, requires more planning and skill on the part of the toolmaker; he or she would need to be aware of principles of symmetry when crafting the tool. One of the most common tool forms, the handaxe, is shown in Figure 11.13. As with the tool illustrated below, handaxes tend to be thicker at the base and then come to a rounded point at the tip. Besides handaxes, forms such as scrapers, cleavers, and flake tools are present at Homo erectus sites.
One striking aspect of Acheulean tools is their uniformity. They are more standardized in form and mode of manufacture than the earlier Oldowan tools. For example, the aforementioned handaxes vary in size, but they are remarkably consistent in regard to their shape and proportions. They were also an incredibly stable tool form over time—lasting well over a million years with little change.
Curiously, the Acheulean tools so prominent at African sites are mostly absent in Homo erectus sites in East Asia. Instead, Oldowan-type choppers and scrapers are found at those sites. If this technology seemed to be so important to African Homo erectus, why didn’t East Asian Homo erectus also use the tools? One reason could be environmental differences between the two regions. It has been suggested that Asian Homo erectus populations used perishable material such as bamboo to make tools. Another possibility is that Homo erectus (or even an earlier hominin) migrated to East Asia before the Acheulean technology developed in Africa. The recent discovery of the 2.1-million-year-old tools in China gives credence to this last explanation.
What (if anything) do the Acheulean tools tell us about the mind of Homo erectus? Clearly, they took a fair amount of skill to manufacture. Apart from the actual shaping of the tool, other decisions made by toolmakers can reveal their use of foresight and planning. Did they just pick the most convenient rocks to make their tools, or did they search out a particular raw material that would be ideal for a particular tool? Analysis of Acheulean stone tools suggest that at some sites, the toolmakers selected their raw materials carefully—traveling to particular rock outcrops to quarry stones and perhaps even removing large slabs of rock at the quarries to get at the most desirable material. Such complex activities would require advanced planning and communication with other individuals. However, other Homo erectus sites lack evidence of such selectivity; instead of traveling even a short distance for better raw material, the hominins tended to use what was available in their immediate area (Shipton et al. 2018).
In contrast to Homo erectus tools, the tools of early modern Homo sapiens during the Upper Paleolithic display tremendous diversity across regions and time periods. Additionally, Upper Paleolithic tools and artifacts communicate information such as status and group membership. Such innovation and social signaling seem to have been absent in Homo erectus, suggesting that they had a different relationship with their tools than did Homo sapiens (Coolidge and Wynn 2017). Some scientists assert that these contrasts in tool form and manufacture may signify key cognitive differences between the species, such as the ability to use a complex language.
Subsistence and Diet
In reconstructing the diet of Homo erectus, researchers can draw from multiple lines of evidence. These include stone tools used by Homo erectus, animal bones and occasionally plant remains from Homo erectus sites, and the bones and teeth of the fossils themselves. These data sources suggest that compared to the australopithecines, Homo erectus consumed more animal protein. Coinciding with the appearance of Homo erectus fossils in Africa are archaeological sites with much more abundant stone tools and larger concentrations of butchered animal bones.

It makes sense that a larger body and brain would be correlated with a dietary shift to more calorically dense foods. This is because the brain is a very energetically greedy organ. Indeed, our own human brains require more than 20% of one’s calorie total intake to maintain. When biologists consider the evolution of intelligence in any animal species, it is often framed as a cost/benefit analysis: For large brains to evolve, there has to be a compelling benefit to having them and a way to generate enough energy to fuel them.
One solution that would allow for an increase in human brain size would be a corresponding reduction in the size of the digestive tract (gut). According to the “expensive tissue hypothesis,” initially formulated by Leslie Aiello and Peter Wheeler (1995), a smaller gut would allow for a larger brain without the need for a corresponding increase in the organism’s metabolic rate. More meat in the diet could also fuel the larger brain and body size seen in the genus Homo. Some researchers also believe that body fat percentages increased in hominins (particularly females) around this time, which would have allowed them to be better buffered against environmental disruption such as food shortages (Anton and Snodgrass 2012).
