14 Race and Human Variation
Michael B. C. Rivera, Ph.D., University of Cambridge
This chapter is a revision from “Chapter 13: Race and Human Variation” by Michael B. C. Rivera. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Illustrate the troubling history of “race” concepts.
- Explain human variation and evolution as the thematic roots of biological anthropology as a discipline.
- Critique earlier “race” concepts based on a contemporary understanding of the apportionment of human genetic variation.
- Explain how biological variation in humans is distributed clinally and in accordance with both isolation-by-distance and Out-of-Africa models.
- Identify phenotypic traits that reflect selective and neutral evolution.
- Extend this more-nuanced view of human variation to today’s research, the implications for biomedical studies, applications in forensic anthropology, and other social/cultural/political concerns.
Humans exhibit biological variation. Humans also have a universal desire to categorize other humans in order to make sense of the world around them. Since the birth of the discipline of biological anthropology, we have been interested in studying how humans vary biologically and what the sources of this variation are. Before we tackle these big problems, first consider this question: Why should we study human variation?

There are certainly academic reasons for studying human variation. First, it is highly interesting and important to consider the evolution of our species (see Chapters 9–12) and how our biological variation may be similar to (or different from) that of other species (e.g., other primates and apes; see Chapters 5 and 6). Second, anthropologists study modern human variation to understand how different biological traits developed over evolutionary time (see Figure 14.1). Suppose we are able to grasp the evolutionary processes that produce the differences in biology, physiology, body chemistry, behavior, and culture (human variation). In that case, we can make more accurate inferences about evolution and adaptation among our hominin ancestors, complementing our study of fossil evidence and the archaeological record. Third, as will be discussed in more detail later on, it is important to consider that biological variation among humans has biomedical, forensic, and sociopolitical implications. For these reasons, the study of human variation and evolution has formed the basis of anthropological inquiry for centuries and continues to be a major source of intrigue and inspiration for scientific research conducted today.
An even-more-important role of the biological anthropologist is to improve public understanding of human evolution and variation—outside of academic circles. Terms such as race and ethnicity are used in everyday conversations and in formal settings within and outside academia. The division of humankind into smaller, discrete categories is a regular occurrence in day-to-day life. This can be seen regularly when governments acquire census data with a heading like “geographic origin” or “ethnicity.” Furthermore, such checkboxes and drop-down lists are commonly seen as part of the identifying information required for surveys and job applications.
According to professors of anthropology, ethnic studies, and sociology, race is often understood as rooted in biological differences, ranging from such familiar traits as skin color or eye shape to variations at the genetic level. However, race can also be studied as an “ideological construct” that goes beyond biological and genetic bases (Fuentes et al. 2019), at different times relating to our ethnicities, languages, religious beliefs, and cultural practices. Sometimes people associate racial identity with the concept of socioeconomic status or position, or they link ideas about race to what passport someone has, how long they have been in a country, or how well they have “integrated” into a population.
Some of these ideas about ethnicity have huge social and political impacts, and notions of race have been part of the motivation behind various forms of racism and prejudice today, as well as many wars and genocides throughout history. Racism manifests in many ways—from instances of bullying between kids on school playgrounds to underpaid minorities in the workforce, and from verbal abuse hurled at people of color to violent physical behaviors against those of a certain race. Prejudice can be characterized as negative views toward another group based on some perceived characteristic that makes all members of that group worthy of disdain, disrespect, or exclusion (not solely along racial lines but also according to [dis]ability, gender, sexual orientation, or socioeconomic background, for example). According to Shay-Akil McLean (2014), “Racism is not something particular to the United States and race is not the same everywhere in the world. Racial categories serve particular contextual purposes depending on the society they are used in, but generally follow the base logic of the supremacy of one type of human body over all others (ordering these human bodies in a hierarchical fashion).” Choosing which biological or nonbiological features to use when discussing race is always a social process (Omi and Winant 2014). Race concepts have no validity to them unless people continue to use them in their daily lives—and, in the worst cases, to use them to justify racist behaviors and problematic ideas about racial difference or superiority/inferiority. Recent work in anthropological genetics has revealed the similarities amongst humans on a molecular level and the relatively few differences that exist between populations (Omi and Winant 2014).
The role of the biological anthropologist becomes crucial in the public sphere, because we may be able to debunk myths surrounding human variation and shed light, for the nonanthropologists around us, on how human variation is actually distributed worldwide (see Figure 14.1). Rooted in scientific observations, our work can help nonanthropologists recognize how common ideas about “race” often have no biological or genetic basis. Many anthropologists work hard to educate students on the history of where race concepts come from, why and how they last in public consciousness, and how we become more conscious of racial issues and the need to fight against racism in our societies. Throughout this chapter, I will highlight how humans cannot actually be divided into discrete “races,” because most traits vary on a continuous basis and human biology is, in fact, very homogenous compared to the greater genetic variation we observe in closely related species. Molecular anthropology, or anthropological genetics, continues to add new layers to our understanding of human biological variation and the evolutionary processes that gave rise to the contemporary patterns of human variation. The study of human variation has not always been unbiased, and thinkers and scientists have always worked in their particular sociohistorical context. For this reason, this chapter opens with a brief overview of race concepts throughout history, many of which relied on unethical and unscientific notions about different human groups.
Special Topic: My Experiences as an Asian Academic

My name is Michael, and I am a biological anthropologist and archaeologist (Figure 14.2). What strikes me as most interesting to investigate is human biological variation today and the study of past human evolution. For instance, some of my research on ancient coastal populations has revealed positive effects of coastal living on dietary health and many unique adaptations in bones and teeth when living near rivers and beaches. I love talking to students and nonscientists about bioanthropologists’ work, through teaching, science communication events, and writing book chapters like this one. I grew up in Hong Kong, a city in southern China. My father is from the Philippines and my mother is from Hong Kong, which makes me a mixed Filipino-Chinese academic. Growing up, I noticed that people came in all shapes, sizes, and colors. My life is very different now in that I have gained the expertise to explain those differences, and I feel a great responsibility to guide those new to anthropology toward their own understandings of diversity.
Biological anthropology is not taught extensively in Hong Kong, so I moved to the United Kingdom to earn my bachelor’s, master’s, and doctorate degrees. It was fascinating to me that we could answer important questions about human variation and history using scientific methods. However, I did not have many minority academic role models to look up to while I was at university. My department was made up almost exclusively of white westerner faculty, and it was hard to imagine I could one day get a job at these western institutions. While pursuing my degrees, I also remember several instances of my research contributions being overlooked or dismissed. Sometimes professors and fellow students would make racist comments toward Asian scholars (including me) and other Black, Indigenous and researchers of color, making us greatly uncomfortable in those spaces. When one of us would work up the courage to tell university leaders our experiences of being stereotyped, dismissed, or insulted, we received little support and were further excluded from research and teaching activities. This is a common experience for Black, Indigenous, and other people of color who pursue biological anthropology, and we face the difficult choice between leaving the field or bearing with such unsafe spaces.
It became important to me at that time to find other academics of color with whom to share experiences and form community. I feel inspired by all my colleagues who advocate for greater representation and diversity in universities (whether they are minority academics or not). I admire many of my fellow researchers who are underrepresented and do a great job of representing minority groups through their cutting-edge research and quality teaching at the undergraduate and graduate levels. Although I no longer work in the West, it has remained my great hope that those in the West and the “Global North” will continue to improve university culture, and I support any efforts there to welcome all scholars.
My current work is based in Hong Kong, where I am deeply dedicated to helping develop biological anthropology in East and Southeast Asia and promoting research from our home regions on the international scene. The study of anthropology really highlights how we share a common humanity and history. Being somebody who is “mixed race” and Asian likely played a key role in steering me toward the study of human variation. As this chapter hopefully shows, there is a lot to discuss about race and ethnicity regarding the discipline’s history and current understandings of human diversity. Some scientific and technological advancements today are misused for reasons to do with money, politics, or the continuation of antiquated ideas. It is my belief, alongside many of my friends and fellow anthropologists, that science should be more about empathy toward all members of our species and contributing to the intellectual and technological nourishment of society. After speaking to many members of the public, as well as my own undergraduate students, I have received lovely messages from other individuals of color expressing thanks and appreciation for my presence and understanding as a minority scientist and mentor figure. Anthropology needs much more diversity as well as to make room for those who have traveled different routes into the discipline. All paths taken into anthropology are valid and valuable. I would encourage everyone to study anthropology—it is a field for understanding and celebrating the intricacies of human diversity.
The History of “Race” Concepts
“Race” in the Classical Era

The earliest classification systems used to understand human variation are evidenced by ancient manuscripts, scrolls, and stone tablets recovered through archaeological, historical, and literary research. The Ancient Egyptians had the Book of Gates, dated to the New Kingdom between 1550 B.C.E. and 1077 B.C.E (Figure 14.3). In one part of this tome dedicated to depictions of the underworld, scribes used pictures and hieroglyphics to illustrate a division of Egyptian people into the four categories known to them at the time: the Aamu (Asiatics), the Nehesu (Nubians), the Reth (Egyptians), and the Themehu (Libyans). Though not rooted in any scientific basis like our current understandings of human variation today, the Ancient Egyptians believed that each of these groups were made of a distinctive category of people, distinguishable by their skin color, place of origin, and even behavioral traits.
Another well-known early document is the Bible, where it is written that all humankind descends from one of three sons of Noah: Shem (the ancestor to all olive-skinned Asians), Japheth (the ancestor to pale-skinned Europeans), and Ham (the ancestor to darker-skinned Africans). Similar to the Ancient Egyptians, these distinctions were based on behavioral traits and skin color. More recent work in historiography and linguistics suggest that the branches of “Hamites,” “Japhethites,” and “Shemites” may also relate to the formation of three independent language groups some time between 1000 and 3000 B.C.E. With the continued proliferation of Christianity, this concept of approximately three racial groupings lasted until the Middle Ages and spread as far across Eurasia as crusaders and missionaries ventured at the time.

There is also the “Great Chain of Being,” conceived by ancient Greek philosophers like Plato (427‒348 B.C.E.) and Aristotle (384‒322 B.C.E.). They played a key role in laying the foundations of empirical science, whereby observations of everything from animals to humans were noted with the aim of creating taxonomic categories. Aristotle describes the Great Chain of Being as a ladder along which all objects, plants, animals, humans, and celestial bodies can be mapped in an overall hierarchy (in the order of existential importance, with humans placed near the top, just beneath divine beings; see Figure 14.4). When he writes about humans, Aristotle expressed the belief that certain people are inherently (or genetically) more instinctive rulers, while others are more natural fits for the life of a worker or enslaved person. Based on research by biological anthropologists, we currently recognize that these early systems of classification and hierarchization are unhelpful in studying human biological variation. Both behavioral traits and physical traits are coded for by multiple genes each, and how we exhibit those traits based on our genetics can vary significantly even between individuals of the same population.
“Race” during the Scientific Revolution
The 1400s to 1600s saw the beginnings of the Scientific Revolution in Western societies, with thinkers like Nicholas Copernicus, Galileo Galilei, and Leonardo Da Vinci publishing some of their most important findings. While by no means the first or only scholars globally to use observation and experimentation to understand the world around them, early scientists living at the end of the medieval period in Europe increasingly employed more experimentation, quantification, and rational thought in their work. This is the main difference between the work of the ancient Egyptians, Romans, and Greeks and that of later scientists such as Isaac Newton and Carl Linnaeus in the 1600s and 1700s.

Linnaeus is the author of Systema Naturae (1758), in which he classified all plants and animals under the formalized naming system known as binomial nomenclature (Figure 14.5). This system is typological, in that organisms are placed into groups according to how they are similar or different to others under study. What was most anthropologically notable about Linnaeus’s typological system was that he was one of the first to group humans with apes and monkeys, based on the anatomical similarities between humans and nonhuman primates. However, Linnaeus viewed the world in line with essentialism, a problematic concept that dictates that there are a unique set of characteristics that organisms of a specific kind must have and that would remove organisms from taxonomic categorizations if they lacked any of the required criteria.
Linnaeus subdivided the human species into four varieties, with overtly racist categories based on skin color and “inherent” behaviors. Some European scientists during this period were not aware of their own biases skewing their interpretations of biological variation, while others deliberately worked to shape public perceptions of human variation in ways that established “otherness” and enforced European domination and the subordination of non-European people. The conclusions and claims at which they arrived, consciously or subconsciously, often fit the times they were living through—the so-called Age of Discovery, when the superiority of European cultures over others was a pervasive idea throughout people’s social and political lives. Although much of Eurasia was linked by spice- and silk-trading routes, the European colonial period between the 1400s and 1700s was marked by many new and unfortunately violent encounters overseas (Figure 14.6). When Europeans arrived by ship on the shores of continents that were already inhabited, it was their first meeting with the Indigenous peoples of the Americas and Australasia, who looked, spoke, and behaved differently from peoples with whom they were familiar. Building on the idea of species and “subspecies,” natural historians of this time invented the term race, from the French rasse meaning “local strain.”

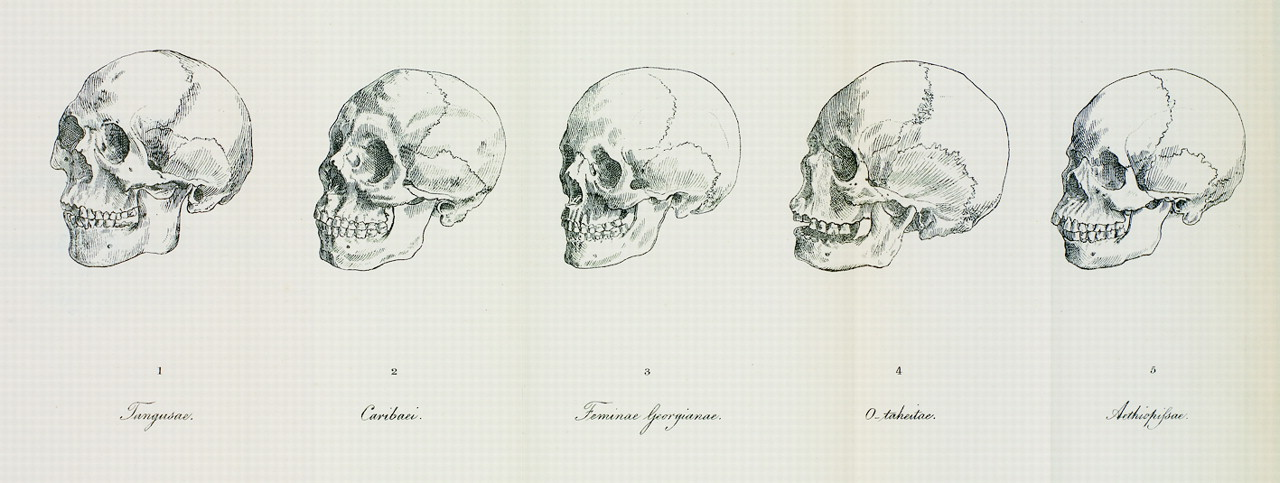
Another scientist of the times, Johann Friedrich Blumenbach (1752‒1840), classified humans into five races based on his observations of cranial form variation as well as skin color. He thus dubbed the “original” form of the human cranium the “Caucasian” form, with the idea that the ideal climate conditions for early humans would have been in the Caucasus region near the Caspian Sea. The key insight Blumenbach presented was that human variation in any particular trait should be more accurately viewed as falling along a gradation (Figure 14.7). While some of his theories were correct according to what we observe today with more knowledge in genetics, they erroneously believed that human “subspecies” were “degenerated” or “transformed” varieties of an ancestral Caucasian or European race. According to them, the Caucasian cranial dimensions were the least changed over human evolutionary time, while the other skull forms represented geographic variants of this “original.” As will be discussed in greater detail later in this chapter, we have genetic and craniometric evidence for sub-Saharan Africa being the origin of the human species instead (see Chapter 12 on the fossil record that places the origins of modern Homo sapiens in north and east Africa). Based on work that shows how most biological characteristics are coded for by nonassociated genes, it is not reasonable to draw links between individuals’ personalities and their skull shapes.
“Race” and the Dawn of Scientific Racism
Between the 1800s and mid-1900s, and contrary to what you might expect, an increased use of scientific methods to justify racial schemes developed in scholarship. Differing from earlier views, which saw all humans as environmentally deviated from one “original” humankind, classification systems after 1800 became more polygenetic (viewing all people as having separate origins) rather than monogenetic (viewing all people as having a single origin). Instead of moving closer to our modern-day understandings of human variation, there was increased support for the notion that each race was created separately and with different attributes (intelligence, temperament, and appearance).
The 1800s were an important precursor to modern biological anthropology as we know it, given that this was when the scientific measurement of human physical features (anthropometry) truly became popularized. However, empirical studies in the 1800s pushed even further the idea that Europeans were culturally and biologically superior to others. While considered one of the pioneers of American “physical” anthropology, Samuel George Morton (1799‒1851) was a scholar who had a large role in perpetuating 1800s scientific racism. By measuring cranial size and shape, he calculated that “Caucasians,” on average, have greater cranial volumes than other groups, such as the Indigenous peoples of the Americas and peoples Morton referred to collectively as “Negros.” Today, we know that cranial size variation depends on such factors as Allen’s and Bergmann’s rules, which give a more likely explanation: in colder environments, it is advantageous for those living there to have larger and rounder heads because they conserve heat more effectively than more slender heads (Beals et al. 1984). The leading figures in craniometry during the 1800s instead were linked heavily with powerful individuals and wealthy sociopolitical institutions and financial bodies. Theories in support of hierarchical racial schemes using “scientific” bases certainly helped continue the exploitative and unethical trafficking and enslavement of Africans between the 1500s and 1800s.
Morton went on to write in his publication Crania Americana (1839) a number of views that fit with a concept called biological determinism. The idea behind biological determinism is that an association exists between people’s physical characteristics and their behavior, intelligence, ability, values, and morals. If the idea is that some groups of people are essentially superior to others in cognitive ability and temperament, then it is easier to justify the unequal treatment of certain groups based on outward appearances. Another such problematic thinker was Paul Broca (1824‒1880), after whom a region of the frontal lobe related to language use is named (Broca’s area). Influenced by Morton, Broca likewise claimed that internal skull capacities could be linked with skin color and cognitive ability. He went on to justify the European colonization of other global territories by purporting it was noble for a biologically more “civilized” population to improve the “humanity” of more “barbaric” populations. Today, these theories of Morton, Broca, and others like them are known to have no scientific basis. If we could speak with them today, they would likely try to emphasize that their conclusions were based on empirical evidence and not a priori reasoning. However, we now can clearly see that their reasoning was biased and affected by prevailing societal views at the time.
“Race” and the Beginnings of Physical Anthropology
In the early 20th century, we saw a number of new figures coming into the science of human variation and shifting the theoretical focus within. Most notably, these included Aleš Hrdlička and Franz Boas.