As indicated above, evidence from archaeology and the inferences about Homo erectus body size suggest increased meat eating. How much hunting did Homo erectus engage in compared to the earlier Oldowan toolmakers? Although experts continue to debate the relative importance of hunting versus scavenging, there seems to be stronger evidence of hunting for these hominins. For example, at sites such as Olorgesailie in Kenya (Figure 11.14), there are numerous associations of Acheulean tools with butchered remains of large animals.
However, Homo erectus certainly ate more than just meat. Studies of the tooth surfaces and microscopic wear patterns on hominin teeth indicate that these hominins ate a variety of foods, including some hard, brittle plant foods (Unger and Scott 2009). This would make sense, considering the environment was changing to be more dominated by grasslands in some areas. Roots, bulbs, and tubers (known as underground storage organs) of open savanna plants may have been a primary food source. Indeed, hunter-gatherer groups such as the Hadza of Tanzania rely heavily on such foods, especially during periods when game is scarce. In the unstable environment of the early Pleistocene, dietary versatility would be a definite advantage.
Tool Use, Cooking, and Fire
One key characteristic of the genus Homo is smaller teeth compared to Australopithecus. Why would teeth get smaller? In addition to new types of foods, changes in how food was prepared and consumed likely led to a decrease in tooth size. Think about how you would eat if you didn’t have access to cutting tools. What you couldn’t rip apart with your hands would have to be bitten off with your teeth—actions that would require bigger, more powerful teeth and jaws. As stone tools became increasingly important, researchers propose that hominins began to cut up, tenderize, and process meat and plants, such that they did not have to use their teeth so vigorously.
Cooking food could also have contributed to the reduction in tooth and jaw size. In fact, anthropologist Richard Wrangham (2009) asserts that cooking played a crucial role in human evolution. Cooking provides a head start in the digestive process because of how heat begins to break down food before food even enters the body, and it can help the body extract more nutrients out of meat and plant foods such as starchy tubers.
Obviously cooking requires fire, and the earliest use of fire is a fascinating topic in the study of human evolution. Fire is not only produced by humans; it occurs naturally as a result of lightning strikes. Like other wild animals, early hominins must have been terrified of wildfires, but at some point in time they learned to control fire and put it to good use. Documenting the earliest evidence of fire has been a contentious issue in archaeology because of the difficulty in distinguishing between human-controlled fire and natural burning at hominin sites. Burned areas and ash deposits must have direct associations with human activity to make a case for deliberate fire use. Unfortunately, such evidence is rare at ancient hominin sites, which have been profoundly altered by humans, animals, and geological forces over millions of years. Recently, newer methods—including microscopic analysis of burned rock and bone—have revealed clear evidence of fire use at Koobi Fora, Kenya, dating to 1.5 million years ago (Hlubik et al. 2017).
Migration out of Africa
Homo erectus is generally thought to be the first hominin species to have left Africa. It is hypothesized that they settled in places in Eurasia, such as the Republic of Georgia, Indonesia, and northern China, where fossil evidence of Homo erectus exists. But why would this species have traveled such vast distances to these far-flung regions? To answer this question, we have to consider what we have learned about the biology, culture, and environmental circumstances of Homo erectus. It is proposed that the larger brain and body size of Homo erectus were fueled by a diet consisting of more meat, and their longer, more powerful legs made it possible to walk and run longer distances to acquire food. It has been suggested that since they were eating higher on the food chain, it was necessary for them to extend their home range to find sufficient game. Cultural developments—including better stone tools and new technology such as fire— might have given them greater flexibility in adapting to different environments. Finally, the major Pleistocene climate shift discussed earlier in the chapter certainly played a role. Changes in air temperature, precipitation, access to water sources, and other habitat alteration had far-reaching effects on animal and plant communities; this included Homo erectus. If hominins were relying more on hunting, the migration patterns of their prey could have led them to traverse increasingly long distances.
Life History
The life history of a species refers to its overall pattern of growth, development, and reproduction during its lifetime, with the assumption that these characteristics have been shaped by natural selection. The field of human behavioral ecology, explored in more detail in Appendix C, examines the roots of human behavior and life history. Our species, Homo sapiens, is characterized by a unique life history pattern of slow development, an extended period of juvenile dependence, and a long lifespan. Whereas the offspring of great apes achieve self-sufficiency early, human children are dependent on their parents long after weaning. Additionally, human fathers and grandparents (particularly postmenopausal grandmothers) devote substantial time and energy to caring for their children.

Human behavioral ecologists who study modern hunter-gatherer societies have observed that foraging is no easy business (Figure 11.15). Members of these groups engage in complex foraging techniques that take many years to master. An extended juvenile period gives children the time to acquire these skills. It also allows time for large human brains to grow and mature. On the back end, a longer developmental period results in skilled, successful adults, capable of living a long time (Hill and Kaplan 1999). Despite the time and energy demands, females could have offspring at more closely spaced intervals if they could depend on help from fathers and grandmothers (Hawkes et al. 1998).
What can the study of Homo erectus reveal about its life history pattern? Well-preserved fossils such as the Nariokotome boy can provide some insights. We know that apes such as chimpanzees reach maturity more quickly than humans, and there is some evidence that the australopithecines had a growth rate more akin to that of chimpanzees. Scientists have conducted extensive studies of the Nariokotome skeleton’s bones and teeth to assess growth and development. On the one hand, examination of the long bone ends (epiphyses) of the skeleton suggested that he was an early adolescent with a relatively large body mass, though growth had not yet been completed. On the other hand, study of the dentition, including measurement of microscopic layers of tooth enamel called perikymata, revealed a much younger age of 8 or 9. According to Christopher Dean and Holly Smith (2009), the best explanation for this discrepancy between the dental and skeletal age is that Homo erectus had its own distinct growth pattern—reaching maturity more slowly than chimpanzees but faster than Homo sapiens. This suggests that the human life history pattern of slow maturation and lengthy dependency was a more recent development. More work remains on refining this pattern for early Homo, but it is an important topic that sheds light on how and when we developed our unique life history characteristics.
The Big Picture of Early Homo
We are discovering that the evolution of the genus Homo is more complex than what was previously thought. The earlier view of a simple progression from Australopithecus to Homo habilis to Homo erectus as clearly delineated stages in human evolution just doesn’t hold up anymore.
As is apparent from the information presented here, there is tremendous variability during this time. While fossils classified as Homo habilis show many of the characteristics of the genus Homo, such as brain expansion and smaller tooth size, the small body size and long arms are more akin to australopithecines. There is also tremendous variability within the fossils assigned to Homo habilis, so there is little consensus on whether it is one or multiple species of Homo, a member of the genus Australopithecus, or even a yet-to-be-defined new genus. Similarly, there are considerable differences in skull morphology and body size and form of Homo erectus, of which some specimens show more similarity to Homo habilis than previously thought.
What does this diversity mean for how we should view early Homo? First, there isn’t an abrupt break between Australopithecus and Homo habilis or even between Homo habilis and Homo erectus. Characteristics we define as Homo don’t appear as a unified package; they appear in the fossil record at different times. This is known as mosaic evolution. Indeed, fossil species such as Australopithecus sediba, as well as Homo naledi and Homo floresiensis (who will be introduced in Chapter 11), have displayed unexpected combinations of primitive and derived traits.
We can consider several explanations for the diversity we see within early Homo from about 2.5 million to 1.5 million years ago. One possibility is the existence of multiple contemporaneous species of early Homo during this period. In light of the pattern of environmental instability discussed earlier, it shouldn’t be surprising to see fossils from different parts of Africa and Eurasia display tremendous variability. Multiple hominin forms could also evolve in the same region, as they diversified in order to occupy different ecological niches. However, even the presence of multiple species of hominin does not preclude their interacting and interbreeding with one another. As you’ll see in Appendix D, sequencing of ancient hominin genomes has led to deeper understanding of genetic relationships between extinct species such as the Neanderthals and Denisovans.
Diversity of brain and body sizes could also reflect developmental plasticity—short-term adaptations within a lifetime (Anton et al. 2014). These have the advantage of being more flexible than genetic natural selection, which could only occur over many generations. For example, among human populations today, different body sizes are thought to be adaptations to different climate or nutritional environments. Under Pleistocene conditions of intense variability, a more flexible strategy of adaptation would be valuable.