Aleš Hrdlička (1869‒1943) was a Czech anthropologist who moved to the United States. In 1903, he established the physical anthropology section of the National Museum of Natural History (Figure 14.8). In 1918, he founded the American Journal of Physical Anthropology, which remains one of the foremost scientific journals disseminating bioanthropological research. As part of his work and the scope of the journal, he differentiated “physical anthropology” from other kinds of anthropology: he wrote that physical anthropology is “the study of racial anatomy, physiology, and pathology” and “the study of man’s variation” (Hrdlička 1918). In some ways, although the scope and technological capabilities of biological anthropologists have changed significantly, Hrdlička established an area of inquiry that has continued and prospered for over a hundred years.
Franz Boas (1858‒1942) was a German American anthropologist who established the four-field anthropology system in the United States and founded the American Anthropological Association in 1902. He argued that the scientific method should be used in the study of human cultures and the comparative method for looking at human biology worldwide. One of Boas’s significant contributions to biological anthropology was the study of skull dimensions with respect to race. After a long-term research project, he demonstrated how cranial form was highly dependent on cultural and environmental factors and that human behaviors were influenced primarily not by genes but by social learning. He wrote in one essay for the journal Science: “While individuals differ, biological differences between races are small. There is no reason to believe that one race is by nature so much more intelligent, endowed with great willpower, or emotionally more stable than another, that the difference would materially influence its culture” (Boas 1931:6). This conclusion directly contrasted with the theories of the past that relied on biological determinism. Biological anthropologists today have found evidence that corroborates Boas’s explanations: societies do not exist on a hierarchy or gradation of “civilizedness” but instead are shaped by the world around them, their demographic histories, and the interactions they have with other groups.
The first half of the 1900s still involved some research that was essentialist and focused on proving racial determinism. Anthropologists like Francis Galton (1822‒1911) and Earnest A. Hooton (1887‒1954) created the field of eugenics as an attempt to formalize the social scientific study of “fitness” and “superiority” among members of 19th-century Europe. As a way of “dealing with” criminals, diseased individuals, and “uncivilized” people, eugenicists recommended prohibiting parts of the population from being married or sterilizing these members of society so they could no longer procreate (Figure 14.9). They instead encouraged “reproduction in individual families with sound physiques, good mental endowments, and demonstrable social and economic capability” (Hooton 1936). In the 1930s, Nazi Germany used this false idea of there being “pure races” to highly destructive effect. The need to be protected against admixture from “unfit” groups was their justification for their blatant racism and purging of citizens that fell under their subjective criteria.
It should be noted that eugenicist ideas were popularly discussed and debated in many non-European contexts, as in the U.S., China, and South Africa, too. The Immigration Restriction Act of 1924 was passed in the United States, with the explicit aim of reducing the country’s “burden” of people considered inferior by restricting immigration of eastern European Jews, Italians, Africans, Arabs, and Asians. In the early 1900s, Chinese scientists and politicians showed great interest in eugenic ideologies, which came to dictate decisions in law-making, family life, and the medical field. Noted American anthropologist Ruth Benedict wrote extensively on Japanese culture and society during and after World War II. Her essentialist portrayals of Japanese people were heavily cited in popular discourse at the time. In 1950s South Africa, interracial marriages and sexual relations were banned by law; antimiscegenation became one of the huge focuses of apartheid resistance activists in later years. We still see the continuation of eugenics-based logic today around the world—in exclusionary immigration laws, cases of incarcerated prison inmates being forcibly sterilized, and the persistence of intelligence testing as a form of measuring people’s “fitness” in a society.
Shortly after World War II and the Nazi Holocaust, the full extent of essentialist, eugenicist thinking became clear. Social constructions of race, and the notion that one can predict psychological or behavioral traits based on external appearance, had become unpopular both within and outside the discipline. It was up to those in the field of physical anthropology at the time to separate physical anthropology from race concepts that supported unscientific and socially damaging agendas. This does not mean that there are no physiological or behavioral differences between different members of the human species. However, going forward, a number of physical anthropologists saw human biological variation as more complicated than simple typologies could describe.
“The New Physical Anthropology”

After 1950, focus steered away from the concept of “race” as a unit of variation and toward understanding why variation exists in populations from an evolutionary perspective. This was outlined by those pioneering the “new physical anthropology,” such as Sherwood Washburn, Theodosius Dobzhansky (Figure 14.10), and Julian Huxley, who borrowed this approach from contemporary population geneticists. Whether using genetic or phenotypic markers as the units of study, “groups” or “clusters” of humans differentiated by these became defined as populations that differ in the frequency of some gene or genes. Anthropologists consider what “makes” a population—a group of individuals potentially capable of or actually interbreeding due to shared geographic proximity, language, ethnicity, culture, and/or values. Put another way, a population is a local interbreeding group with reduced gene flow between themselves and other groups of humans. Members of the same population may be expected to share many genetic traits (and, as a result, many phenotypic traits that may or may not be visible outwardly).

Thinking of humans in terms of populations was part of Julian Huxley’s (1942) “Modern Synthesis”—so named because it helped to reconcile two fundamental principles about evolution that had not been made sense of together before (Figure 14.11). As discussed in Chapter 3, Gregor Mendel (1822‒1884) was able to show that inheritance was mediated by discrete particles (or genes) and not blended in the offspring. However, it was difficult for some 19th-century scientists to accept this model of genetic inheritance at the time because much of biological variation appeared to be continuous and not particulate (take skin color or height as examples). In the 1930s, it was demonstrated that traits could be polygenic and that multiple alleles could be responsible for any one phenotypic trait, thus producing the continuous variation in traits such as eye color that we see today. Thus, Huxley’s “Modern Synthesis” outlines not only how human populations are capable of exchanging genes at the microevolutionary level but also how multiple alleles for one trait (polygenic exchanges) can cause gradual macroevolutionary changes.
Human Variation in Biological Anthropology Today
Many Human Traits Are Nonconcordant
In our studies of human (genetic) variation today, we understand most human traits to be nonconcordant (Figure 14.12). “Nonconcordance” is a term used to describe how biological traits vary independent of each other—that is, they do not get inherited in a correlative manner with other genetically controlled traits. For example, if you knew an individual had genes that coded for tall height, you would not be able to predict if they are lighter-skinned or have red hair. This is different from earlier essentialist views of human variation: the idea that skin color could predict one’s brain function or even “temperament” and tendencies toward criminal behavior.
Human Variation Is Clinal/Continuous (Not Discrete)
Frank B. Livingstone (1928‒2005) wrote: “There are no races, only clines” (1962: 279). A cline is a gradation in the frequency of an allele/trait between populations living in different geographic regions. Human variation cannot be broken into discrete “races,” because most physical traits vary on a continuous or “clinal” basis. One obvious example of this is how human height does not only come in three values (“short,” “medium,” and “tall”) but instead varies across a spectrum of vertical heights achievable by humans all over the world. On the one hand, we can describe human height as exhibiting continuous variation, forming a continuous pattern, but height does not vary according to where people live across the globe and does not exhibit a clinal pattern. On the other hand, skin color variation between populations does show patterning that fits quite well on to how near or far they are from each other on a world map. This makes a trait like skin color clinally distributed worldwide. When large numbers of genetic loci for large numbers of samples were sampled from human populations distributed worldwide during the 1960s and 1970s, the view that certain facets of human diversity were clinally distributed was further supported by genetic data.
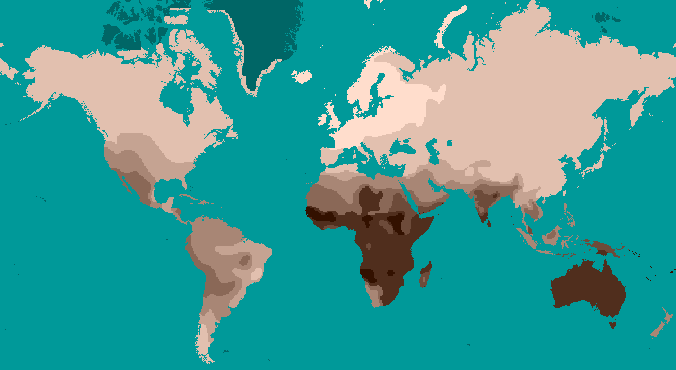
To study human traits that are clinally distributed, genetic tests must be performed to uncover the true frequencies of an allele or trait across a certain geographic space. One easily visible example of a clinal distribution seen worldwide is the patterning of human variation in skin color. Whether in southern Asia, sub-Saharan Africa, or Australia, dark brown skin is found. Paler skin tones are found in higher-latitude populations such as those who have lived in areas like Europe, Siberia, and Alaska for millennia. Skin color is easily observable as a phenotypic trait exhibiting continuous variation.
A clinal distribution still derives from genetic inheritance; however, clines often correspond to some gradually changing environmental factor. Clinal patterns arise when selective pressures in one geographic area differ from those in another as well as when people procreate and pass on genes together with their most immediate neighbors. There are several mechanisms, selective and neutral, that can lead to the clinal distribution of an allele or a biological trait. Natural selection is the mechanism that produced a global cline of skin color, whereby darker skin color protects equatorial populations from high amounts of UV radiation; there is a transition of lessening pigmentation in individuals that reside further and further away from the tropics (Jablonski 2004; Jablonski and Chaplin 2000; see Figure 14.13). The ability and inability to digest lactose (milk sugar) among different world communities varies according to differential practices and histories of milk and dairy-product consumption (Gerbault et al. 2011; Ingram et al. 2009). Where malaria seems to be most prevalent as a disease stressor on human populations, a clinal gradient of increasing sickle cell anemia experience toward these regions has been studied extensively by genetic anthropologists (Luzzatto 2012). Sometimes, culturally defined mate selection based on some observable trait can lead to clinal variation between populations as well.
Two neutral microevolutionary processes that may produce a cline in a human allele or trait are gene flow and genetic drift (see Chapter 4). The ways in which neutral processes can produce clinal distributions is seen clearly when looking at clinal maps for different blood groups in the human ABO blood group system (Figure 14.14). For instance, scientists have identified an East-to-West cline in the distribution of the blood type B allele across Eurasia. The frequency of B allele carriers decreases gradually westward when we compare the blood groups of East and Southeast Asian populations with those in Europe. This shows how populations residing nearer to one another are more likely to interbreed and share genetic material (i.e., undergo gene flow). We also see 90%‒100% of native South American individuals, as well as between 70%‒90% of Aboriginal Australian groups, carrying the O allele (Mourant, Kopeć, and Domaniewska-Sobczak 1976). These high frequencies are likely due to random genetic drift and founder effects, in which population sizes were severely reduced by the earliest O allele-carrying individuals migrating into those areas. Over time, the O blood type has remained predominant.
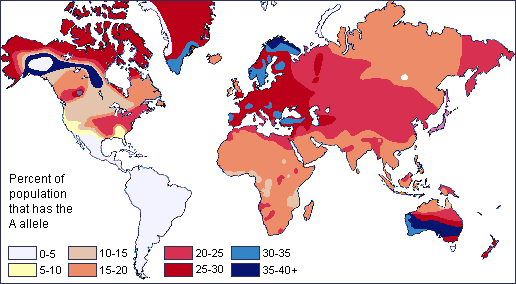
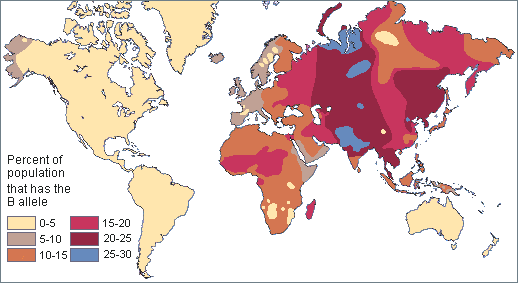
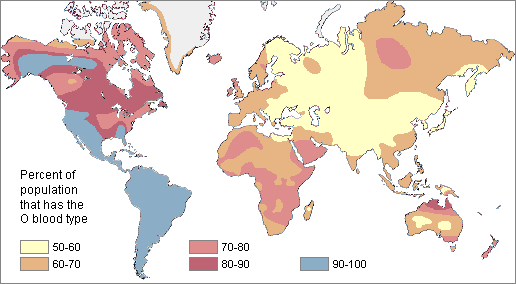
Genetic Variation Is Greater Within Group than Between Groups
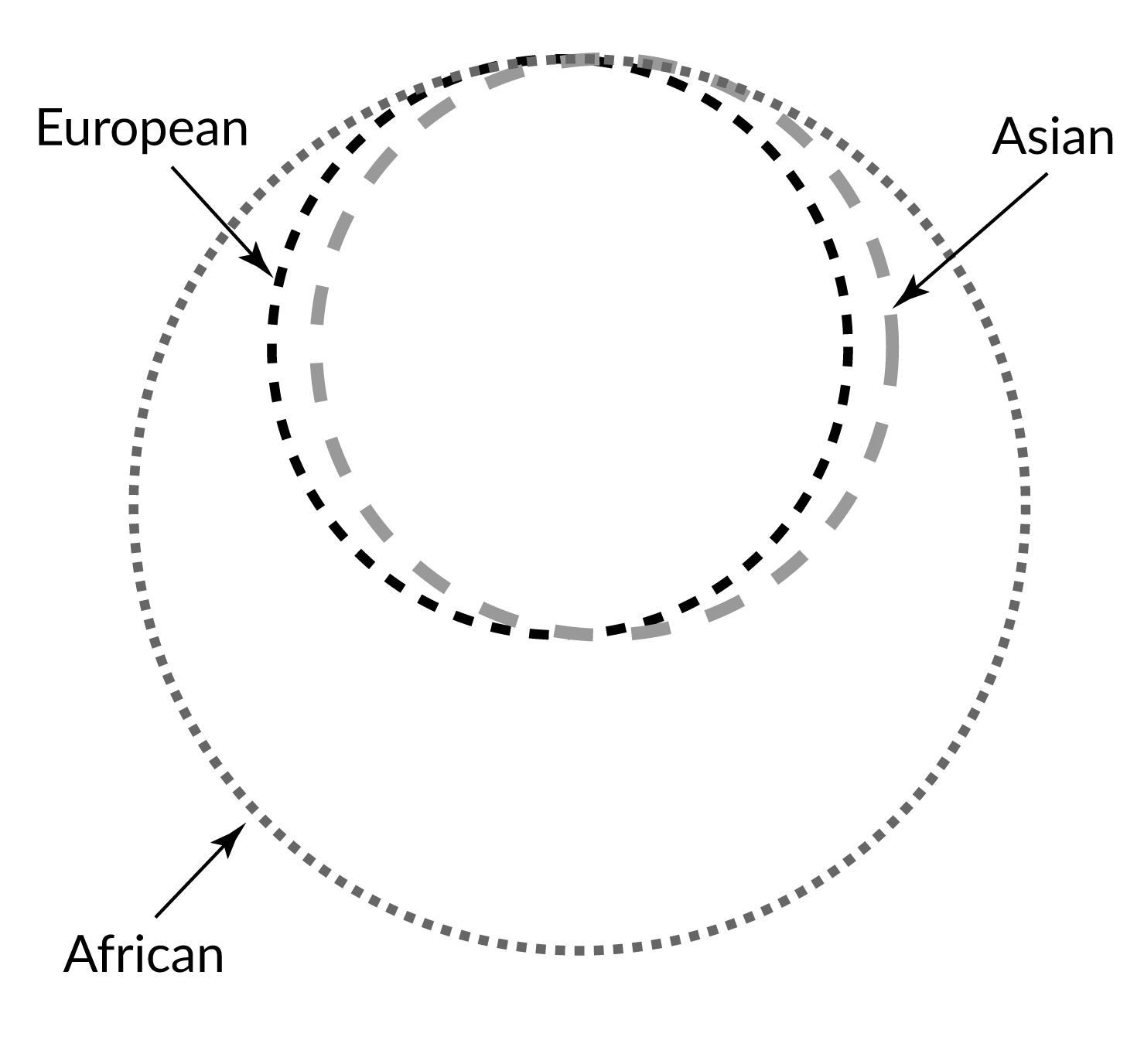
One problem with race-based classifications is they relied on an erroneous idea that individuals with particular characteristics would share more similar genes with each other within a particular “race” and share less with individuals of other “races” possessing different traits and genetic makeups. However, since around 50 years ago, scientific studies have shown that the majority of human genetic differences worldwide exist within groups (or “races”) individually rather than between groups. Indeed, most genetic variation we see occurs in Africa, and many variants are shared among individuals on all continents (Figure 14.15).
In 2002, a landmark article by Noah Rosenberg and colleagues explored worldwide human genetic variation using an even-greater genetic data set. They used 377 highly variable markers in the human genome and sampled from 1,056 individuals representative of 52 populations. The markers chosen for study were not ones that code for any expressed genes. Because these regions of the human genome were made of unexpressed genes, we may understand these markers as neutrally derived (as opposed to selectively derived) because they do not code for functional advantages or disadvantages. These neutral genetic markers likely reflect an intricate combination of regional founder effects and population histories. Analyses of these neutral markers allowed scientists to identify that 93%‒95% of global genetic differences, referred to as variance, can be accounted for by within-population differences, while only a small proportion of genetic variance (3%‒5%) can be attributed to differences among major groups (Rosenberg et al. 2002). This research supports the theory that distinct biological races do not exist, even though misguided concepts of race may still have real social and political consequences.
Biological Data Fit Isolation-By-Distance and Out-of-Africa Models
One further note is that the world’s population may be genetically divided into “groups,” “subsets,” “clumps,” or “clusters” that reflect some degree of genetic similarity. These identifiable clusters reflect genetic or geographic distances—either with gene flow facilitated by proximity between populations or impeded by obstacles like oceans or environmentally challenging habitats (Rosenberg et al. 2005). Sometimes, inferred clusters using multiple genetic loci are interpreted by nongeneticists literally as “ancestral populations.” However, it would be wrong to assume from these genetic results that highly differentiated and “pure” ancestral groups ever existed. These groupings reflect differences that have arisen over time due to clinal patterning, genetic drift, and/or restricted or unrestricted gene flow (Weiss and Long 2009). The clusters identified by scientists are arbitrary and the parameters used to split up the global population into groups is subjective and dependent on the particular questions or distinctions being brought into focus (Relethford 2009).
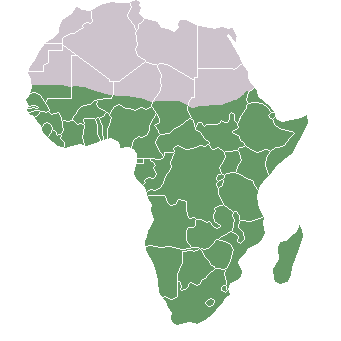
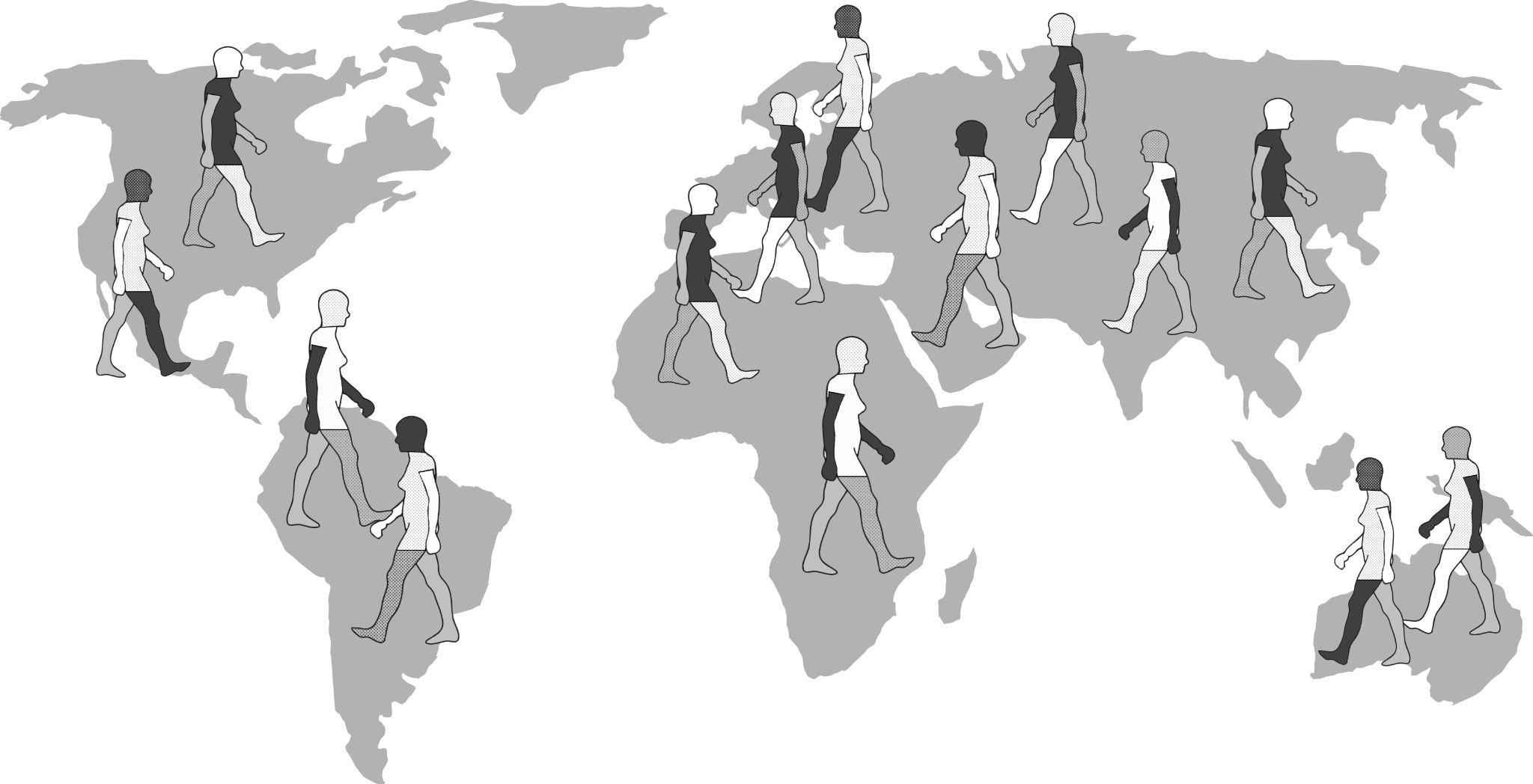
Additionally, research on worldwide genetic variation has shown that human variation decreases with increasing distance from sub-Saharan Africa, where there is evidence for this vast region being the geographical origin of anatomically modern humans (Liu et al. 2006; Prugnolle, Manica, and Balloux 2005; see Figures 14.16 and 14.17). Genetic differentiation decreases in human groups the further you sample data from relative to sub-Saharan Africa because of serial founder effects (Relethford 2004). Over the course of human colonization of the rest of the world outside Africa, populations broke away in expanding waves across continents into western Asia, then Europe and eastern Asia, followed by Oceania and the Americas. As a result, founder events occurred whereby genetic variation was lost, as the colonization of each new geographical region involved a smaller number of individuals moving from the original larger population to establish a new one (Relethford 2004). The most genetic variation is found across populations residing in different parts of sub-Saharan Africa, while other current populations in places like northern Europe and the southern tip of South America exhibit some of the least genetic differentiation relative to all global populations (Campbell and Tishkoff 2008).
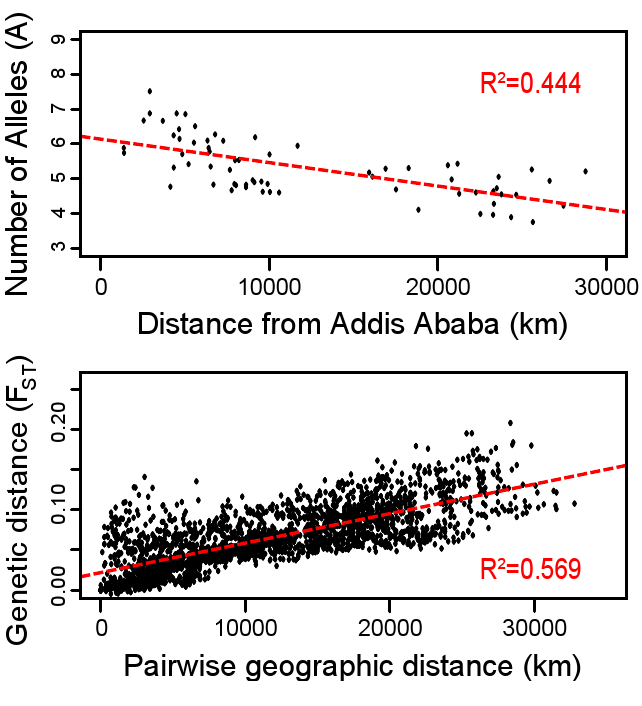
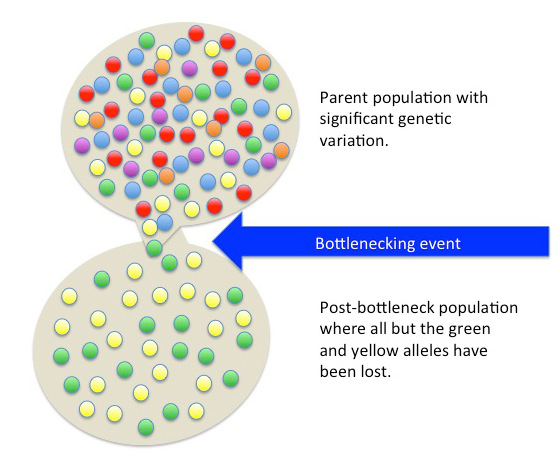
Besides fitting nicely into the Out-of-Africa model, worldwide human genetic variation conforms to an isolation-by-distance model, which predicts that genetic similarity between groups will decrease exponentially as the geographic distance between them increases (Kanitz et al. 2018). This is because of the greater and greater restrictions to gene flow presented by geographic distance, as well as cultural and linguistic differences that occur as a result of certain degrees of isolation. Since genetic data conform to isolation-by-distance and Out-of-Africa models, these findings support the abolishment of “race” groupings. This research demonstrates that human variation is continuous and cannot be differentiated into geographically discrete categories. There are no “inherent” or “innate” differences between human groups; instead, variation derives from some degree of natural selection, as well as neutral processes like population bottle-necking (Figure 14.18), random mutations in the DNA, genetic drift, and gene flow through between-mate interbreeding.
Humans Have Higher Homogeneity Compared to Many Other Species
An important fact to bear in mind is that humans are 99.9% identical to one another. This means that the apportionments of human variation discussed above only concern that tiny 0.1% of difference that exists between all humans globally. Compared to other mammalian species, including the other great apes, human variation is remarkably lower. This may be surprising given that the worldwide human population has already exceeded seven billion, and, at least on the surface level, we appear to be quite phenotypically diverse. Molecular approaches to human and primate genetics tells us that external differences are merely superficial. For a proper appreciation of human variation, we have to look at our closest relatives in the primate order and mammalian class. Compared to chimpanzees, bonobos, gorillas and other primates, humans have remarkably low average genome-wide heterogeneity (Osada 2005).
When we look at chimpanzee genetic variation, it is fascinating that western, central, eastern, and Cameroonian chimpanzee groups have substantially more genetic variation between them than large global samples of human DNA (Bowden et al. 2012; Figure 14.19). This is surprising given that all of these chimpanzee groups live relatively near one another in Africa, while measurements of human genetic variation have been conducted using samples from entirely different continents. First, geneticists suppose that this could reflect differential experiences of the founder effect between humans and chimpanzees. As it has been argued that all non-African human populations descended from a small number of anatomically modern humans who left Africa, it would be expected that all groups descended from that smaller ancestral group would be similar genetically. Second, our species is really young, given that we have only existed on the planet for around 150,000 to 300,000 years. This gave humans little time for random genetic mutations to occur as genes get passed down through genetic interbreeding and meiosis. Chimpanzees, however, have inhabited different ecological niches, and less interbreeding has occurred between the four chimpanzee groups over the past six to eight million years compared to the amount of gene flow that occurred between worldwide human populations (Bowden et al. 2012).
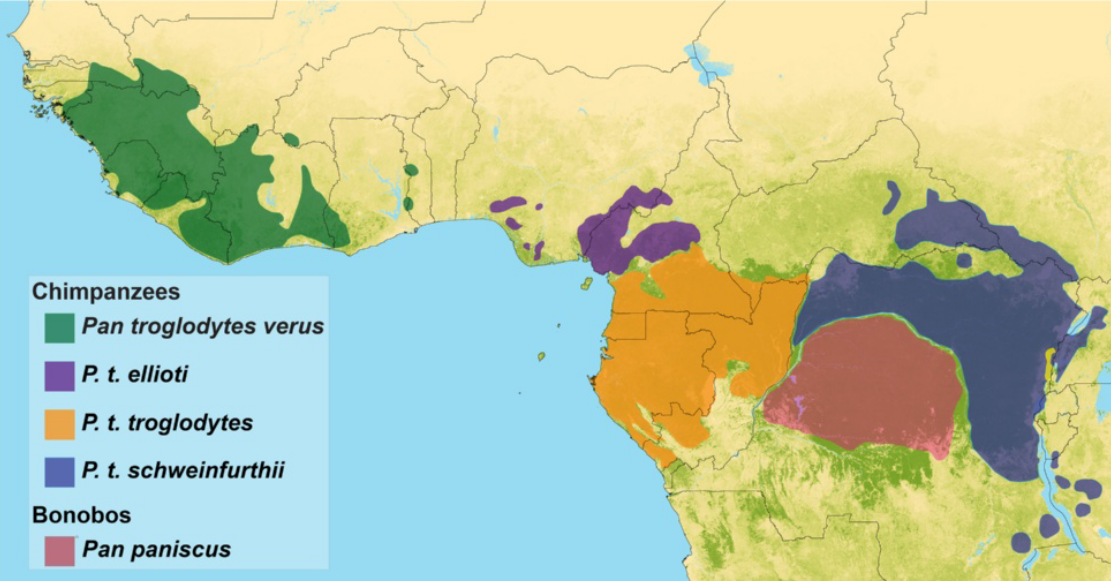
Recent advances have now enabled the attainment of genetic samples from the larger family of great apes and the evaluation of genetic variation among bonobos, orangutans, and gorillas alongside that of chimpanzees and humans (Prado-Martinez et al. 2013). Collecting such data and analyzing primate genetic variation has been important not only to elucidate how different ecological, demographic, and climatic factors have shaped our evolution but also to inform upon conservation efforts and medical research. Genes that may code for genetic susceptibilities to tropical diseases that affect multiple primates can be studied through genome-wide methods. Species differences in the genomes associated with speech, behavior, and cognition could tell us more about how human individuals may be affected by genetically derived neurological or speech-related disorders and conditions (Prado-Martinez et al. 2013; Staes et al. 2017). In 2018, a great ape genomic study also reported genetic differences between chimpanzees and humans related to brain cell divisions (Kronenberg et al. 2018). From these results, it may be inferred that cognitive or behavioral variation between humans and the great apes might relate to an increased number of cortical neurons being formed during human brain development (Kronenberg et al. 2018). Comparative studies of human and nonhuman great ape genetic variation highlight the complex interactions of population histories, environmental changes, and natural selection between and within species. When viewed in the context of overall great ape variation, we may reconsider how variable the human species is relatively and how unjustified previous “race” concepts really were.
Phenotypic Traits That Reflect Neutral Evolution
Depending on the trait being observed, different patterns of phenotypic variation may be found within and among groups worldwide. In this subsection, some phenotypic traits that reflect the aforementioned patterns of genetic variation will be discussed.

Looking beyond genetic variation briefly, recent studies have revisited biological anthropology’s earlier themes of externally observable traits, such as skull shape. In the last 20 or so years, anthropologists have evaluated the level to which human cranial shape variation reflects the results from genetic markers, such as those used previously to fit against Out-of-Africa models (Relethford 2004) or those used in the apportionment of human variation between and within groups (Lewontin 1972; Rosenberg et al. 2002). Using larger sample sizes of cranial data collected from thousands of skulls worldwide and a long list of cranial measurements, studies demonstrate a similar decrease in variation with distance from Africa and show that a majority of cranial variation occurs within populations rather than between populations (Betti et al. 2009; Betti et al. 2010; Manica et al. 2007; Relethford 2001; von Cramon-Taubadel and Lycett 2008; see Figure 14.20). The greatest cranial variation is found among skulls of sub-Saharan African origin, while the least variation is found among populations inhabiting places like Tierra del Fuego at the southern tip of Argentina and Chile. While ancient and historical thinkers previously thought “race” categories could reasonably be determined based on skull dimensions, modern-day analyses using more informative sets of cranial traits simply show that migrations out of Africa and the relative distances between populations can explain a majority of worldwide cranial variation (Betti et al. 2009).
This same patterning in phenotypic variation has even been found in studies examining shape variation of the pelvis (Betti et al. 2013; Betti et al. 2014), the teeth (Rathmann et al. 2017), and the human bony labyrinth of the ear (Ponce de León et al. 2018;Figure 14.21). The skeletal morphology of these bones still varies worldwide, but a greater proportion of that variation can still be attributed to the ways in which human populations migrated across the world and exchanged genes with those closer to them rather than those further away. Human skeletal variation in these parts of the body is continuous and nondiscrete. Given the important functions of the cranium and these other skeletal parts, we may infer that the genes that underpin their development have been relatively conserved by neutral evolutionary processes such as genetic drift and gene flow. It is also important to note that while some traits such as height, weight, cranial dimensions, and body composition are determined, in part, by genes, the underlying developmental processes behind these traits are underpinned by complex polygenic mechanisms that have led to the continuous spectrum of variation in such variables among modern-day human populations.
Phenotypic Traits That Reflect Natural Selection
Even though 99.9% of our DNA is the same across all humans worldwide, and many traits reflect neutral processes, there are parts of that remaining 0.1% of the human genome that code for individual and regional differences. Similarly to craniometric analyses that have been conducted in recent decades, human variation in skin color has also been reassessed using new methods and in light of greater knowledge of biological evolution.
New technologies allow scientists to use color photometry to sample and quantify the visible wavelength of skin color, in a way 19th- and 20th-century readers could not. In one report, it was found that 87.9% of global skin color variation can be attributed to genetic differences between groups, 3.2% to those among local populations within regions, and 8.9% within local populations (Relethford 2002). This apportionment differs significantly and is the reverse situation found in the distribution of genetic differences we see when we examine genetic markers such as blood type–related alleles. However, this pattern of human skin color worldwide is not surprising, given that we now understand that past selection has occurred for darker skin near the equator and lighter skin at higher latitudes (Jablonski 2004; Jablonski and Chaplin 2000). While most genetic variation reflects neutral variation due to population migrations, geographic isolation, and restricted gene flow dynamics, some human genetic/phenotypic variation is best explained as local adaptation to environmental conditions (i.e., selection). Given that skin color variation is atypical compared to other genetic markers and biological traits, this, in fact, goes against earlier “race” typologies. This is because recent studies ironically show how so much of genetic variation relates to neutral processes, while skin color does not. It follows that skin color cannot be viewed as useful in making inferences about other human traits.

It is also true that some populations have not been studied extensively in skin pigmentation genetics (e.g., African, Austronesian, Melanesian, Southeast Asian, Indigenous American, and Pacific Islander populations, according to Lasisi and Shriver 2018). Earlier dispersals of these populations, and their local genetic varition, will have contributed to worldwide genetic variation, inclusive of skin pigmentation variation. Gene loci we did not previously appreciate as being linked to pigmentation are now being recognized thanks to better tools, more diverse genetic samples, and more accessible datasets (Quillen et al. 2018). Biological anthropologists look forward to further discoveries elucidating the different selective pressures and population dynamics that influence skin pigmentation evolution.
Social Implications
To finish this chapter, we will consider the social, economic, political, and biological implications of poor understandings of race and the deliberate perpetuation of social and medical racism.
The Black Lives Matter movement (BLM) of 2013 began with the work of racial justice activists and community organizers Alicia Garza, Opal Tometi, and Patrissa Cullors. First incited by the murder of Trayvon Martin, a 17-year-old African American, and the acquittal of the man who shot him, BLM went on to protest against the deaths of numerous Black individuals, most of whom were killed by police officers (for example, Ahmaud Arbery was killed in February of 2020 by two white non-police officers). Some key characteristics of BLM from the start were its decentralized grassroots structure, the role of university students and social media in spreading awareness of the movement, and its embrace of other movements (e.g., climate justice, ending police brutality, feminist campaigns, queer activism, immigration reform, etc.). When George Floyd was murdered by a white police officer on May 25, 2020, the BLM gained new momentum, across 2,000-plus cities in the United States, and among many protesting against historic racism and police brutality in other contexts around the globe. Many in the biological anthropology community have responded to these events with a great dedication to working against systemic racism in society and institutions (American Association of Biological Anthropologists 2020).
BLM continues to be an important movement, as is evidenced in the degree of community organizing, mutual aid efforts, calls for political reform, progress toward curriculum reform and equality, inclusion and diversity (EDI) work in businesses and universities, the removal of monuments honoring historical figures associated with slavery and racism, and many other important actions. Garza (2016) writes: “The reality is that race in the United States operates on a spectrum from black to white … the closer you are to white on that spectrum, the better off you are.” Tometi (2016) has stated: “We need [a human rights movement that challenges systemic racism] because the global reality is that Black people are subject to all sorts of disparities in most of our challenging issues of our day. I think about climate change, and how six of the ten worst impacted nations by climate change are actually on the continent of Africa.” In the words of Cullors (2016), “Black Lives Matter is our call to action. It is a tool to reimagine a world where Black people are free to exist, free to live. It is a tool for our allies to show up differently for us.” We gather from their words the importance of learning from the egregious role that anthropologists have played in the past, recognizing the legacies of “scientific” justifications for eugenics and racism in our society today, and proactively working toward environmental and social equity.
Another major industry that engages in the quantification and interpretation of human variation is medical and clinical work (National Research Council [U.S.] Committee on Human Genome Diversity 1997). Large-scale genomic studies sampling from human populations distributed worldwide have produced detailed knowledge on variation in disease resistance or susceptibility between and within populations. Let’s think about drug companies who develop medicines for Black patients particularly. The predispositions to particular diseases are higher among people of African descent than some pharmaceutical businesses have taken into account. Through targeted sampling of various world groups, clinical geneticists may also identify genetic risk factors of certain common disorders such as chronic heart disease, asthma, diabetes, autoimmune diseases, and behavioral disorders. Having an understanding of population-specific biology is crucial in the development of therapies, medicines, and vaccinations, as not all treatments may be suitable for every human, depending on their genotype. During diagnosis and treatment, it is important to have an evolutionary perspective on gene-environment relationships in patients. Typological concepts of “race” are not useful, given that most racial groups (whether self-identified or not) popularly recognized lack homogeneity and are, in fact, variable. Cystic fibrosis, for instance, occurs in all world populations but can often be underdiagnosed in populations with African ancestry because it is thought of as a “white” disease (Yudell et al. 2016).
Sociologists, law scholars, and professors of race studies have written extensively on how genetic/technological/medical revolutions impact people of color. In her book, Fatal Invention: How Science, Politics, and Big Business Re-create Race in the Twenty-First Century (2013), Professor Dorothy E. Roberts writes about how technological advances have been used in resuscitating race as a biological category for dividing humans in essentialist ways (Figure 14.23). She notes how members of law enforcement have engaged in racial profiling, sometimes with the use of machine-learning and facial-recognition technologies. Ancestry-testing services also purport to tell us “what” we are and to insist that this information is “written” in our genes. Such advertising campaigns obscure the nuances of genetic variation with the primary motive of tapping into people’s desire to “know themselves” and driving up profits for their businesses. Commercial genetic testing reinforces the idea that genes map neatly onto race, all while generating massive stores of data in DNA databases. In Roberts’s view, the myth of the biological concept of race being perpetuated in these ways undermines a just society and reproduces racial inequalities.