New discoveries are also questioning old assumptions about the behavior of Homo habilis and Homo erectus. Just as the fossil evidence doesn’t neatly separate Australopithecus and Homo, evidence of the lifeways of early Homo show similar diversity. For example, one of the traditional dividing lines between Homo and Australopithecus was thought to be stone tools: Homo made them; Australopithecus didn’t. However, the recent discovery of stone tools from Kenya dating to 3.3 million years ago challenges this point of view. Similarly, the belief that Homo erectus was the first species to settle outside Africa may now come into question with the report of 2.1-million-year-old stone tools from China. If this find is supported by additional evidence, it may cause a reevaluation of Homo erectus being the first to leave Africa. Instead, there could have been multiple earlier migrations of hominins such as Homo habilis or even Australopithecus species.
These various lines of evidence about the genus Homo point out the need for a more nuanced view of this period of human evolution. Rather than obvious demarcations between species and their corresponding behavioral advancements, it now looks like many behaviors were shared among species. Earlier hominins that we previously didn’t think had the capability could have been doing things like expanding out of Africa or using stone tools. Meanwhile, some other hominins that we had considered more advanced didn’t actually have the full suite of “human” characteristics previously expected.
From a student’s perspective, all this complexity probably seems frustrating. It would be ideal if the human story were a straightforward, sequential narrative. Unfortunately, it seems that human evolution was not a nice, neat trajectory of increasingly humanlike traits and behaviors; rather, it is emblematic of the untidy but exciting nature of the study of human evolution.
Despite some haziness dominating the early Homo narrative, we can identify some overall trends for the million-year period associated with early Homo. These trends include brain expansion, a reduction in facial prognathism, smaller jaw and tooth size, larger body size, and evidence of full terrestrial bipedalism. These traits are associated with a key behavioral shift that emphasizes culture as a flexible strategy to adapt to unpredictable environmental circumstances. Included in this repertoire are the creation and use of stone tools to process meat obtained by scavenging and later hunting, a utilization of fire and cooking, and the roots of the human life history pattern of prolonged childhood, cooperation in child raising, and the practice of skilled foraging techniques. In fact, it’s apparent that the cultural innovations are driving the biological changes, and vice versa, fueling a feedback loop that continues during the later stages of human evolution.
Hominin Species Summaries
|
Hominin |
Homo habilis |
|
Dates |
2.5 million years ago to 1.7 million years ago |
|
Region(s) |
East and South Africa |
|
Famous discoveries |
Olduvai Gorge, Tanzania; Koobi Fora, Kenya; Sterkfontein, South Africa |
|
Brain size |
650 cc average (range from 510 cc to 775 cc) |
|
Dentition |
Smaller teeth with thinner enamel compared to Australopithecus; parabolic dental arcade shape |
|
Cranial features |
Rounder cranium and less facial prognathism than Australopithecus |
|
Postcranial features |
Small stature; similar body plan to Australopithecus |
|
Culture |
Oldowan tools |
|
Other |
N/A |
|
Hominin |
Homo erectus |
|
Dates |
1.8 million years ago to about 110,000 years ago |
|
Region(s) |
East and South Africa; West Eurasia; China and Southeast Asia |
|
Famous discoveries |
Lake Turkana, Olorgesailie, Kenya; Java, Indonesia; Zhoukoudian, China; Dmanisi, Republic of Georgia |
|
Brain size |
Average 900 cc; range between 650 cc and 1,100 cc |
|
Dentition |
Smaller teeth than Homo habilis |
|
Cranial features |
Long, low skull with robust features including thick cranial vault bones and large brow ridge, sagittal keel, and occipital torus |
|
Postcranial features |
Larger body size compared to Homo habilis; body proportions (longer legs and shorter arms) similar to Homo sapiens |
|
Culture |
Acheulean tools (in Africa); evidence of increased hunting and meat-eating; use of fire; migration out of Africa |
|
Other |
N/A |
Review Questions
- Describe the climate during the early Pleistocene. Explain why climate is important for understanding the evolution of early Homo.
- List the key anatomical characteristics that are generally agreed to define the genus Homo.
- Why has classification of early Homo fossils proved difficult? What are some explanations for the variability seen in these fossils?
- Compare and contrast the Oldowan and Acheulean tool industries.