The COVID-19 pandemic has had a significant impact on the world’s population, particularly people living in the economic Global South and many Black, Indigenous and communities of color residing in the Global North. We have witnessed disproportionately high numbers of COVID-related deaths and infection cases among marginalized groups. Many immigrants and ethnic minorities in various societies have also experienced scapegoating and blame directed at them for being the source of COVID-19 spread.
To inform us on how to interpret this current worldwide pandemic, historians and anthropologists are looking back at the lessons learned from past instances of racist medicine (discriminatory practices based on broader social discrimination) and medical racism (application of discriminatory practices justified on medical grounds). Historically, who could become doctors and medical professionals was often racialized, gendered, and class specific. This made it difficult for many to overcome prejudices against women, Black people, Indigenous individuals, or other people of color from becoming doctors and clinical researchers in places such as South Africa and the United States. This, in turn, affects the sorts of information we know about health levels and health outcomes among these very groups. In the past decade, long-overdue attention is finally being paid to how race affects biological outcomes. For instance, researchers have focused on the negative legacies of racial discrimination and racism-induced stress on hormone (im)balances, mental health disorders, cardiovascular disease prevalence, and other health outcomes (Kuzawa and Sweet 2009; Shonkoff, Slopen, and WIlliams 2021; Williams 2018). The technology and standards of protocol in medical testing have been scrutinized (for more on how pulse oximeters were not designed with nonwhite patients in mind, for example, see Sjoding et al. 2020). Scholars of race and medicine have also written on how illness and disease spread have often been used to perpetuate societal prejudices. This manifests as xenophobic tendencies at a societal level, such as the blaming of “outgroups” and increased “in-group” protectiveness. Overreliance on the idea that people are “inherently” disease carriers due to genetic or biological reasons leads to improper accounting for socioeconomic or infrastructural issues that lead to differential disease prevalence amongst minority communities. (For more on race and COVID, see Tsai 2021 as well as this textbook’s Chapter 16: Contemporary Topics: Human Biology and Health.)
It is important to remember that while it is possible to look for clues about one’s ancestry or geographic origin based on skull morphology, again, the amount of distinctiveness in any given sample makes it impossible to distinguish whether a cranium belongs to one group (Relethford 2009). Individuals can vary in their skeletal dimensions by continental origin, country origin, regional origin, sex, age, environmental factors, and the time period in which they lived, making it difficult to assign individuals to particular categories in a completely meaningful way (Ousley, Jantz, and Freid 2009). When forensic reports and scientific journal articles give an estimation of ancestry, it is crucial to keep in mind that responsible assignments of ancestry will be done through robust statistical testing and stated as a probability estimate. Today, we also live in a more globalized world where a skeletal individual may have been born originally to parents of two separate traditional racial categories. In contexts of great heterogeneity within populations, this definitely adds difficulty to the work of forensic scientists and anthropologists preparing results for the courtroom (genetic testing may be comparatively more helpful in such situations).
Dig Deeper: Measuring FST
Richard Lewontin (1929‒) is a biologist and evolutionary geneticist who authored an article evaluating where the total genetic variation in humans lies. Titled “The Apportionment of Human Diversity” (Lewontin 1972), the article addressed the following question: On average, how genetically similar are two randomly chosen people from the same group when compared to two randomly chosen people from different groups?
Lewontin studied this problem by using genetic data. He obtained data for a large number of different human populations worldwide using 17 genetic markers (including alleles that code for various important enzymes and proteins, such as blood-group proteins). The statistical analysis he ran used a measure of human genetic differences in and among populations known as the fixation index (FST).
Technically, FST can be defined as the proportion of total genetic variance within a subpopulation relative to the total genetic variance from an entire population. Therefore, FST values range from 0 to 1 (or, sometimes you will see this stated as a percentage between 0% and 100%). The closer the FST value of a population (e.g., the world’s population) approaches 1, the higher the degree of genetic differentiation among subpopulations relative to the overall population (see Figure 14.24 for a detailed illustration).
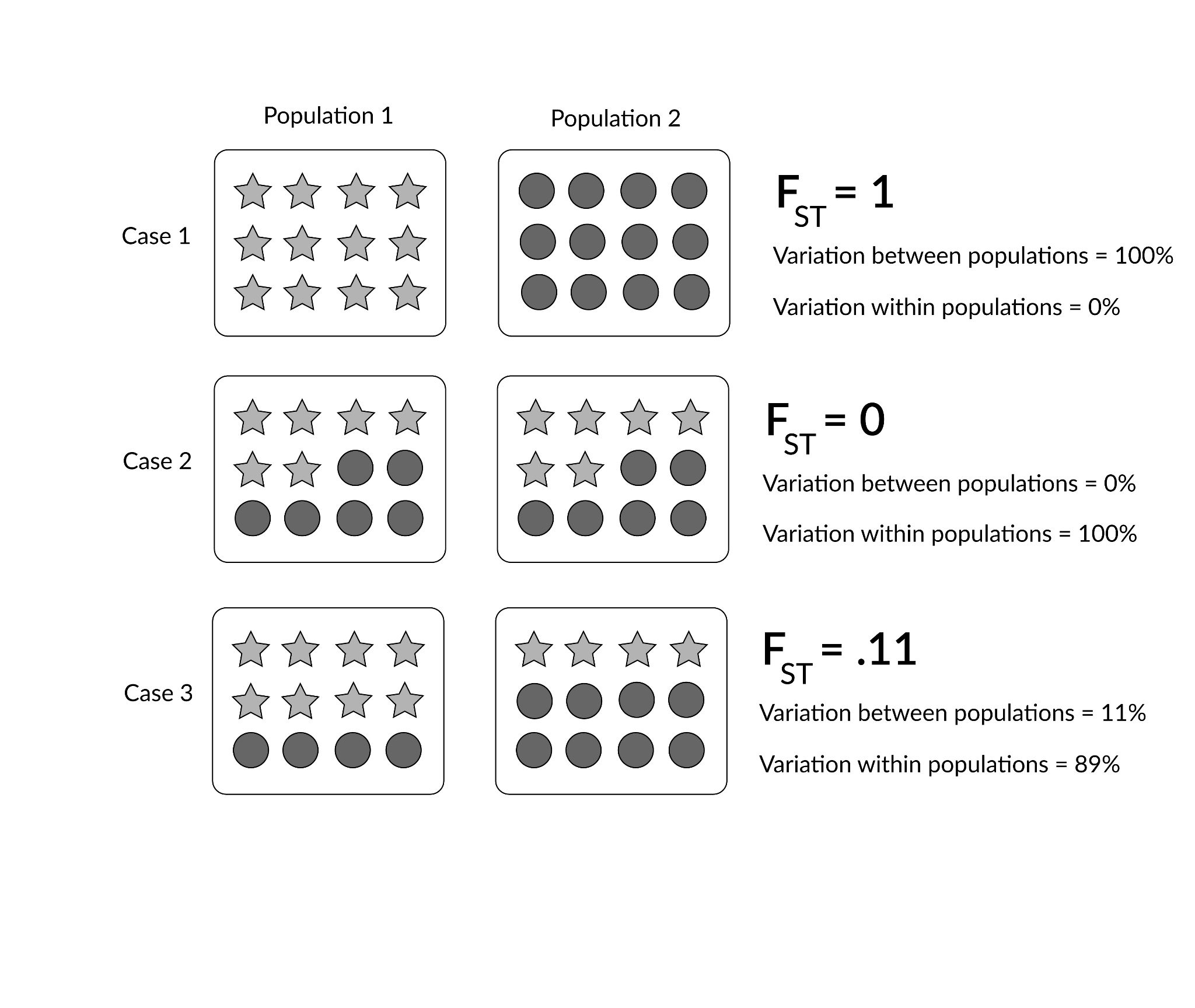
In his article, Lewontin (1972) identified that most of human genetic differences (85.4%) were found within local subpopulations (e.g., the Germans or Easter Islanders), whereas 8.3% were found between populations within continental human groups, and 6.3% were attributable to traditional “race” groups (e.g., “Caucasian” or “Amerind”). These findings have been important for scientifically rejecting the existence of biological races (Long and Kittles 2003).
Talking About Human Biological Variation Going Forward
To conclude, utilizing the term races to describe human biological variation is not accurate or productive. Using a select few hundred genetic loci, or perhaps a number of phenotypic traits, it may be possible to assign individuals to a geographic ancestry, but what constitutes a bounded genetic or geographical grouping is both arbitrary and potentially harmful owing to ethical and historical reasons. The discipline of biological anthropology has moved past typological frameworks that shoehorn continuously variable human populations into discrete and socially constructed subsets. Improvements in the number of markers, the genetic technologies used to study variation, and the number of worldwide populations sampled have led to more nuanced understandings of human variation. It is of utmost importance that scientists make the following points clear to the public:
- Today, we refer to different local human groups as “populations.” What constitutes a population should be carefully defined in scientific reports based on some geographical, linguistic, or cultural criteria and some degree of relativity to other closely or distantly related human groups.
- Humans have significantly less genetic variation than other primates and mammals, and all human beings on Earth share 99.9% of their overall DNA. Some of the remaining 0.1% of human variation varies on a clinal or continuous basis, such as can be seen when looking at ABO blood-type polymorphisms worldwide.
- Many biological characteristics in humans are actually determined nonconcordantly and/or polygenically. Therefore, superiority or inferiority in human behavior or body form cannot justifiably be linked to fixed and innate differences between groups.
- Genetic distances are correlated with geographic distances among the global human population. This is especially apparent when we consider that genetic variation is highest in sub-Saharan Africa, and average genetic heterogeneity decreases in populations further away from the African continent in accordance with the migratory history of anatomically modern Homo sapiens.
- The effects of gene flow, genetic drift, and population bottlenecking are reflected in some phenotypic traits, such as cranial shape.
- We recognize other traits, like skin color and lactase persistence, to be the products of many millennia of natural selective pressures influencing human biology from the external environment.
Taken together, genetic analyses of human variation do not support 20th-century (or even-earlier) concepts of race. In discussions about human variation, these genomic results help clarify how biological variation is distributed across the human population today. Taking care to think about and debate the nature of human variation is important, because although the effects and events that produced genetic differences among groups occurred in the ancient past, sociocultural concepts about race and ethnicity continue to have real social, economic, and political consequences.
Beyond talking about variation in the university setting, it is important that teachers, researchers, and students of anthropology recognize and assume the responsibility of influencing public perspectives of human variation. Race-based classification systems were developed during the colonial era, the transatlantic trafficking of kidnapped Africans and the so-called “Scientific Revolution” by the first “anthropologists” and scholars of humankind’s variation. Unfortunately, some of their early ideas have persisted and evolved into present-day lived realities. Some of today’s politicians and socioeconomic bodies have racially charged agendas that promote racism or certain kinds of economic or racial inequalities. As anthropologists, we must acknowledge that while human “races” are not a biological reality, their status as a (misguided) social construction does have real consequences for many people (Antrosio 2011).
In other words, while “race” is a sociocultural invention, the treatment different individuals receive due to their perceived “race” can have significant financial, emotional, sociopolitical, and physiological costs. However—and importantly assuming a “color-blind” position when it comes to the topics of “race” and ethnicity (especially in political discussions) is actually counterproductive, because the negative social consequences of modern “race” ideas could be ignored, making it harder to examine and address instances of discrimination properly (Wise 2010). Rather than shy away from these topics, we can use our scientific findings to establish socially relevant and biologically accurate ideas concerning human diversity. Today, research into genetic and phenotypic differentiation among and within various human populations continues to expand in its scope, its technological capabilities, its sample sizes, and its ethical concerns. It is thanks to such scientific work done in the past few decades that we now have a deeper understanding not only of how humans vary but also of how we are biologically a rather homogenous, intermixing world population.
Summary
Historically, most concepts of race were shaped by religious and early ‘scientific’ attempts to classify people, many of which justified inequality and racism. Today, biological anthropology emphasizes that human variation is continuous (clinal), polygenic, and shaped by both evolutionary pressures and neutral processes. Misunderstandings of race, however, continue to have serious social and medical consequences; evident in systemic racism and inequities in health care.
Biological anthropologists now play a critical role in discrediting myths about race. By studying human variation, we can begin to understand evolutionary processes, adaptation, and the social implications of differences among human populations. Modern research shows that that humans are far more genetically homogenous than many other species, reinforcing a conclusion that ‘race’ is not a biological reality but a powerful social construct with real effects. Through evolutionary history and genetics, human diversity can be researched and understood, rather than than through racial categories.
Review Questions
- How is the genetic variation of the human species distributed worldwide?
- What evolutionary processes are responsible for producing genotypic/phenotypic variation within and between human populations?
- Should we continue to attribute any value to “race” concepts older than 1950, based on our current understandings of human biological variation?
- How should we communicate scientific findings about human biological variation more accurately and responsibly to those outside the anthropological discipline?
Key Terms
Age of Discovery: A period between the late 1400s and late 1700s when European explorers and ships sailed extensively across the globe in pursuit of new trading routes and territorial conquest.
Ancestry: Biogeographical information about an individual, traced either through the study of an individual’s genome, skeletal characteristics, or some other form of forensic/archaeological evidence. Anthropologists carry out probabilistic estimates of ancestry. They attribute sets of human remains to distinctive “ancestral” groups using careful statistical testing and should report ancestry estimations with statistical probability values.
Binomial nomenclature: A system of naming living things developed by Linnaeus in the 1700s. It employs a scientific name made up of two italicized Latin or Greek words, with the first word capitalized and representative of an organism’s genus and the second word indicating an organism’s species (e.g., Homo sapiens, Australopithecus afarensis, Pongo tapanuliensis, etc.).
Biological anthropology: A branch of study under anthropology (the study of humankind) that focuses on when and where humans and our human ancestors first originated, how we have evolved and adapted globally over time, and the reasons why we see biological variation among humans worldwide today.
Biological determinism: The erroneous concept that an individual’s behavioral characteristics are innate and determined by genes, brain size, or other physiological attributes—and, notably, without the influence of social learning or the environment around the individual during development.
Bony labyrinth: A system of interconnected canals within the auditory (ear- or hearing-related) apparatus, located in the inner ear and responsible for balance and the reception of sound waves.
Cline: A gradient of physiological or morphological change in a single character or allele frequency among a group of species across environmental or geographical lines (e.g., skin color varies clinally, as, over many generations, human groups living nearer the equator have adapted to have more skin pigmentation).
Continuous variation: This term refers to variation that exists between individuals and cannot be measured using distinct categories. Instead, differences between individuals within a population in relation to one particular trait are measurable along a smooth, continuous gradient.
Cystic fibrosis: A genetic disorder in which one defective gene causes overproduction and buildup of mucus in the lungs and other bodily organs. It is most common in northern Europeans (but also occurs in other world populations).
Ecological niche: The position or status of an organism within its community and/or ecosystem, resulting from the organism’s structural and functional adaptations (e.g., bipedalism, omnivorous diets, lactose digestion, etc.).
Essentialism: A belief or view that an entity, organism, or human grouping has a specific set of characteristics that are fundamentally necessary to its being and classification into definitive categories.
Ethnicity: A term used commonly in an interchangeable way with the term race, complicated because how different people define this term depends on the qualities and characteristics they use to assign a label or identity to themselves and/or others (which may include aspects of family background, skin color, language(s) spoken, religion, physical proportions, behavior and temperament, etc.).
Eugenics: A set of beliefs and practices that involves the controlled selective breeding of human populations with the hope of improving their heritable qualities, especially through surgical procedures like sterilization and legal rulings that affect marriage rights for interracial couples.
Gene flow: A neutral (or nonselective) evolutionary process that occurs when genes get shared between populations.
Genetic drift: A neutral evolutionary process in which allele frequencies change from generation to generation due to random chance.
Heterogeneity: The quality of being diverse genetically.
Homogenous: The quality of being uniform genetically.
Human diversity: Human diversity is a measure of variation that may describe how many different forms of human there are, separated or clustered into groups according to some genetic, phenotypic, or cultural trait(s). The term can be applied to culture (in which case humans can be described as significantly diverse) or genetics (in which case humans are not diverse because all humans on Earth share a majority of their genes).
Human variation: Differences in biology, physiology, body chemistry, behavior, and culture. By measuring these differences, we understand the degrees of variation between individuals, groups, populations, or species.
Isolation-by-distance model: A model that predicts a positive relationship between genetic distances and geographical distances between pairs of populations.
Monogenetic: Pertaining to the idea that the origin of a species is situated in one geographic region or time (as opposed to polygenetic).
Mutation: A gene alteration in the DNA sequence of an organism. As a random, neutral evolutionary process that occurs over the course of meiosis and early cell development, gene mutations are possible sources of variation in any given human gene pool. Genetic mutations that occur in more than 1% of a population are termed polymorphisms.
Natural selection: An evolutionary process whereby certain traits are perpetuated through successive generations, likely owing to the advantages they give organisms in terms of chances of survival and/or reproduction.
Nonconcordance: The fact of genes or traits not varying with one another and instead being inherited independently.
Otherness: In postcolonial anthropology, we now understand “othering” to mean any action by someone or some group that establishes a division between “us” and “them” in relation to other individuals or populations. This could be based on linguistic or cultural differences, and it has largely been based on external characteristics throughout history.
Out-of-Africa model: A model that suggests that all humans originate from one single group of Homo sapiens in (sub-Saharan) Africa who lived between 100,000 and 315,000 years ago and who subsequently diverged and migrated to other regions across the globe.
Physical anthropology: This used to be the more common name given to the subdiscipline of anthropology centered upon the study of human origins, evolution and variation (also see biological anthropology above). This name for the field has gradually become less popular due to two reasons: first, it may not reflect our interests in other aspects of humankind that are not physical (such as those behavioral, cultural and spiritual), and second, using this term popular in the early decades of our field may be viewed by some as harkening back to a time when biological anthropologists conducted their work in unethical ways.
Polygenetic: Having many different ancestries, as in older theories about human origins that involved multiple traditional groupings of humans evolving concurrently in different parts of the world before they merged into one species through interbreeding and/or intergroup warfare. These earlier suggestions have now been overwhelmed by insurmountable evidence for a single origin of the human species in Africa (see the “Out-of-Africa model”).
Polymorphism: A genetic variant within a population (caused either by a single gene or multiple genes) that occurs at a rate of over 1% among the population. Polymorphisms are responsible for variation in phenotypic traits such as blood type and skin color.
Population: A group of humans living in a particular geographical area, with more local interbreeding within-group than interbreeding with other groups. A limited or restricted amount of gene flow between populations can occur due to geographical, cultural, linguistic, or environmental factors.
Population bottlenecking: An event in which genetic variation is significantly reduced owing to a sharp reduction in population size. This can occur when environmental disaster strikes or as a result of human activities (e.g., genocides or group migrations). An important example of this loss in genetic variation occurred over the first human migrations out of Africa and into other continental regions.
Prejudice: An unjustified attitude toward an individual or group that is not based on reason, whether positive (and showing preference for one group of people over another) or negative (and resulting in harm or injury to others).
Race: The identification of a group based on a perceived distinctiveness that makes that group more similar to each other than they are to others outside the group. This may be based on cultural differences, genetic parentage, physical characteristics, behavioral attributes, or something arbitrarily and socially constructed. As a social or demographic category, perceptions of “race” can have real and serious consequences for different groups of people. This is despite the fact that biological anthropologists and geneticists have demonstrated that all humans are genetically homogenous and that more differences can be found within populations than between them in the overall apportionment of human biological variation. This term is sometimes used interchangeably with ethnicity.
Racism: Any action or belief that discriminates against someone based on perceived differences in race or ethnicity.
Scientific Revolution: A period between the 1400s and 1600s when substantial shifts occurred in the social, technological, and philosophical sense, when a scientific method based on the collection of empirical evidence through experimentation was emphasized and inductive reasoning was used to test hypotheses and interpret their results.
Typological: Of or describing an assortment system that relies on the interpretation of qualitative similarities or differences in the study of variation among objects or people. The categorization of cultures or human groups according to “race” was performed with a typological approach in the earliest practice of anthropology, but this practice has since been discredited and abandoned.
Variance: In statistics, variance measures the dispersal of a set of data around the mean or average value.
For Further Exploration
Videos
American Medical Association (AMA). 2020. “Examining Race-Based Medicine.” YouTube, October 29. Accessed June 4, 2023.
Crenshaw, Kimberlé. 2016. “The Urgency of Intersectionality.” YouTube, December 7. Accessed June 4, 2023.
Golash-Boza, Tanya. 2018. “What Is Race? What Is Ethnicity? Is There a Difference?.” YouTube, October 28. Accessed June 4, 2023.
Lasisi, Tina. 2020. “How Hair Reveals the Futility of Race Categories.” National Museum of Natural History webinar, October 21.
Lasisi, Tina. 2022. “Where Does My Skin Color Come From?.” PBS Terra, August 18. Accessed June 4, 2023.
PBS Origins. 2018. “The Origin of Race in the USA.” YouTube, April 3. Accessed June 4, 2023.
Roberts, Dorothy. 2016. “The Problem with Race-Based Medicine.” YouTube, March 4. Accessed June 4, 2023.
Vox. 2015. “The Myth of Race, Debunked in 3 Minutes.” YouTube, January 13. Accessed June 4, 2023.
Podcast Episodes
Kwong, Emily, and Rebecca Ramirez. 2021. “Here’s a Better Way to Talk about Hair: A 16 Minute Listen with Tina, Lasisi” NPR Short Wave, October 6. Accessed June 4, 2023.
Speaking of Race. 2020. “Race and Health series.” Speaking of Race, April 10. Accessed June 4, 2023.
Websites
Choices Program. 2023. “An Interactive Timeline: Black Activism and the Long Fight for Racial Justice.” Choices Program, Brown University [Interactive Timeline], Updated February, 2023.
References
American Association of Biological Anthropologists. 2020. “An Open Letter to Our Community in Response to Police Brutality against African-Americans and a Call to Antiracist Action”. American Association of Biological Anthropologists, June 10, 2020. Accessed June 4, 2023.
Antrosio, Jason. 2011. “‘Race Reconciled’: Race Isn’t Skin Color, Biology, or Genetics.” Living Anthropologically (website), June 5, 2011; updated May 20, 2020. Accessed June 4, 2023.
Beals, Kenneth L., Courtland L. Smith, Stephen M. Dodd, J. Lawrence Angel, Este Armstrong, Bennett Blumenberg, Fakhry G. Girgis, et al. 1984. “Brain Size, Cranial Morphology, Climate, and Time Machines [and Comments and Reply].” Current Anthropology 25 (3): 301‒330.
Betti, Lia, François Balloux, Tsunehiko Hanihara, and Andrea Manica. 2010. “The Relative Role of Drift and Selection in Shaping the Human Skull.” American Journal of Physical Anthropology 141 (1): 76‒82. https://doi.org/10.1002/ajpa.21115.
Betti, Lia, François Balloux, William Amos, Tsunehiko Hanihara, and Andrea Manica. 2009. “Distance from Africa, Not Climate, Explains Within-Population Phenotypic Diversity in Humans.” Proceedings: Biological Sciences 276 (1658): 809‒814. https://doi.org/10.1098/rspb.2008.1563.
Betti, Lia, Noreen von Cramon-Taubadel, Andrea Manica, and Stephen J. Lycett. 2013. “Global Geometric Morphometric Analyses of the Human Pelvis Reveal Substantial Neutral Population History Effects, Even across Sexes.” PloS ONE 8 (2): e55909. https://doi.org/10.1371/journal.pone.0055909.
Betti, Lia, Noreen von Cramon-Taubadel, Andrea Manica, and Stephen J. Lycett. 2014. “The Interaction of Neutral Evolutionary Processes with Climatically Driven Adaptive Changes in the 3D Shape of the Human Os Coxae.” Journal of Human Evolution 73 (August): 64‒74. https://doi.org/10.1016/j.jhevol.2014.02.021.
Boas, Franz. 1931. “Race and Progress.” Science 74 1905): 1‒8.
Bowden, Rory, Tammie S. MacFie, Simon Myers, Garrett Hellenthal, Eric Nerrienet, Ronald E. Bontrop, Colin Freeman, Peter Donnelly, and Nicholas I. Mundy. 2012. “Genomic Tools for Evolution and Conservation in the Chimpanzee: Pan troglodytes ellioti Is a Genetically Distinct Population.” PLoS Genetics 8 (3): e1002504. https://doi.org/10.1371/journal.pgen.1002504.
Campbell, Michael C., and Sarah A. Tishkoff. 2008. “African Genetic Diversity: Implications for Human Demographic History, Modern Human Origins, and Complex Disease Mapping.” Annual Review of Genomics and Human Genetics 9: 403‒433.
Clee, Paul R. Sesink, Ekwoge E. Abwe, Ruffin D. Ambahe, Nicola M. Anthony, Roger Forso, Sabrina Locatelli, Fiona Maisels, et al. 2015. “Chimpanzee Population Structure in Cameroon and Nigeria Is Associated with Habitat Variation That May Be Lost Under Climate Change.” BMC Evolutionary Biology 15: 2. https://doi.org/10.1186/s12862-014-0275-z.
Cullors, Patrisse. 2016. “An Interview with the Founders of Black Lives Matter.” TED Talks 2016, October 26‒28. Accessed June 15, 2023. https://www.ted.com/talks/alicia_garza_patrisse_cullors_and_opal_tometi_an_interview_with_the_founders_of_black_lives_matter/up-next.
Fuentes, Agustín, Rebecca Rogers Ackermann, Sheela Athreya, Deborah Bolnick, Tina Lasisi, Sang-Hee Lee, Shay-Akil McLean, and Robin Nelson. 2019. “AAPA Statement on Race and Racism.” American Journal of Physical Anthropology 169 (3): 400‒402.
Garza, Alicia. 2016. “An Interview with the Founders of Black Lives Matter.” TED Talks 2016, October 26‒28. Accessed June 15, 2023. https://www.ted.com/talks/alicia_garza_patrisse_cullors_and_opal_tometi_an_interview_with_the_founders_of_black_lives_matter/up-next.
Gerbault, Pascale, Anke Liebert, Yuval Itan, Adam Powell, Mathias Currat, Joachim Burger, Dallas M. Swallow, and Mark G. Thomas. 2011. “Evolution of Lactase Persistence: An Example of Human Niche Construction.” Philosophical Transactions of the Royal Society B 366 (1566): 863‒877. https://doi.org/10.1098/rstb.2010.0268.
Hooton, Earnest A. 1936. “Plain Statements about Race.” Science 83 (2161): 511‒513.
Hrdlička, Aleš. 1918. “Physical Anthropology: Its Scope and Aims; Its History and Present Status in America. A: Physical Anthropology; Its Scopes and Aims.” American Journal of Physical Anthropology 1 (1): 3‒23.
Huxley, Julian. 1942. Evolution: The Modern Synthesis. London: Allen and Unwin.
Ingram, Catherine J. E., Charlotte A. Mulcare, Yuval Itan, Mark G. Thomas, and Dallas M. Swallow. 2009. “Lactose Digestion and the Evolutionary Genetics of Lactase Persistence.” Human Genetics 124 (6): 579‒591. https://doi.org/10.1007/s00439-008-0593-6.
Jablonski, Nina G. 2004. “The Evolution of Human Skin and Skin Color.” Annual Review of Anthropology 33: 585‒623. https://doi.org/10.1146/annurev.anthro.33.070203.143955.
Jablonski, Nina G., and George Chaplin. 2000. “The Evolution of Human Skin Coloration.” Journal of Human Evolution 39 (1): 57‒106. https://doi.org/10.1006/jhev.2000.0403.
Kanitz, Ricardo, Elsa G. Guillot, Sylvain Antoniazza, Samuel Neuenschwander, and Jérôme Gedout. 2018. “Complex Genetic Patterns in Human Arise from a Simple Range-Expansion Model over Continental Landmasses.” PLoS ONE 13 (2): e0192460.
Kronenberg, Zev N., Ian T. Fiddes, David Gordon, Shwetha Murali, Stuart Cantsilieris, Olivia S. Meyerson, Jason G. Underwood, et al. 2018. “High-Resolution Comparative Analysis of Great Ape Genomes.” Science 360 (6393): eaar6343. https://doi.org/10.1126/science.aar6343.
Kuzawa, Christopher W., and Elizabeth Sweet. 2009. “Epigenetics and the Embodiment of Race: Development Origins of US Racial Disparities in Cardiovascular Health.” American Journal of Human Biology 21 (1) : 2‒15.
Lasisi, Tina, and Mark D. Shriver. 2018. “Focus on African Diversity Confirms Complexity of Skin Pigmentation Genetics.” Genomic Biology 19: 13.
Lewontin, Richard. 1972. “The Apportionment of Human Diversity.” In Evolutionary Biology, vol. 6, edited by Theodosius Dobzhansky, Max K. Hecht, and William C. Steere, 381‒398. New York: Springer.
Linnaeus, Carl. 1758. Systema Naturae. Stockholm: Laurentius Salvius. https://www.cabdirect.org/abstracts/20057000018.html.
Liu, Hua, Franck Prugnolle, Andrea Manica, and François Balloux. 2006. “A Geographically Explicit Genetic Model of Worldwide Human-Settlement History.” American Journal of Human Genetics 79 (2): 230‒237.
Livingstone, Frank B. 1962. “On the Nonexistence of Human Races.” Current Anthropology 3 (3): 279‒281.
Long, Jeffery C., and Rick A. Kittles. 2003. “Human Genetic Diversity and the Nonexistence of Biological Races.” Human Biology 75 (4): 449‒471.
Luzzatto, Lucio. 2012. “Sickle Cell Anaemia and Malaria.” Mediterranean Journal of Hematology and Infectious Diseases 4 (1). https://doi.org/10.4084/MJHID.2012.065.
Manica, Andrea, William Amos, François Balloux, and Tsunehiko Hanihara. 2007. “The Effect of Ancient Population Bottlenecks on Human Phenotypic Variation.” Nature 448 (7151): 346‒348. https://doi.org/10.1038/nature05951.
McLean, Shay-Akil. 2014. “‘Race, Ethnicity, & Racism.” Decolonize ALL The Things Website, Accessed January 10, 2023. https://decolonizeallthethings.com/learning-tools/race-ethnicity-racism/.
Morton, Samuel George. 1839. Crania Americana, or, A Comparative View of the Skulls of Various Aboriginal Nations of North and South America. Philadelphia: J. Dobson.
Mourant, A. E., Ada C. Kopeć, and Kazimiera Domaniewska-Sobczak. 1976. The Distribution of the Human Blood Groups and Other Polymorphisms, 2nd edition. Oxford: Oxford University Press.
National Research Council (U.S.) Committee on Human Genome Diversity. 1997. Evaluating Human Genetic Diversity. Washington, D.C.: National Academies Press.
Omi, Michael, and Howard Winant. 2014. “The Theory of Racial Formation.” In Racial Formation in the United States,3rd edition, edited by Michael Omi and Howard Winant, 105‒126. Routledge: New York.
Osada, Naoki. 2015. “Genetic Diversity in Humans and Non-Human Primates and Its Evolutionary Consequences.” Genes and Genetic Systems 90 (3): 133‒145.
Ousley, Stephen D., Richard L. Jantz, and Donna Freid. 2009. “Understanding Race and Human Variation: Why Forensic Anthropologists Are Good at Identifying Race.” American Journal of Physical Anthropology 139 (1): 68‒76. https://doi.org/10.1002/ajpa.21006.
Ponce de León, Marcia S., Toetik Koesbardiati, John David Weissmann, Marco Millela, Carlos S. Reyna-Blanco, Gen Suwa, Osamu Kondo, Anna-Sapfo Malaspinas, Tim D. White, and Christoph P. E. Zollikofer. 2018. “Human Bony Labyrinth Is an Indicator of Population History and Dispersal from Africa.” Proceedings of the National Academy of Sciences 115 (16): 4128‒4133. https://doi.org/10.1073/pnas.1808125115.
Prado-Martinez, Javier, Peter H. Sudmant, Jeffrey M. Kidd, Heng Li, Joanna L. Kelley, Belen Lorente-Galdos, Krishna R. Veeramah, et al. 2013. “Great Ape Genetic Diversity and Population History.” Nature 499 (7459): 471–475. https://doi.org/10.1038/nature12228.
Prugnolle, Franck, Andrea Manica, and François Balloux. 2005. “Geography Predicts Neutral Genetic Diversity of Human Populations.” Current Biology 15 (5): 159‒160.
Quillen, Ellen E., Heather L. Norton, Esteban J. Parra, Frida Lona-Durazo, Khai C. Ang, Florin Mircea Illiescu, Laurel N. Pearson, et al. 2018. “Shades of Complexity: New Perspectives on the Evolution and Genetic Architecture of Human Skin.” American Journal of Physical Anthropology 168 (S67): 4–26.
Rathmann, Hannes, Hugo Reyes-Centeno, Silvia Ghirotto, Nicole Creanza, Tsunehiko Hanihara, and Katerina Harvati. 2017. “Reconstructing Human Population History from Dental Phenotypes.” Scientific Reports 7: 12495. https://doi.org/10.1038/s41598-017-12621-y.
Relethford, John H. 2001. “Global Analysis of Regional Differences in Craniometric Diversity and Population Substructure.” Human Biology 73 (5): 629‒636. https://doi.org/10.1353/hub.2001.0073.
Relethford, John H. 2002. “Apportionment of Global Human Genetic Diversity Based on Craniometrics and Skin Color.” American Journal of Physical Anthropology 118 (4): 393‒398. https://doi.org/10.1002/ajpa.10079.
Relethford, John H. 2004. “Global Patterns of Isolation by Distance Based on Genetic and Morphological Data.” Human Biology 76 (4): 499‒513. https://doi.org/10.1353/hub.2004.0060.
Relethford, John H. 2009. “Race and Global Patterns of Phenotypic Variation.” American Journal of Physical Anthropology 139 (1): 16‒22. https://doi.org/10.1002/ajpa.20900.
Roberts, Dorothy. 2013. Fatal Invention: How Science, Politics, and Big Business Re-Create Race in the Twenty-First Century. New York: The New Press.
Rosenberg, Noah A., Saurabh Mahajan, Sohini Ramachandran, Chengfeng Zhao, Jonathan K. Pritchard, and Marcus W. Feldman. 2005. “Clines, Clusters, and the Effect of Study Design on the Inference of Human Population Structure.” PLoS Genetics 1 (6): e70. https://doi.org/10.1371 /journal.pgen.0010070.
Rosenberg, Noah A., Jonathan K. Pritchard, James L. Weber, Howard M. Cann, Kenneth K. Kidd, Lev A. Zhivotovsky, and Marcus W. Feldman. 2002. “Genetic Structure of Human Populations.” Science 298 (5602): 2381‒2385.
Sauer, Norman J. 1992. “Forensic Anthropology and the Concept of Race: If Races Don’t Exist, Why Are Forensic Anthropologists So Good at Identifying Them?” Social Science and Medicine 34 (2): 107‒111. https://doi.org/10.1016/0277-9536(92)90086-6.
Shonkoff, Jack P., Natalie Slopen, and David R. Williams. 2021. “Early Childhood Adversity, Toxic Stress, and the Impacts of Racism on the Foundations of Health.” Annual Review of Public Health 42: 115‒134.
Sjoding, Michael W., Robert P. Dickson, Theodore J. Iwashyna, Steven E. Gay, and Thomas S. Valley. 2020. “Racial Bias in Pulse Oximetry Measurement.” The New England Journal of Medicine 383: 2477-2478.
Staes, Nicky, Chet C. Sherwood, Katharine Wright, Marc de Manuel, Elaine E. Guevara, Tomas Marques-Bonet, Michael Krützen, et al. 2017. “FOXP2 Variation in Great Ape Populations Offers Insight into the Evolution of Communication Skills.” Scientific Reports 7 (1): 1‒10. https://doi.org/10.1038/s41598-017-16844-x.
Tomati, Opal. 2016. “An Interview with the Founders of Black Lives Matter.” TED Talks 2016, October 26‒28. Accessed June 15, 2023. https://www.ted.com/talks/alicia_garza_patrisse_cullors_and_opal_tometi_an_interview_with_the_founders_of_black_lives_matter/up-next.
Tsai, Jennifer. 2021. “COVID-19 Is Not a Story of Race, but a Record of Racism—Our Scholarship Should Reflect That Reality.” The American Journal of Bioethics 21 (2): 43‒47. https://doi.org/10.1080/15265161.2020.1861377.
von Cramon-Taubadel, Noreen, and Stephen J. Lycett. 2008. “Brief Communication: Human Cranial Variation Fits Iterative Founder Effect Model with African Origin.” American Journal of Physical Anthropology 136 (1): 108‒113. https://doi.org/10.1002/ajpa.20775.
Weiss, Kenneth M., and Jeffrey C. Long. 2009. “Non-Darwinian Estimation: My Ancestors, My Genes’ Ancestors.” Genome Research 19: 703‒710. https://doi.org/10.1101/gr.076539.108.19.
Williams, David W. 2018. “Stress and the Mental Health of Populations of Color: Advancing Our Understanding of Race-related Stressors.” Journal of Health and Social Behavior 59 (4): 466‒485.
Wise, Tim. 2010. Colorblind: The Rise of Post-Racial Politics and the Retreat from Racial Equity. San Francisco: City Lights.
Yudell, Michael, Dorothy Roberts, Rob DeSalle, and Sarah Tishkoff. 2016. “Taking Race out of Human Genetics.” Science 351 (6273): 564‒565. https://doi.org/10.1126/science.aac4951.
Jonathan M. G. Perry, Ph.D., Western University of Health Sciences
Stephanie L. Canington, Ph.D., University of Pennsylvania
This chapter is a revision from "Chapter 8: Primate Evolution” by Jonathan M. G. Perry and Stephanie L. Canington. In Explorations: An Open Invitation to Biological Anthropology, first edition, edited by Beth Shook, Katie Nelson, Kelsie Aguilera, and Lara Braff, which is licensed under CC BY-NC 4.0.
Learning Objectives
- Understand the major trends in primate evolution from the origin of primates to the origin of our own species.
- Learn about primate adaptations and how they characterize major primate groups.
- Discuss the kinds of evidence that anthropologists use to find out how extinct primates are related to each other and to living primates.
- Recognize how the changing geography and climate of Earth have influenced where and when primates have thrived or gone extinct.
The first fifty million years of primate evolution was a series of adaptive radiations leading to the diversification of the earliest lemurs, monkeys, and apes. The primate story begins in the canopy and understory of conifer-dominated forests, with our small, furtive ancestors subsisting at night, beneath the notice of day-active dinosaurs.
From the ancient plesiadapiforms (archaic primates) to the earliest groups of true primates (euprimates) (Bloch and Boyer 2002), the origin of our own order is characterized by the struggle for new food sources and microhabitats in the arboreal setting. Climate change forced major extinctions as the northern continents became increasingly dry, cold, and seasonal and as tropical rainforests gave way to deciduous forests, woodlands, and eventually grasslands. Lemurs, lorises, and tarsiers—once diverse groups containing many species—became rare, except for lemurs in Madagascar, where there were no anthropoid competitors and perhaps few predators. Meanwhile, anthropoids (monkeys and apes) likely emerged in Asia and then dispersed across parts of the northern hemisphere, Africa, and ultimately South America. The movement of continents, shifting sea levels, and changing patterns of rainfall and vegetation contributed to the developing landscape of primate biogeography, morphology, and behavior. Today’s primates provide modest reminders of the past diversity and remarkable adaptations of their extinct relatives. This chapter explores the major trends in primate evolution from the origin of the Order Primates to the beginnings of our own lineage, providing a window into these stories from our ancient past.
Major Hypotheses About Primate Origins
For many groups of mammals, there is a key feature that led to their success. A good example is powered flight in bats. Primates lack a feature like this (see Chapter 5). Instead, if there is something unique about primates, it is probably a group of features rather than one single thing. Because of this, anthropologists and paleontologists struggle to describe an ecological scenario that could explain the rise and success of our own order. Three major hypotheses have been advanced to consider the origin of primates and to explain what makes our order distinct among mammals (Figure 9.1); these are described below.
Arboreal Hypothesis
In the 1800s, many anthropologists viewed all animals in relation to humans. That is, animals that were more like humans were considered to be more “advanced” and those lacking humanlike features were considered more “primitive.” This way of thinking was particularly obvious in studies of primates. A more modern way of referring to members of a group that lack certain evolutionary innovations seen in other members is to call them plesiomorphic (literally “anciently shaped”). The state of their morphological features is sometimes referred to as ancestral traits.
Thus, when anthropologists sought features that separate primates from other mammals, they focused on features that were least developed in lemurs and lorises, more developed in monkeys, and most developed in apes (Figure 9.2). Frederic Wood Jones, one of the leading anatomist-anthropologists of the early 1900s, is usually credited with the Arboreal Hypothesis of primate origins (Jones 1916). This hypothesis holds that many of the features of primates evolved to improve locomotion in the trees; this way of getting around is referred to as arboreal. For example, the grasping hands and feet of primates are well suited to gripping tree branches of various sizes and our flexible joints are good for reorienting the extremities in many different ways. A mentor of Jones, Grafton Elliot Smith, had suggested that the reduced olfactory system, acute vision, and forward-facing eyes of primates are adaptations for making accurate leaps and bounds through a complex, three-dimensional canopy (Smith 1912). The forward orientation of the eyes in primates causes the visual fields to overlap, enhancing depth perception, especially at close range. Evidence to support this hypothesis includes the facts that many extant primates are arboreal, and the plesiomorphic members of most primate groups are dedicated arborealists. The Arboreal Hypothesis was well accepted by most anthropologists at the time and for decades afterward.
Visual Predation Hypothesis
In the late 1960s and early 1970s, Matt Cartmill studied and tested the idea that the characteristic features of primates evolved in the context of arboreal locomotion. Cartmill noted that squirrels climb trees (and even vertical walls) very effectively, even though they lack some of the key adaptations of primates. As members of the Order Rodentia, squirrels also lack the hand and foot anatomy of primates. They have claws instead of flattened nails and their eyes face more laterally than those of primates. Cartmill reasoned that there must be some other explanation for the unique traits of primates. He noted that some nonarboreal animals share at least some of these traits with primates; for example, cats and predatory birds have forward-facing eyes that enable visual field overlap. Cartmill suggested that the unique suite of features in primates is an adaptation to detecting insect prey and guiding the hands (or feet) to catch insects (Cartmill 1972). His hypothesis emphasizes the primary role of vision in prey detection and capture; it is explicitly comparative, relying on form-function relationships in other mammals and nonmammalian vertebrates. According to Cartmill, many of the key features of primates evolved for preying on insects in this special manner (Cartmill 1974).
Angiosperm-Primate Coevolution Hypothesis
The visual predation hypothesis was unpopular with some anthropologists. One reason for this is that many primates today are not especially predatory. Another is that, whereas primates do seem well adapted to moving around in the smallest, terminal branches of trees, insects are not necessarily easier to find there. A counterargument to the visual predation hypothesis is the angiosperm-primate coevolution hypothesis. Primate ecologist Robert Sussman (Sussman 1991) argued that the few primates that eat mostly insects often catch their prey on the ground rather than in tree branches. Furthermore, predatory primates often use their ears more than their eyes to detect prey. Finally, most early primate fossils show signs of having been omnivorous rather than insectivorous. Instead, he argued, the earliest primates were probably seeking fruit. Fruit (and flowers) of angiosperms (flowering plants) often develop in the terminal branches. Therefore, any mammal trying to access those fruits must possess anatomical traits that allow them to maintain their hold on thin branches and avoid falling while reaching for the fruits. Primates likely evolved their distinctive visual traits and extremities in the Paleocene (approximately 65 million to 54 million years ago) and Eocene (approximately 54 million to 34 million years ago) epochs, just when angiosperms were going through a revolution of their own—the evolution of large, fleshy fruit that would have been attractive to a small arboreal mammal. Sussman argued that, just as primates were evolving anatomical traits that made them more efficient fruit foragers, angiosperms were also evolving fruit that would be more attractive to primates to promote better seed dispersal. This mutually beneficial relationship between the angiosperms and the primates was termed coevolution or more specifically diffuse coevolution.
At about the same time, D. Tab Rasmussen noted several parallel traits in primates and the South American woolly opossum, Caluromys. He argued that early primates were probably foraging on both fruits and insects (Rasmussen 1990). As is true of Caluromys today, early primates probably foraged for fruits in the terminal branches of angiosperms, and they probably used their visual sense to aid in catching insects. Insects are also attracted to fruit (and flowers), so these insects represent a convenient opportunity for a primarily fruit-eating primate to gather protein. This solution is a compromise between the visual predation hypothesis and the angiosperm-primate coevolution hypothesis. It is worth noting that other models of primate origins have been proposed, and these include the possibility that no single ecological scenario can account for the origin of primates.
The Origins of Primates
Paleocene: Mammals in the Wake of Dinosaur Extinctions
Placental mammals, including primates, originated in the Mesozoic Era (approximately 251 million to 65.5 million years ago), the Age of Dinosaurs. During this time, most placental mammals were small, probably nocturnal, and probably avoided predators via camouflage and slow, quiet movement. It has been suggested that the success and diversity of the dinosaurs constituted a kind of ecological barrier to Mesozoic mammals. The extinction of the dinosaurs (and many other organisms) at the end of the Cretaceous Period (approximately 145.5–65.5 million years ago) might have opened up these ecological niches, leading to the increased diversity and disparity in mammals of the Tertiary Period (approximately 65.5–2.5 million years ago).
The Paleocene was the first epoch in the Age of Mammals. Soon after the Cretaceous-Tertiary (K-T) extinction event, new groups of placental mammals appear in the fossil record. Many of these groups achieved a broad range of sizes and lifestyles as well as a great number of species before declining sometime in the Eocene (or soon thereafter). These groups were ultimately replaced by the modern orders of placental mammals (Figure 9.3). It is unknown whether these replacements occurred gradually, for example by competitive exclusion, or rapidly, perhaps by sudden geographic dispersals with replacement. In some senses, the Paleocene might have been a time of recovery from the extinction event; it was cooler and more seasonal globally than the subsequent Eocene.

Plesiadapiforms, the Archaic Primates
The Paleocene epoch saw the emergence of several families of mammals that have been implicated in the origin of primates. These are the plesiadapiforms, which are archaic primates, meaning they possessed some primate features and lacked others. The word plesiadapiform means “almost adapiform,” a reference to some similarities between some plesiadapiforms and some adapiforms (or adapoids; later-appearing true primates)—mainly in the molar teeth. Because enamel fossilizes better than other parts of the body, the molar teeth are the parts most often found and first discovered for any new species. Thus, dental similarities were often the first to be noticed by early mammalian paleontologists, partly explaining why plesiadapiforms were thought to be primates. Major morphological differences between plesidapiforms and euprimates (true primates) were observed later when more parts of plesiadapiform skeletons were discovered. Many plesiadapiforms have unusual anterior teeth and most have digits possessing claws rather than nails. So far, no plesiadapiform ever discovered has a postorbital bar (seen in extant strepsirrhines) or septum (as seen in haplorhines), and whether or not the auditory bulla was formed by the petrosal bone remains unclear for many plesiadapiform specimens. Nevertheless, there are compelling reasons (partly from new skeletal material) for including plesiadapiforms within the Order Primates.
Geographic and Temporal Distribution
Purgatorius is generally considered to be the earliest primate. This Paleocene mammal is known from teeth that are very plesiomorphic for a primate. It has some characteristics that suggest it is a basal plesiadapiform, but there is very little to link it specifically with euprimates (see Clemens 2004). Its ankle bones suggest a high degree of mobility, signaling an arboreal lifestyle (Chester et al. 2015). Purgatorius is plesiomorphic enough to have given rise to all primates, including the plesiadapiforms. However, new finds suggest that this genus was more diverse and had more differing tooth morphologies than previously appreciated (Wilson Mantilla et al. 2021). Plesiadapiform families were numerous and diverse during parts of the Paleocene in western North America and western Europe, with some genera (e.g., Plesiadapis; see Figure 9.4) living on both continents (Figure 9.5). Thus, there were probably corridors for plesiadapiform dispersal between the two continents, and it stands to reason that these mammals were living all across North America, including in the eastern half of the continent and at high latitudes. A few plesiadapiforms have been described from Asia (e.g., Carpocristes), but the affinities of these remain uncertain.
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Paromomyidae |
Ignacius |
Long, dagger-like, lower incisor. |
North America and Europe |
Early Paleocene to Late Eocene |
|
Carpolestidae |
Carpolestes |
Plagiaulacoid dentition. Limb adaptations to terminal branch feeding. Grasping big toe. |
North America, Europe, and Asia |
Middle Paleocene to Early Eocene |
|
Plesiadapidae |
Plesiadapis |
Mitten-like upper incisor. Diastema. Large body size for group. |
North America and Europe |
Middle Paleocene to Early Eocene |
|
1 Derived from Fleagle 2013. |
|
|||
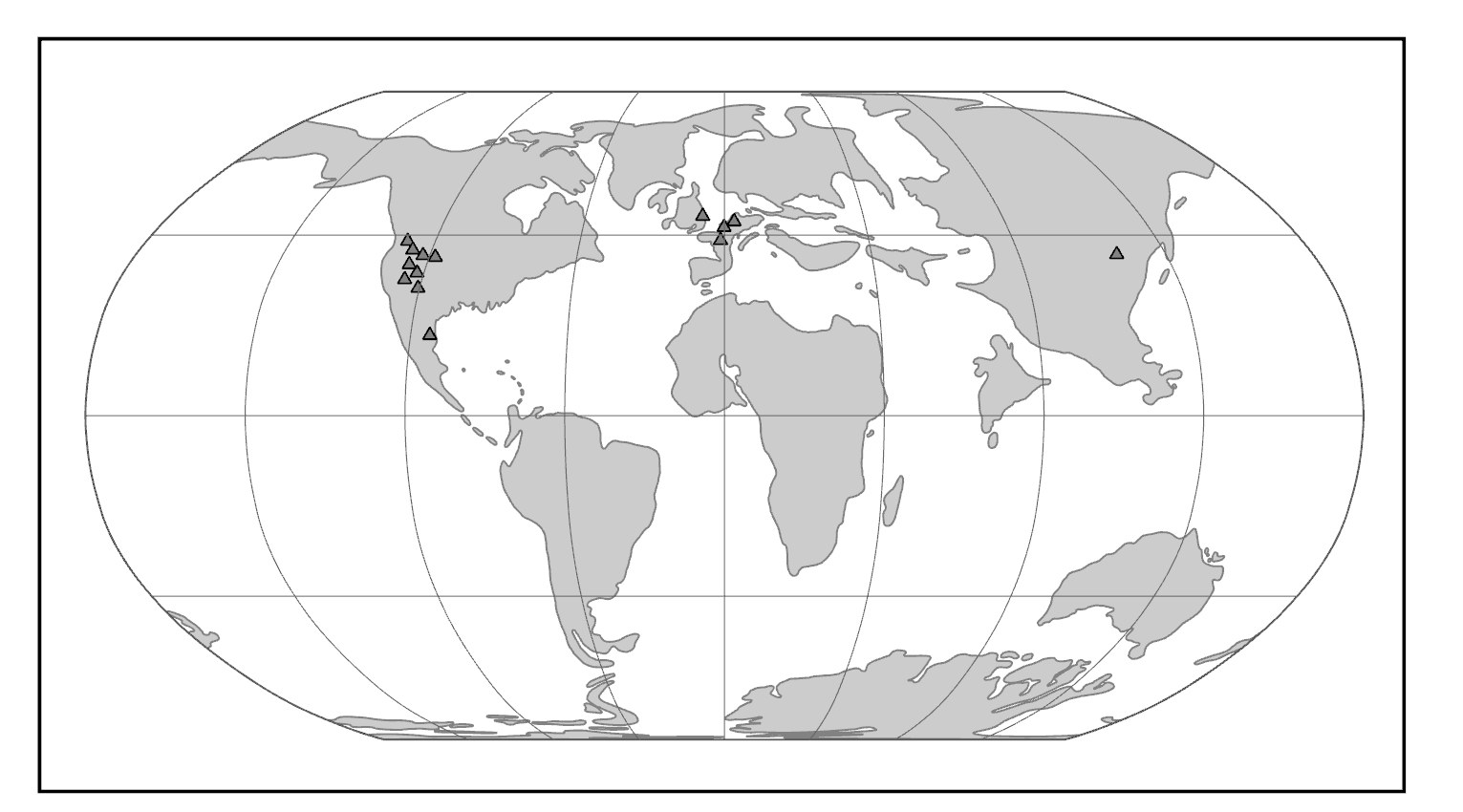
General Morphological Features
Although there is much morphological variation among the families of plesiadapiforms, some common features unite the group. Most plesiadapiforms were small, the largest being about three kilograms (approximately 7 lbs.; Plesiadapis cookei). They had small brains and fairly large snouts, with eyes that faced more laterally than in euprimates. Many species show reduction and/or loss of the canine and anterior premolars, with the resulting formation of a rodent-like diastema (a pronounced gap between the premolars and the incisors, with loss of at least the canine); this probably implies a herbivorous diet. Some families appear to have had very specialized diets, as suggested by unusual tooth and jaw shapes.
Arguably the most interesting and unusual family of plesiadapiforms is the Carpolestidae. They are almost exclusively from North America (with a couple of possible members from Asia), and mainly from the Middle and Late Paleocene. Their molars are not very remarkable, being quite similar to those of some other plesiadapiforms (e.g., Plesiadapidae). However, their lower posterior premolars (p4) are laterally compressed and blade-like with vertical serrations topped by tiny cuspules. This unusual dental morphology is termed plagiaulacoid (Simpson 1933). The upper premolar occlusal surfaces are broad and are covered with many small cuspules; the blade-like lower premolar might have cut across these cuspules, between them, or both.