- Name some specific behaviors associated with Homo erectus in the areas of tool use, subsistence practices, migration patterns, and other cultural innovations.
Key Terms
Acheulean: Tool industry characterized by teardrop-shaped stone handaxes flaked on both sides.
Developmental plasticity: The capability of an organism to modify its phenotype during development in response to environmental cues.
Human behavioral ecology: The study of human behavior from an evolutionary and ecological perspective.
Life history: The broad pattern of a species’ life cycle, including development, reproduction, and longevity.
Mosaic evolution: Different characteristics evolve at different rates and appear at different stages.
Occipital torus: A ridge on the occipital bone in the back of the skull.
Oldowan: Earliest stone-tool industry consisting of simple flakes and choppers.
Perikymata: Microscopic ridges on the surface of tooth enamel that serve as markers of tooth development.
Pleistocene: Geological epoch dating from 2.6 million years ago to about 11,000 years ago.
Pliocene: Geological epoch dating from 5.3 to 2.6 million years ago.
Prognathism: Condition where the lower face and jaw protrude forward from a vertical plane.
Sagittal keel: A thickened area along the top of the skull.
FOR FURTHER EXPLORATION
Boaz, Noel Thomas, and Russell L. Ciochon. 2004. Dragon Bone Hill: An Ice-Age Saga of Homo erectus. New York: Oxford University Press.
Human Evolution by the Smithsonian Institution. Produced by the Smithsonian National Museum of Natural History, this website covers many aspects of human evolution including 3-D models of hominin fossils.
Lewin, Roger, and Robert A. Foley. 2004. Principles of Human Evolution. Oxford, UK: Blackwell Publishing.
Mutu, Kari. “Honour Finds Kenya’s Oldest Fossil Hunter Kamoya Kimeu.” The East African, July 19, 2021.
Nordling, Linda. “Raising Up African Paleoanthropologists.” SAPIENS, September 28, 2021. Accessed February 24, 2023. https://www.sapiens.org/biology/african-paleoanthropologists/.
Risen, Clay. “Kamoya Kimeu, Fossil-Hunting ‘Legend’ in East Africa Is Dead.” New York Times, August 11, 2022. Accessed February 24, 2023. https://www.nytimes.com/2022/08/11/science/kamoya-kimeu-dead.html/.
Stoneking, Mark. 2015. “Of Lice and Men: The Molecular Evolution of Human Lice.” Lecture, Center for Academic Research & Training in Anthropogeny, San Diego, California, October 16, 2015. Accessed February 24, 2023. https://carta.anthropogeny.org/events/unique-features-human-skin.
Tarlach, Gemma. 2015. “The First Humans to Know Winter.” Discover, February 26. https://www.discovermagazine.com/planet-earth/the-first-humans-to-know-winter
Ungar, Peter S. 2017. Evolution's Bite: A Story of Teeth, Diet, and Human Origins. Princeton, NJ: Princeton University Press.
References
Aiello, Leslie C., and Peter Wheeler. 1995. “The Expensive-Tissue Hypothesis.” Current Anthropology 36 (2): 199–221.
Anton, Susan C., Richard Potts, and Leslie C. Aiello. 2014. “Evolution of Early Homo: An Integrated Biological Perspective.” Science 345 (6192) doi: 10.1126/science.1236828.
Anton, Susan C., and J. Josh Snodgrass. 2012. “Origins and Evolution of Genus Homo: New Perspectives.” Current Anthropology 53 (S6): S479–S496.
Ashton, Nick, Simon G. Lewis, Isabelle De Groote, Sarah M. Duffy, Martin Bates, Richard Bates, Peter Hoare, et al. 2014. “Hominin Footprints from Early Pleistocene Deposits at Happisburgh, UK.” PLOS ONE 9 (2): e88329.
Belmaker, Miriam. 2010. “Early Pleistocene Faunal Connections between Africa and Eurasia: An Ecological Perspective.” In Out of Africa I: The First Hominin Colonization of Eurasia, edited by John G. Fleagle, John J. Shea, Frederick E. Grine, Andrea L. Baden, and Richard E. Leakey, 183–205. Dordrecht: Springer Netherlands.