Many plesiadapiforms have robust limb bones with hallmarks of arboreality. Instead of having nails, most taxa had sharp claws on most or all of the digits. The extremities show grasping abilities comparable to those of primates and some arboreal marsupials. Nearly complete skeletons have yielded a tremendous wealth of information on locomotor and foraging habits. Many plesiadapiforms appear to have been able to cling to vertical substrates (like a broad tree trunk) using their sharp claws, propelling themselves upward using powerful hindlimbs, bounding along horizontal supports, grasping smaller branches, and moving head-first down tree trunks. In carpolestids in particular, the skeleton appears to have been especially well adapted to moving slowly and carefully in small terminal branches (Figure 9.6).
Dig Deeper: Debate: Relationship of Plesiadapiforms to True Primates
In the middle of the twentieth century, treeshrews (Order Scandentia) were often considered part of the Order Primates, based on anatomical similarities between some treeshrews and primates. For many people, plesiadapiforms represented intermediates between primates and treeshrews, so plesiadapiforms were included in Primates as well.
Studies of reproduction and brain anatomy in treeshrews and lemurs suggested that treeshrews are not primates (e.g., Martin 1968). This was soon followed by the suggestion to also expel plesiadapiforms (Martin 1972) from the Order Primates. Like treeshrews, plesiadapiforms lack a postorbital bar, nails, and details of the ear region that characterize true primates. Many paleoanthropologists were reluctant to accept this move to banish plesiadapiforms (e.g., F. S. Szalay, P. D. Gingerich).
Later, K. Christopher Beard (1990) found that in some ways, the digits of paromomyid plesiadapiforms are actually more similar to those of dermopterans (Order Dermoptera), the closest living relatives of primates, than they are to those of primates themselves (but see Krause 1991). At the same time, Richard Kay and colleagues (1990) found that cranial circulation patterns and auditory bulla morphology in the paromomyid, Ignacius (see Figure 9.4), are more like those of dermopterans than of primates.
For many anthropologists, this one-two punch effectively removed plesiadapiforms from the Order Primates. In the last two decades, the tide of opinion has turned again, with many researchers reinstating plesiadapiforms as members of the Order Primates. New and more complete specimens demonstrate that the postcranial skeletons of plesiadapiforms, including the hands and feet, were primate-like, not dermorpteran-like (Bloch and Boyer 2002, 2007). New fine-grained CT scans of relatively complete plesiadapiform skulls revealed that they share some key traits with primates to the exclusion of other placental mammals (Bloch and Silcox 2006). Most significant was the suggestion that Carpolestes simpsoni possessed an auditory bulla formed by the petrosal bone, like in all living primates.
The debate about the status of plesiadapiforms continues, owing to a persistent lack of key bones in some species and owing to genuine complexity of the anatomical traits involved. Maybe plesiadapiforms were the ancestral stock from which all primates arose, with some plesiadapiforms (e.g., carpolestids) nearer to the primate stem than others.
Adapoids and Omomyoids, the First True Primates
Geographic and Temporal Distribution
The first universally accepted fossil primates are the adapoids (Superfamily Adapoidea) and the omomyoids (Superfamily Omomyoidea). These groups become quite distinct over evolutionary time, filling mutually exclusive niches for the most part. However, the earliest adapoids are very similar to the earliest omomyoids.
The adapoids were mainly diurnal and herbivorous, with some achieving larger sizes than any plesiadapiforms (10 kg; 22 lbs.). By contrast, the omomyoids were mainly nocturnal, insectivorous and frugivorous, and small.
Both groups appear suddenly at the start of the Eocene, where they are present in western North America, western Europe, and India (Figure 9.7). This wide dispersal of early primates was probably due to the presence of rainforest corridors extending far into northern latitudes.
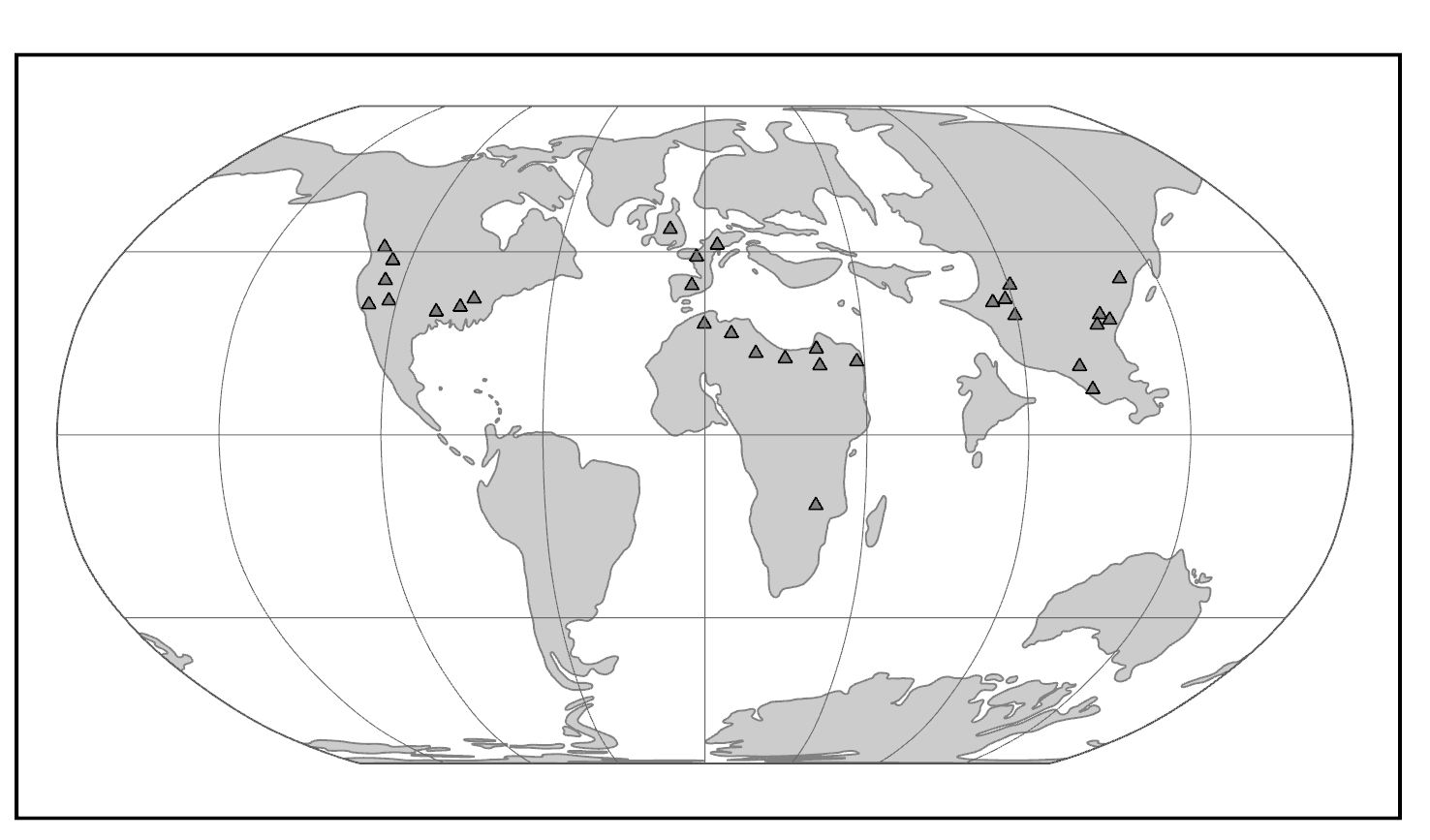
In North America and Europe, both groups achieved considerable diversity in the Middle Eocene, then mostly died out at the end of that epoch (Figure 9.8). In some Eocene rock formations in the western United States, adapoids and omomyoids make up a major part of the mammalian fauna. The Eocene of India has yielded a modest diversity of euprimates, some of which are so plesiomorphic that it is difficult to know whether they are adapoids or omomyoids (or even early anthropoids).
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Cercamoniidae |
Donrussellia |
Variable in tooth number and jaw shape. |
Europe and Asia |
Early to Late Eocene |
|
Asiadapidae2 |
Asiadapis |
Plesiomorphic teeth and jaw resemble early Omomyids. |
Asia |
Early Eocene |
|
Caenopithecidae3 |
Darwinius |
Robust jaws with crested molars. Fewer premolars. |
Europe, Africa, North America, and Asia |
Middle to Late Eocene |
|
Adapidae |
Adapis |
Fused mandible. Long molar crests. Large size and large chewing muscles. |
Europe |
Late Eocene to Early Oligocene |
|
Sivaladapidae |
Sivaladapis |
Some large with robust jaws. |
Asia |
Middle Eocene to Late Miocene |
|
Notharctidae4 |
Notharctus |
Canine sexual dimorphism. Lemur-like skull. Clinging and leaping adaptations. |
North America and Europe |
Early to Middle Eocene |
|
Omomyidae5 |
Teilhardina |
Small, nocturnal, frugivorous or insectivorous. Tarsier-like skull in some. |
North America, Europe, and Asia |
Early Eocene to Early Miocene |
|
Microchoeridae6 |
Necrolemur |
Long bony ear tubes. Tarsier-like lower limb adaptations for leaping. |
Europe and Asia |
Early Eocene to Early Oligocene |
|
1 Derived from Fleagle 2013. 2 See Dunn et al. 2016 and Rose et al. 2018. 3 See Kirk and Williams 2011 and Seiffert et al. 2009. 4 See Gregory 1920. 5 See Beard and MacPhee 1994 and Strait 2001. 6 See Schmid 1979. |
|
|||
Adapoids and omomyoids barely survived the Eocene-Oligocene extinctions, when colder temperatures, increased seasonality, and the retreat of rainforests to lower latitudes led to changes in mammalian biogeography. In North America, one genus (originally considered an omomyoid but recently reclassified as Adapoidea) persisted until the Miocene: Ekgmowechashala (Rose and Rensberger 1983). This taxon has highly unusual teeth and might have been a late immigrant to North America from Asia. In Asia, one family of adapoids, the Sivaladapidae, retained considerable diversity as late as the Late Miocene.
Adapoid Diversity
Adapoids were very diverse, particularly in the Eocene of North America and Europe. They can be divided into six families, with a few species of uncertain familial relationship. As a group, adapoids have some features in common, although much of what they share is plesiomorphic. Important features include the hallmarks of euprimates: postorbital bar, flattened nails, grasping extremities, and a petrosal bulla (Figures 9.9 and 9.10). In addition, some adapoids retain the ancestral dental formula of 2.1.4.3; that is, in each quadrant of the mouth, there are two incisors, one canine, four premolars, and three molars. In general, the incisors are small compared to the molars, but the canines are relatively large, with sexual dimorphism in some species. Cutting crests on the molars are well developed in some species, and the two halves of the mandible were fused at the midline in some species. Some adapoids were quite small (Anchomomys at a little over 100 g), and some were quite large (Magnadapis at 10 kg; 22 lbs.). Furthermore, the spaces and attachment features for the chewing muscles were truly enormous in some species, suggesting that these muscles were very large and powerful. Taken together, this suggests an overall adaptive profile of diurnal herbivory. The canine sexual dimorphism in some species suggests a possible mating pattern of polygyny, as males in polygynous primate species often compete with each other for mates and have especially large canine teeth.


Omomyoid Diversity
Like adapoids, omomyoids appeared suddenly at the start of the Eocene and then became very diverse with most species dying out before the Oligocene. Omomyoids are known from thousands of jaws with teeth, relatively complete skulls for about a half-dozen species, and very little postcranial material. Omomyoids were relatively small primates, with the largest being less than three kilograms (approximately 7 lbs.; Macrotarsius montanus). All known crania possess a postorbital bar, which in some has been described as “incipient closure.” Some—but not all—known crania have an elongated bony ear tube extending lateral to the location of the eardrum, a feature seen in living tarsiers and catarrhines. The anterior teeth tend to be large, with canines that are usually not much larger than the incisors. Often it is difficult to distinguish closely related species using molar morphology, but the premolars tend to be distinct from one species to another. The postcranial skeleton of most omomyoids shows hallmarks of leaping behavior reminiscent of that of tarsiers. In North America, omomyoids became very diverse and abundant. In fact, omomyoids from Wyoming are sufficiently abundant and from such stratigraphically controlled conditions that they have served as strong evidence for the gradual evolution of anatomical traits over time (Rose and Bown 1984).
Teilhardina (Figure 9.11; see Figure 9.2) is one of the earliest and arguably the most plesiomorphic of omomyoids. Teilhardina has several species, most of which are from North America, with one from Europe (T. belgica) and one from Asia (T. asiatica). The species of this genus are anatomically similar and the deposits from which they are derived are roughly contemporaneous. Thus, this small primate likely dispersed across the northern continents very rapidly (Smith et al. 2006).
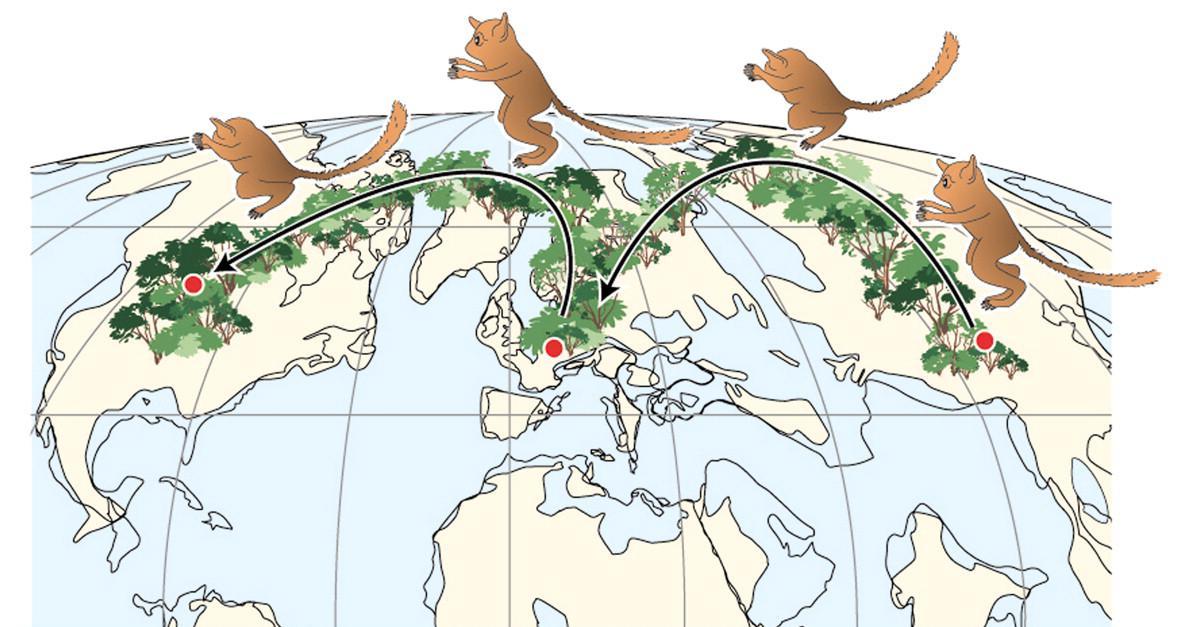
The Emergence of Modern Primate Groups
Origins of Crown Strepsirrhines
Until the turn of this century, very little was known about the origins of the crown (living) strepsirrhines. The Quaternary record of Madagascar contains many amazing forms of lemurs, including giant sloth-like lemurs, lemurs with perhaps monkey-like habits, lemurs with koala-like habits, and even a giant aye-aye (Godfrey and Jungers 2002). However, in Madagascar, early Tertiary continental sediments are lacking, and there is no record of lemur fossils before the Pleistocene.
The fossil record of galagos is slightly more informative. Namely, there are Miocene African fossils that are very likely progenitors of lorisids (Simpson 1967). However, these are much like modern galagos and do not reveal anything about the relationship between crown strepsirrhines and Eocene fossil primates (but see below regarding Propotto). A similar situation exists for lorises in Asia: there are Miocene representatives, but these are substantially like modern lorises. The discovery of the first definite toothcomb canine (a hallmark of stresirrhines) in 2003 provided the “smoking gun” for the origin of crown strepsirrhines (Seiffert et al. 2003). Recently, several other African primates have been recognized as having strepsirrhine affinities (Marivaux et al. 2013; Seiffert 2012). The enigmatic Fayum primate Plesiopithecus is known from a skull that has been compared to aye-ayes and to lorises (Godinot 2006; Simons and Rasmussen 1994a).
The now-recognized diversity of stem strepsirrhines from the Eocene and Oligocene of Afro-Arabia is strong evidence to suggest that strepsirrhines originated in that geographic area. This implies that lorises dispersed to Asia subsequent to an African origin. It is unknown what the first strepsirrhines in Madagascar were like. However, it seems likely that the lemuriform-lorisiform split occurred in continental Africa, followed by dispersal of lemuriform stock to Madagascar. Recent evidence suggests that Propotto, a Miocene primate from Kenya originally described as a potto antecedent, actually forms a clade with Plesiopithecus and the aye-aye; this might suggest that strepsirrhines dispersed to Madagascar from continental Africa more than once (Gunnell et al. 2018).
The Fossil Record of Tarsiers
Tarsiers are so unusual that they fuel major debates about primate taxonomy. Tarsiers today are moderately diverse but geographically limited and not very different in their ecological habits—especially considering that the split between them and their nearest living relative probably occurred over 50 million years ago. If omomyoids are excluded, then the fossil record of tarsiers is very limited. Two fossil species from the Miocene of Thailand have been placed in the genus Tarsius, as has an Eocene fossil from China (Beard et al. 1994). These, and Xanthorhysis from the Eocene of China, are all very tarsier-like. In fact, it is striking that Tarsius eocaenus from China was already so tarsier-like as early as the Eocene. This suggests that tarsiers achieved their current morphology very early in their evolution and have remained more or less the same while other primates changed dramatically. Two additional genera, Afrotarsius from the Oligocene of Egypt and Libya and Afrasia from the Eocene of Myanmar, have also been implicated in tarsier origins, though the relationship between them and tarsiers is unclear (Chaimanee et al. 2012). More recently, a partial skeleton of a small Eocene primate from China, Archicebus achilles (dated to approximately 55.8 million to 54.8 million years ago), was described as the most basal tarsiiform (Ni et al. 2013). This primate is reconstructed as a diurnal insectivore and an arboreal quadruped that did some leaping—but not to the specialized degree seen in living tarsiers. The anatomy of the eye in living tarsiers suggests that their lineage passed through a diurnal stage, so Archicebus (and diurnal omomyoids) might represent such a stage.
Climate Change and the Paleogeography of Modern Primate Origins
Changing global climate has had profound effects on primate dispersal patterns and ecological habits over evolutionary time. Primates today are strongly tied to patches of trees and particular plant parts such as fruits, seeds, and immature leaves. It is no surprise, then, that the distribution of primates mirrors the distribution of forests. Today, primates are most diverse in the tropics, especially in tropical rainforests. Global temperature trends across the Tertiary have affected primate ranges. Following the Cretaceous-Tertiary extinction event, cooler temperatures and greater seasonality characterized the Paleocene. In the Eocene, temperatures (and probably rainfall) increased globally and rainforests likely extended to very high latitudes. During this time, euprimates became diverse. With cooling and increased aridity at the end of the Eocene, many primate extinctions occurred in the northern continents and the surviving primates were confined to lower latitudes in South America, Afro-Arabia, Asia, and southern Europe. Among these survivors are the progenitors of the living groups of primates: lemurs and lorises, tarsiers, platyrrhines (monkeys of the Americas), and catarrhines (monkeys and apes of Africa and Asia) (Figure 9.12).
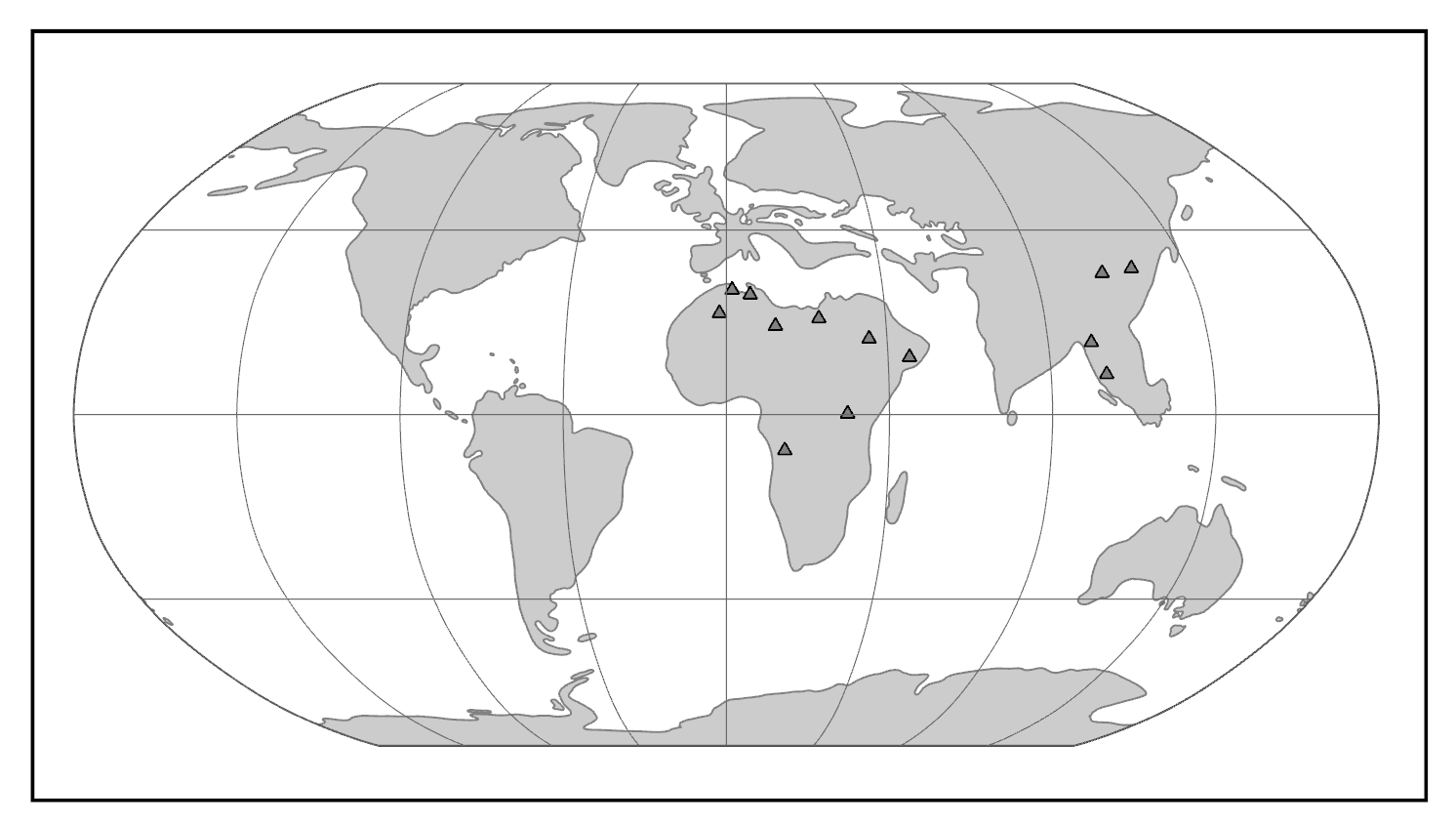
Competing Hypotheses for the Origin of Anthropoids
There is considerable debate among paleoanthropologists as to the geographic origins of anthropoids. In addition, there is debate regarding the source group for anthropoids. Three different hypotheses have been articulated in the literature. These are the adapoid origin hypothesis, the omomyoid origin hypothesis, and the tarsier origin hypothesis (Figure 9.13).
Adapoid Origin Hypothesis
Resemblances between some adapoids and some extant anthropoids include fusion of the mandibular symphysis, overall robusticity of the chewing system, overall large body size, features that signal a diurnal lifestyle (like relatively small eye sockets), and ankle bone morphology. Another feature in common is canine sexual dimorphism, which is present in some species of adapoids (probably) and in several species of anthropoids.
These features led some paleoanthropologists in the last half of the 20th century to suggest that anthropoids came from adapoid stock (Gingerich 1980; Simons and Rasmussen 1994b). One of the earliest supporters of the link between adapoids and anthropoids was Hans Georg Stehlin, who described much of the best material of adapoids and compared these Eocene primates to South American monkeys (Stehlin 1912). In more recent times, the adapoid origin hypothesis was reinforced by resemblances between these European adapoids (especially Adapis and Leptadapis) and some early anthropoids from the Fayum Basin (e.g., Aegyptopithecus, see below; Figure 9.14).
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Propliopithecidae2 |
Aegyptopithecus |
Large size. Cranial sexual dimorphism, large canines. Robust jaws and rounded molars. Partially ossified ear tube (in some). Robust skeleton; quadruped. |
Africa |
Late Eocene to Early Oligocene |
|
Parapithecidae3 |
Apidium |
Medium size. Retention of three premolars per quadrant. Rounded molars and premolars with large central cusps. Adaptations for leaping in the lower limb. |
Africa |
Late Eocene to Late Oligocene |
|
Proteopithecidae4 |
Proteopithecus |
Small size. Retention of three premolars per quadrant. Arboreal quadrupeds that ate fruit. |
Africa |
Late Eocene |
|
Oligopithecidae5 |
Catopithecus |
Small size. Skull has postorbital septum and unfused mandible. Deep jaws. Diet of fruits. Generalized quadruped. |
Africa |
Late Eocene |
|
Eosimiidae |
Eosimias |
Deep jaw with vertical unfused symphysis. Pointed incisors and canines. Crowded premolars. |
Asia |
Middle Eocene |
|
Amphipithecidae6 |
Pondaungia |
Deep jaws. Molars generally rounded with wide basins. |
Asia |
Middle Eocene to Early Oligocene |
|
1 Derived from Fleagle 2013. 2 See Gebo and Simons 1987 and Simons et al. 2007. 3 See Feagle and Simons 1995 and Simons 2001. 4 See Simons and Seiffert 1999. 5 See Simons and Rasmussen 1996. 6 See Kay et al. 2004. |
|
|||
Unfortunately for the adapoid hypothesis, most of the shared features listed above probably emerged independently in the two groups as adaptations to a diet of hard and/or tough foods. For example, fusion of the mandibular symphysis likely evolved as a means to strengthen the jaw against forces that would pull the two halves away from each other, in the context of active chewing muscles on both sides of the head generating great bite forces. This context would also favor the development of robust jaws, large chewing muscles, shorter faces, and some other features shared by some adapoids and some anthropoids.
As older and more plesiomorphic anthropoids were found in the Fayum Basin, it became clear that the earliest anthropoids from Africa did not possess these features of jaw robusticity (Seiffert et al. 2009). Furthermore, many adapoids never evolved these features. Fusion of the mandibular symphysis in adapoids is actually quite different from that in anthropoids and probably occurred during juvenile development in the former (Beecher 1983; Ravosa 1996). Eventually, the adapoid origin hypothesis fell out of favor among most paleoanthropologists, although the description of Darwinius is a recent revival of that idea (Franzen et al. 2009; but see Seiffert et al. 2009, Williams et al. 2010b).
Omomyoid Origin Hypothesis
Similarities in cranial and hindlimb morphology between some omomyoids and extant tarsiers have led to the suggestion that tarsiers arose from some kind of omomyoid. In particular, Necrolemur has many features in common with tarsiers, as does the North American Shoshonius, which is known from a few beautifully preserved (although distorted) crania. Tarsiers and Shoshonius share exclusively some features of the base of the cranium; however, Shoshonius does not have any sign of postorbital closure, and it lacks the bony ear tube of tarsiers. Nevertheless, some of the resemblances between some omomyoids and tarsiers suggest that tarsiers might have originated from within the Omomyoidea (Beard 2002; Beard and MacPhee 1994). In this scenario, although living tarsiers and living anthropoids might be sister taxa, they might have evolved from different omomyoids, possibly separated from each other by more than 50 million years of evolution, or from anthropoids evolved from some non-omomyoid fossil group. The arguments against the omomyoid origin hypothesis are essentially the arguments for the tarsier origin hypothesis (see below). Namely, tarsiers and anthropoids share many features (especially of the soft tissues) that must have been retained for many millions of years or must have evolved convergently in the two groups. Furthermore, a key hard-tissue feature shared between the two extant groups, the postorbital septum, was not present in any omomyoid. Therefore, that feature must have arisen convergently in the two extant groups or must have been lost in omomyoids. Neither scenario is very appealing, although recent arguments for convergent evolution of the postorbital septum in tarsiers and anthropoids have arisen from embryology and histology of the structure (DeLeon et al. 2016).
Tarsier Origin Hypothesis
Several paleoanthropologists have suggested that there is a relationship between tarsiers and anthropoids to the exclusion of omomyoids and adapoids (e.g., Cartmill and Kay 1978; Ross 2000; Williams and Kay 1995). Tarsiers and anthropoids today share several traits, including many soft-tissue features related to the olfactory system (e.g., the loss of a hairless external nose and loss of the median cleft running from the nose to the mouth, as possessed by strepsirrhines), and aspects of the visual system (e.g., the loss of a reflective layer at the back of the eye, similarities in carotid circulation to the brain, and mode of placentation). Unfortunately, none of these can be assessed directly in fossils. Some bony similarities between tarsiers and anthropoids include an extra air-filled chamber below the middle ear cavity, reduced bones within the nasal cavity, and substantial postorbital closure; these can be assessed in fossils, but the distribution of these traits in omomyoids does not yield clear answers. Furthermore, several similarities between tarsiers and anthropoids are probably due to similarities in sensory systems, which might have evolved in parallel for ecological reasons. Although early attempts to resolve the crown primates with molecular data were sometimes equivocal or in disagreement with one another, more recent analyses (including those of short interspersed elements) suggest that tarsiers and anthropoids are sister groups to the exclusion of lemurs and lorises (Williams et al. 2010a). However, this does not address omomyoids, all of which are far too ancient for DNA extraction.
The above three hypotheses are not the only possibilities for anthropoid origins. It may be that anthropoids are neither the closest sister group of tarsiers, nor evolved from adapoids or omomyoids. In recent years, two new groups of Eocene Asian primates have been implicated in the origin of anthropoids: the eosimiids and the amphipithecids. It is possible that one or the other of these two groups gave rise to anthropoids. Regardless of the true configuration of the tree for crown primates, the three major extant groups probably diverged from each other quite long ago (Seiffert et al. 2004).
Early Anthropoid Fossils in Africa