Blumenschine, Robert, Henry T. Bunn, Valerius Geist, Fumiko Ikawa-Smith, Curtis W. Marean, Anthony G. Payne, John Tooby, J. Nikolaas, and Van Der Merwe. 1987. “Characteristics of an Early Hominid Scavenging Niche [and Comments and Reply].” Current Anthropology 28 (4): 383–407.
Bower, Bruce. 2004. “Evolution’s Buggy Ride.” Science News 166 (15): 230–230.
Bramble, Dennis M., and Daniel E. Lieberman. 2004. “Endurance Running and the Evolution of Homo.” Nature 432 (7015): 345–352.
Coolidge, Frederick L., and Thomas Grant Wynn. 2017. The Rise of Homo Sapiens: The Evolution of Modern Thinking. New York: Oxford University Press.
Dean, M. Christopher, and B. Holly Smith. 2009. “Growth and Development of the Nariokotome Youth, KNM-WT 15000.” In The First Humans–Origin and Early Evolution of the Genus Homo: Contributions from the Third Stony Brook Human Evolution Symposium and Workshop October 3–7, 2006, edited by Frederick E. Grine, John G. Fleagle, and Richard E. Leakey, 101–120. Dordrecht: Springer Netherlands.
deMenocal, Peter B. 2014. “Climate Shocks.” Scientific American 311 (3): 48–53.
Dennell, Robin, and Wil Roebroeks. 2005. “An Asian Perspective on Early Human Dispersal from Africa.” Nature 438 (7071): 1099–1104.
Hawkes, Kristen, James F. O’Connell, Nicholas G. Blurton Jones, Helen Alvarez, and Eric L. Charnov. 1998. “Grandmothering, Menopause, and the Evolution of Human Life Histories.” Proceedings of the National Academy of Sciences 95 (3): 1336–1339.
Herries, A. I. R., J. M. Martin, A. B. Leece, J. W. Adams, G. Boschian, R. Joannes-Boyau, T. R. Edwards, et al. 2020. "Contemporaneity of Australopithecus, Paranthropus, and early Homo erectus in South Africa." Science 368 (6486). https://doi.org/10.1126/science.aaw7293
Hill, Kim, and Hillard Kaplan. 1999. “Life History Traits in Humans: Theory and Empirical Studies.” Annual Review of Anthropology 28: 397–430.
Hlubik, Sarah, Francesco Berna, Craig Feibel, David Braun, and John W. K. Harris. 2017. “Researching the Nature of Fire at 1.5 Mya on the Site of FxJj20 AB, Koobi Fora, Kenya, Using High-Resolution Spatial Analysis and FTIR Spectrometry.” Current Anthropology 58 (S16): S243–S257.
Jablonski, Nina G. 2010. “The Naked Truth.” Scientific American 302 (2): 42–49.
Kamberov, Yana G., Elinor K. Karlsson, Gerda L. Kamberova, Daniel E. Lieberman, Pardis C. Sabeti, Bruce A. Morgan, and Clifford J. Tabin. 2015. “A Genetic Basis of Variation in Eccrine Sweat Gland and Hair Follicle Density.” Proceedings of the National Academy of Sciences 112 (32): 9932–9937.
Kittler, R., M. Kayser, and M. Stoneking. 2003. "Molecular Evolution of Pediculus Humanus and the Origin of Clothing." Current Biology 13 (16): 1414–1417.
Leakey, Louis S. B., Phillip V. Tobias, and John R. Napier. 1964. “A New Species of Genus Homo from Olduvai Gorge.” Nature 202: 308–312.
Lemorini, Cristina, Thomas W. Plummer, David R. Braun, Alyssa N. Crittenden, Peter W. Ditchfield, Laura C. Bishop, Fritz Hertel, et al. 2014. “Old Stones’ Song: Use-Wear Experiments and Analysis of the Oldowan Quartz and Quartzite Assemblage from Kanjera South (Kenya).” Journal of Human Evolution 72: 10–25.
Lisiecki, Lorraine E., and Maureen E. Raymo. 2005. "A Pliocene-Pleistocene stack of 57 globally distributed benthic δ18O records." Paleoceanography 20 (1)
Lordkipanidze, David, Marcia S. Ponce de León, Ann Margvelashvili, Yoel Rak, G. Philip Rightmire, Abesalom Vekua, and Christoph P. E. Zollikofer. 2013. “A Complete Skull from Dmanisi, Georgia, and the Evolutionary Biology of Early Homo.” Science 342 (6156): 326–333.
Matsu'ura, S., M. Kondo, T. Danhara, S. Sakata, H. Iwano, T. Hirata, I. Kurniawan, et al. 2020. "Age Control of the First Appearance Datum for Javanese Homo erectus in the Sangiran Area." Science 367 (6474): 210–214.
Qiu, Jane. 2016. “How China Is Rewriting the Book on Human Origins.” Nature 535: 22–25.
Reed, David L., Jessica E. Light, Julie M. Allen, and Jeremy J. Kirchman. 2007. "Pair of Lice Lost or Parasites Regained: The Evolutionary History of Anthropoid Primate Lice." BMC Biology 5 (1): 7. doi: 10.1186/1741-7007-5-7.
Rizal, Y., K. E. Westaway, Y. Zaim, G. D. van den Bergh, E. A. Bettis, 3rd, M. J. Morwood, O. F. Huffman, R. Grün, et al. 2020. "Last Appearance of Homo erectus at Ngandong, Java, 117,000–108,000 Years Ago." Nature 577 (7790): 381–385.
Roche, Helene, Robert J. Blumenschine, and John J. Shea. 2009. “Origins and Adaptations of Early Homo: What Archeology Tells Us.” In The First Humans: Origin and Early Evolution of the Genus Homo, edited by Frederick E. Grine, John G. Fleagle, and Richard E. Leakey, 135–147. New York: Springer.
Rogers, Alan R., David Iltis, and Stephen Wooding. 2004. “Genetic Variation at the MC1R l Locus and the Time since Loss of Human Body Hair.” Current Anthropology 45 (1): 105–108.
Ruff, Christopher. 2009. “Relative Limb Strength and Locomotion in Homohabilis.” American Journal of Physical Anthropology 138 (1): 90–100.
Shipton, Ceri, James Blinkhorn, Paul S. Breeze, Patrick Cuthbertson, Nick Drake, Huw S. Groucutt, Richard P. Jennings, et al. 2018. “Acheulean Technology and Landscape Use at Dawadmi, Central Arabia.” PloS one 13 (7): e0200497–e0200497.
Simpson, Scott W., Jay Quade, Naomi E. Levin, Robert Butler, Guillaume Dupont-Nivet, Melanie Everett, and Sileshi Semaw. 2008. “A Female Homoerectus Pelvis from Gona, Ethiopia.” Science 322 (5904): 1089–1092.
Ungar, Peter S., and Robert S. Scott. 2009. “Dental Evidence for Diets of Early Homo.” In The First Humans: Origin and Early Evolution of the Genus Homo, edited by Frederick E. Grine, John G. Fleagle, and Richard E. Leakey, 121–134. New York: Springer.
Villmoare, Brian, William H. Kimbel, Chalachew Seyoum, Christopher J. Campisano, Erin N. DiMaggio, John Rowan, David R. Braun, J. Ramón Arrowsmith, and Kaye E. Reed. 2015. “Early Homo at 2.8 Ma From Ledi-Geraru, Afar, Ethiopia.” Science 347 (6228): 1352–1355.
Wood, Bernard. 2014. “Human Evolution: Fifty Years after Homohabilis.” Nature 508 (7494): 31–33.
Wood, Bernard, and Mark Collard. 1999. “The Changing Face of Genus Homo.” Evolutionary Anthropology 8 (6): 195–207.
Wrangham, Richard. 2009. Catching Fire: How Cooking Made Us Human. New York: Basic Books.
Zhu, Zhaoyu, Robin Dennell, Weiwen Huang, Yi Wu, Shifan Qiu, Shixia Yang, and Zhiguo Rao. 2018. “Hominin Occupation of the Chinese Loess Plateau Since about 2.1 Million Years Ago.” Nature 559: 608–612.
Acknowledgments
The author gratefully acknowledges funding from the California Community Colleges Chancellor’s Office Zero Textbook Cost Degree Grant Program—Implementation Phase 2.