The classic localities yielding the greatest wealth of early anthropoid fossils are those from the Fayum Basin in Egypt (Simons 2008; Figure 9.15). The Fayum is a veritable oasis of fossil primates in an otherwise spotty early Tertiary African record. Since the 1960s, teams led by E. L. Simons have discovered several new species of early anthropoids, some of which are known from many parts of the skeleton and several individuals (Figure 9.16).
The Fayum Jebel Qatrani Formation and Birket Qarun Formation between them have yielded a remarkable array of terrestrial, arboreal, and aquatic mammals. These include ungulates, bats, sea cows, elephants, hyraces, rodents, whales, and primates. Also, many other vertebrates, like water birds, were present. The area at the time of deposition (Late Eocene through Early Oligocene) was probably very wet, with slow-moving rivers, standing water, swampy conditions, and lots of trees (see Bown and Kraus 1988). In short, it was an excellent place for primates.
General Morphology of Anthropoids
The anthropoids known from the Fayum (and their close relatives from elsewhere in East Africa and Afro-Arabia) bear many of the anatomical hallmarks of extant anthropoids; however, there are plesiomorphic forms in several families that lack one or more anthropoid traits. All Fayum anthropoids known from skulls possess postorbital closure, most had fused mandibular symphyses, and most had ring-like ectotympanic bones. Tooth formulae were generally either 2.1.3.3 or 2.1.2.3. Fayum anthropoids ranged in size from the very small Qatrania and Biretia (less than 500 g) to the much-larger Aegyptopithecus (approximately 7 kg; 15 lbs.). Fruit was probably the main component of the diet for most or all of the anthropoids, with some of them supplementing with leaves (Kay and Simons 1980; Kirk and Simons 2001; Teaford et al. 1996). Most Fayum anthropoids were probably diurnal above-branch quadrupeds. Some of them (e.g., Apidium; see Figure 9.14) were probably very good leapers (Gebo and Simons 1987), but none show specializations for gibbon-style suspensory locomotion. Some of the Fayum anthropoids are known from hundreds of individuals, permitting the assessment of individual variation, sexual dimorphism, and in some cases growth and development. The description that follows provides greater detail for the two best known Fayum anthropoid families, the Propliopithecidae and the Parapithecidae; the additional families are summarized briefly.
Fayum Anthropoid Families
The Propliopithecidae (see Figure 9.14) include the largest anthropoids from the fauna, and they are known from several crania and some postcranial elements. They have been suggested to be stem catarrhines, although perhaps near the split between catarrhines and platyrrhines. The best known propliopithecid is Aegyptopithecus, known from many teeth, crania, and postcranial elements (Figure 9.17) .
Parapithecidae are an extremely abundant and unusual family of anthropoids from the Fayum. The parapithecid Apidium is known from many jaws with teeth, crushed and distorted crania, and several skeletal elements. Parapithecus is known from cranial material including a beautiful, undistorted cranium. This genus shows extreme reduction of the incisors, including complete absence of the lower incisors in P. grangeri (Simons 2001). This trait is unique among primates. Parapithecids were once thought to be the ancestral stock of platyrrhines; however, their platyrrhine-like features are probably ancestral retentions, so the most conservative approach is to consider them stem anthropoids.
The Proteopithecidae were small frugivores that probably mainly walked along horizontal branches on all fours. They are considered stem anthropoids. The best known genus, Proteopithecus, is represented by dentitions, crania, and postcranial elements.
The Oligopithecidae share a mixture of traits that makes them difficult to classify more specifically within anthropoids. The best known member, Catopithecus, is known from crania that demonstrate a postorbital septum and from mandibles that lack symphyseal fusion. They share the catarrhine tooth formula of 2.1.2.3 and have a canine honing complex that involves the anterior lower premolar. The postcranial elements known for the group suggest generalized arboreal quadrupedalism. The best known member, Catopithecus, is known from crania that demonstrate a postorbital septum and from mandibles that lack symphyseal fusion (Simons and Rasmussen 1996). The jaws are deep, with broad muscle attachment areas and crested teeth. Catopithecus was probably a little less than a kilogram in weight.
Other genera of putative anthropoids from the Fayum include the very poorly known Arsinoea, the contentious Afrotarsius, and the enigmatic Nosmips. The last of these possesses traits of several major primate clades and defies classification (Seiffert et al. 2010).
Early Anthropoid Fossils in Asia
For the last half of the 1900s, researchers believed that Africa was the unquestioned homeland of early anthropoids (see Fleagle and Kay 1994). However, two very different groups of primates from Asia soon began to change that. One was an entirely new discovery (Eosimiidae), and the other was a poorly known group discovered decades prior (Amphipithecidae). Soon, attention on anthropoid origins began to shift eastward (see Ross and Kay 2004; Simons 2004). If anthropoids arose in Asia instead of Africa, then this implies that the African early anthropoids either emigrated from Asia or evolved their anthropoid traits in parallel with living anthropoids.
Eosimiids
First described in the 1990s, the eosimiids are best represented by Eosimias (see Figure 9.14; Figure 9.18). This tiny “dawn monkey” is known from relatively complete jaws with teeth, a few small fragments of the face, and some postcranial elements (Beard et al. 1994; Beard et al. 1996; Gebo et al. 2000). Eosimias (along with the other less-well-known genera in its family) bears some resemblance to tarsiers as well as anthropoids. Unfortunately, no good crania are known for this family, and the anatomy of, for example, the posterior orbital margin could be very revealing as to higher-level relationships.
Amphipithecids
Amphipithecids are small- to medium-size primates (up to 10 kg; 22 lbs.). Most are from the Eocene Pondaung Formation in Myanmar (Early–Middle Eocene), but one genus is known from Thailand. Some dental similarities with anthropoids were noted early on, such as deep jaws and wide basins that separate low molar cusps. The best known genera were Pondaungia and Amphipithecus (Ciochon and Gunnell 2002; see Figure 9.14). Another amphipithecid, Siamopithecus from Thailand, has very rounded molars and was probably a seed-eater (Figure 9.19). In addition to teeth and jaws, some cranial fragments, ankle material, and ends of postcranial bones have been found for Pondaungia. There are important resemblances between the postcranial bones of Pondaungia and those of adapoids, suggesting adapoid affinities for the amphipithecidae. This would imply that the resemblances with anthropoids in the teeth are convergent, based on similarities in diet (see Ciochon and Gunnell 2002). Unfortunately, the association between postcranial bones and teeth is not definite. With other primates in these faunas (including eosimiids), one cannot be certain that the postcranial bones belong with the teeth. Some researchers suggest that some bones belong to a sivaladapid (or asiadapid) and others to an early anthropoid (Beard et al. 2007; Marivaux et al. 2003). Additional well-associated material of amphipithecids would help to clear up this uncertainty.
Platyrrhine Dispersal to South America
Today there is an impressive diversity of primates in South and Central America. These are considered to be part of a single clade, the Platyrrhini. Primates colonized South America sometime in the Eocene from an African source. In the first half of the 20th century, the source of platyrrhines was a matter of major debate among paleontologists, with some favoring a North American origin (e.g., Simpson 1940).
Part of the reason for this debate is that South America was an island in the Eocene. Primates needed to cross open ocean to get there from either North America or Africa, although the distance from the former was shorter. Morphology yields clues to platyrrhine origins. The first known primates in South America have more in common morphologically with African primates than with North American ones. At the time, anthropoids were popping up in North Africa, whereas the only euprimates in North America were adapoids and omomyoids. Despite lacking a bony ear tube, early platyrrhines shared a great deal with other anthropoids, including full postorbital closure and fusion of the mandibular symphysis.
The means by which a population of small North African primates managed to disperse across the Atlantic and survive to colonize South America remains a mystery. The most plausible scenario is one of rafting. That is, primates must have been trapped on vegetation that was blown out to sea by a storm. The vegetation then became a sort of life raft, which eventually landed ashore, dumping its passengers in South America. Rodents probably arrived in South America in the same way (Antoine et al. 2012).
Once ashore, platyrrhines must have crossed South America fairly rapidly because the earliest-known primates from that continent are from Peru (Bond et al. 2015). Soon after that, platyrrhines were in Bolivia, namely Branisella. By the Miocene, platyrrhines were living in extreme southern Argentina and were exploiting a variety of feeding niches. The Early Miocene platyrrhines were all somewhat plesiomorphic in their morphology, but some features that likely arose by ecological convergence suggest (to some) relationships with extant platyrrhine families. This has led to a lively debate about the pattern of primate evolution in South America (Kay 2015; Kay and Fleagle 2010; Rosenberger 2010). By the Middle Miocene, clear representatives of modern families were present in a diverse fauna from La Venta, Colombia (Wheeler 2010). The Plio-Pleistocene saw the emergence of giant platyrrhines as well as several taxa of platyrrhines living on Caribbean islands (Cooke et al. 2016).
The story of platyrrhines seems to be one of amazing sweepstakes dispersal, followed by rapid diversification and widespread geographic colonization of much of South America. After that, dramatic extinctions resulted in the current, much-smaller geographic distribution of platyrrhines. These extinctions were probably caused by changing climates, leading to the contraction of forests. Platyrrhines dispersed to the Caribbean and to Central America, with subsequent extinctions in those regions that might have been related to interactions with humans. Unlike anthropoids of Africa and Asia, platyrrhines do not seem to have evolved any primarily terrestrial forms and so have always been highly dependent on forests.
Special Topic: Jonathan Perry and Primates of the Extreme South
Many primates are very vulnerable to ecological disturbance because they are heavily dependent on fruit to eat and trees to live in. This is one reason why so many primates are endangered today and why many of them went extinct due to climatic and vegetational changes in the past. I (Jonathan Perry) have conducted paleontological research focusing on primates that lived on the edge of their geographic distribution. This research has taken me to extreme environments in the Americas: southern Patagonia, the Canadian prairies, western Wyoming, and the badlands of eastern Oregon.
Santa Cruz Province in Argentina is as far south as primates have ever lived. The Santa Cruz fauna of the Miocene has yielded a moderate diversity of platyrrhines, each with slightly different dietary adaptations. These include Homunculus, first described by Florentino Ameghino in 1891 (Figure 9.20). Recent fieldwork by my colleagues and I in Argentina has revealed several skulls of Homunculus as well as many parts of the skeleton (Kay et al. 2012). The emerging profile of this extinct primate is one of a dedicated arboreal quadruped that fed on fruits and leaves. Many of the foods eaten by Homunculus must have been very tough and were probably covered and impregnated with grit; we suspect this because the cheek teeth are very worn down, even in young individuals, and because the molar tooth roots were very large, presumably to resist strong bite forces (Perry et al. 2010, 2014).
I began working in Argentina while a graduate student at Duke University. I participated as a field assistant in a team led by my Ph.D. advisor, Richard F. Kay, and Argentine colleagues Sergio F. Vizcaíno and M. Susana Bargo. Most of the localities examined belong to a suite of beach sites known since the 1800s and visited by many field parties from various museums in the early 1900s. Since 2003, our international team of paleontologists from the U.S. and Argentina has visited these localities every single year (Figure 9.21). Over time, new fossils and new students have led to new projects and new approaches, including the use of microcomputed tomography (microCT) to visualize and analyze internal structures of the skeleton.

Planet of Apes
Geologic Activity and Climate Change in the Miocene
The Miocene Epoch was a time of mammalian diversification and extinction, global climate change, and ecological turnover. In the Miocene, there was an initial warming trend across the globe with the expansion of subtropical forests, followed by widespread cooling and drying with the retreat of tropical forests and replacement with more open woodlands and eventually grasslands. It was also a time of major geologic activity. On one side of the globe, South America experienced the rise of the Andes Mountains. On the other side, the Indian subcontinent collided with mainland Asia, resulting in the rise of the Himalayan Mountains. In Africa, volcanic activity promoted the development of the East African Rift System. Critical to the story of ape evolution was the exposure of an intercontinental landbridge between East Africa and Eurasia, permitting a true planet of apes (Figure 9.22).
Geographic Distribution: Africa, Asia, Europe
The world of the Miocene had tremendous ape diversity compared to today. The earliest records of fossil apes are from Early Miocene deposits in Africa. However, something dramatic happened around 16 million years ago. With the closure of the ancient Tethys Sea, the subsequent exposure of the Gomphotherium Landbridge, and a period of global warming, the Middle–Late Miocene saw waves of emigration of mammals (including primates) out of Africa and into Eurasia, with evidence of later African re-entry for some (Harrison 2010). Some of the mammals that dispersed from Africa to Eurasia and back were apes. Though most of these early apes left no modern descendants, some of them gave rise to the ancestors of modern apes—including hominins (Figure 9.23).
Where Are the Monkeys? Diversity in the Miocene
Whereas the Oligocene deposits in the Fayum of Egypt have yielded the earliest-known catarrhine fossils, the Miocene demonstrates some diversification of Cercopithecoidea. However, compared to the numerous and diverse Miocene apes (see below), monkeys of the Miocene are very rare and restricted to a single extinct family, the Victoriapithecidae (Figure 9.24). This family contains the earliest definite cercopithecoids. These monkeys are found from northern and eastern Africa between 20 million and 12.5 million years ago (Miller et al. 2009). The best known early African monkey is Victoriapithecus (Figure 9.25), a small-bodied (approximately 7 kg; 15 lbs.), small-brained monkey. Bilophodonty, known to be a hallmark of molar teeth of modern cercopithecoid, was present to some extent in Victoriapithecids. Victoriapithecus has been reconstructed as being more frugivorous and perhaps spent more time on the ground (terrestrial locomotion) than in the trees (arboreal locomotion; Blue et al. 2006). The two major groups of cercopithecoids today are cercopithecines and colobines. The earliest records demonstrating clear members of each of these two groups are at the end of the Miocene. Examples include the early colobine Microcolobus from Kenya and the early cercopithecine Pliopapio from Ethiopia.
|
Family
|
Genera
|
Morphology
|
Location
|
Age1
|
|
Victoriapithecidae2 |
Victoriapithecus |
Long, sloping face. Round, narrowly spaced orbits. Deep cheek bones. Well-developed sagittal crest. |
Africa |
Early to Middle Miocene |
|
Proconsulidae3 |
Proconsul |
Short face. Generalized dentition. Arboreal quadruped. Probably tailless. |
Africa and Arabia |
Early to Middle Miocene |
|
Pongidae |
Gigantopithecus |
Largest primate ever. Deep jaws and low rounded molars. |
Asia |
Miocene to Present |
|
1 Derived from Fleagle 2013. 2 See Benefit and McCrossin 1997 and Fleagle 2013. 3 See Begun 2007. |
|
|||
The Story of Us, the Apes
African Ape Diversity
The Early Miocene of Africa has yielded around 14 genera of early apes (Begun 2003). Many of these taxa have been reconstructed as frugivorous arboreal quadrupeds (Kay 1977). One of the best studied of these genera is the East African Proconsul (Family Proconsulidae; see Figure 9.24). Several species have been described, with body mass reconstructions ranging from 17 to 50 kg (approximately 37–110 lbs.). A paleoenvironmental study reconstructed the habitat of Proconsul to be a dense, closed-canopy tropical forest (Michel et al. 2014). No caudal vertebrae (tail bones) have been found in direct association with Proconsul postcrania, and the morphology of the sacrum is consistent with Proconsul lacking a tail (Russo 2016; Ward et al. 1991).
Overall, the African ape fossil record in the Late Miocene is sparse, with seven fossil localities dating between eleven and five million years ago (Pickford et al. 2009). Nevertheless, most species of great apes live in Africa today. Where did the progenitors of modern African apes arise? Did they evolve in Africa or somewhere else? The paucity of apes in the Late Miocene of Africa stands in contrast to the situation in Eurasia. There, ape diversity was high. Furthermore, several Eurasian ape fossils show morphological affinities with modern hominoids (apes). Because of this, some paleoanthropologists suggest that the ancestors of modern African great apes recolonized Africa from Eurasia toward the end of the Miocene (Begun 2002). However, discoveries of Late Miocene hominoids like the Kenyan Nakalipithecus (9.9 million to 9.8 million years ago), the Ethiopian Chororapithecus (10.7 million to 10.1 million years ago), and the late-Middle Miocene Namibian Otavipithecus (13 million to 12 million years ago) fuel an alternative hypothesis—namely that African hominoid diversity was maintained throughout the Miocene and that one of these taxa might, in fact, be the last common ancestor of extant African apes (Kunimatsu et al. 2007; Mocke et al. 2002). The previously underappreciated diversity of Late Miocene apes in Africa might be due to poor sampling of the fossil record in Africa.
Eurasian Ape Diversity
With the establishment of the Gomphotherium Landbridge (a result of the closure of the Eastern Mediterranean seaway; Rögl 1999), the Middle Miocene was an exciting time for hominoid radiations outside of Africa (see Figure 9.23). Eurasian hominoid species exploited their environments in many different ways in the Miocene. Food exploitation ranged from soft-fruit feeding in some taxa to hard-object feeding in others, in part owing to seasonal fluctuations and the necessary adoptions of fallback foods (DeMiguel et al. 2014). For example, the molars of Oreopithecus bambolii (Family Hominidae) have relatively long lower-molar shearing crests, suggesting that this hominoid was very folivorous (Ungar and Kay 1995). Associated with variation in diet, there is great variation in the degree to which cranial features (e.g., zygomatic bone or supraorbital tori) are developed across the many taxa (Cameron 1997); however, Middle Miocene fossils tend to exhibit relatively thick molar enamel and relatively robust jaws (Andrews and Martin 1991).
In Spain, the cranium with upper dentition, part of a mandible, and partial skeleton of Pliobates (Family Pliobatidae), a small-bodied ape (4–5 kg; 9–11 lbs.), was discovered in deposits dating to 11.6 million years ago (Alba et al. 2015). It is believed to be a frugivore with a relative brain size that overlaps with modern cercopithecoids. The fossilized postcrania of Pliobates suggest that this ape might have had a unique style of locomotion, including the tendency to walk across the branches of trees with its palms facing downward and flexible wrists that permitted rotation of the forearm during climbing. However, the anatomy of the distal humerus differs from those of living apes in ways that suggest that Pliobates was less efficient at stabilizing its elbow while suspended (Benefit and McCrossin 2015). Two other recently described apes from Spain, Pierolapithecus and Anoiapithecus, are known from relatively complete skeletons. Pierolapithecus had a very projecting face and thick molar enamel as well as some skeletal features that suggest (albeit controversially) a less suspensory locomotor style than in extant apes (Moyà-Solà et al. 2004). In contrast to Pierolapithecus, the slightly younger Anoiapithecus has a very flat face (Moyà-Solà et al. 2009).
Postcranial evidence for suspensory or well-developed orthograde behaviors in apes does not appear until the Late Miocene of Europe. Primary evidence supporting these specialized locomotor modes includes the relatively short lumbar vertebrae of Oreopithecus (Figure 9.26) and Dryopithecus (Maclatchy 2004). Further, fossil material of the lower torso of O. bambolii (which dates to the Pan-hominin divergence) conveys a higher degree of flexion-extension abilities in the lumbar region (lower back) than what is possible in extant apes. Additionally, the hindlimb of O. bambolii is suggested to have supported powerful hip adduction during climbing (Hammond et al. 2020). The Late Miocene saw the extinction of most of the Eurasian hominoids in an event referred to as the Vallesian Crisis (Agustí et al. 2003). Among the latest surviving hominoid taxa in Eurasia were Oreopithecus and Gigantopithecus, the latter of which held out until the Pleistocene in Asia and was probably even sympatric with Homo erectus (Cachel 2015).

The Origins of Extant Apes
The fossil record of the extant apes is somewhat underwhelming: it ranges from being practically nonexistent for some taxa (e.g., chimpanzees) to being a little better for others (e.g., humans). There are many possible reasons for these differences in fossil abundance, and many are associated with the environmental conditions necessary for the fossilization of bones. One way to understand the evolution of extant apes that is not so dependent on the fossil record is via molecular evolutionary analyses. This can include counting up the differences in the genetic sequence between two closely related species to estimate the amount of time since these species shared a common ancestor. This is called a molecular clock, and it is often calibrated using fossils of known absolute age that stand in for the last common ancestor of a particular clade. Molecular clock estimates have placed the Hylobatidae and Hominidae split between 19.7 million and 24.1 million years ago, the African ape and Asian ape split between 15.7 million and 19.3 million years ago, and the split of Hylobatidae into its current genera between 6.4 million and 8 million years ago (Israfil et al. 2011).
Small Ape Origins and Fossils
Unfortunately, the fossil record for the small (formerly “lesser”) apes is meager, particularly in Miocene deposits. One possible early hylobatid is Laccopithecus robustus, a Late Miocene catarrhine from China (Harrison 2016). Although it does share some characteristics with modern gibbons and siamangs (including an overall small body size and a short face), Laccopithecus most likely represents a plesiomorphic stem catarrhine and is therefore distantly related to extant apes (Jablonski and Chaplin 2009). A more likely candidate for the hylobatid stem is another Late Miocene taxon from China, Yuanmoupithecus xiaoyuan. Interpretation of its phylogenetic standing, however, is complicated by contradicting dental features—some of them quite plesiomorphic—which some believe best place Yuanmoupithecus as a stem hylobatid (Harrison 2016). Recently, a Middle Miocene Indian fossil ape, Kapi ramnagarensis, has extended the fossil record of small apes by approximately five million years. Its teeth are suggestive of a shift to a more frugivorous diet and it is likely a stem hylobatid (Gilbert et al. 2020). The history of Hylobatidae becomes clearer in the Pleistocene, with fossils representing extant genera.
Great Ape Origins and Fossils
The most extensive fossil record of a modern great ape is that of our own genus, Homo. However, the evolutionary history of the Asian great ape, the orangutan (Pongo), is becoming clearer. Today, orangutans are found only on the islands of Borneo and Sumatra. However, Pleistocene-aged teeth, attributed to Pongo, have been found in Cambodia, China, Laos, Peninsular Malaysia, and Vietnam—demonstrating the vastness of the orangutan’s previous range (Ibrahim et al. 2013; Wang et al. 2014). Sivapithecus from the Miocene of India and Pakistan is represented by many specimens, including parts of the face. Sivapithecus is very similar to Pongo, especially in the face, and it probably is closely related to ancestral orangutans (Pilbeam 1982). Originally, jaws and teeth belonging to the former genus Ramapithecus were thought to be important in the origin of humans (Simons 1961), but now these are recognized as specimens of Sivapithecus (Kelley 2002). Postcranial bones of Sivapithecus, however, suggest a more generalized locomotor mode—including terrestrial locomotion—than seen in Pongo (Pilbeam et al. 1990). Stable carbon and oxygen isotope data from dental enamel have reconstructed the paleoecological space of Sivapithecus (as well as the contemporaneous Late Miocene pongine Khoratpithecus) within the canopies of forested habitats (Habinger et al. 2022).
A probable close relative of Sivapithecus is the amazing Gigantopithecus (see Figure 9.24). Known only from teeth and jaws from China and India (e.g., Figure 9.27), this ape probably weighed as much as 270 kg (595 lbs.) and was likely the largest primate ever (Bocherens et al. 2017). Because of unique features of its teeth (including molarized premolars and patterns of wear) and its massive size, it has been reconstructed as a bamboo specialist, somewhat like the modern panda. Small silica particles (phytoliths) from grasses have been found stuck to the molars of Gigantopithecus (Ciochon et al. 1990). Recent studies evaluating the carbon isotope composition of the enamel sampled from Gigantopithecus teeth suggest that this ape exploited a wide range of vegetation, including fruits, leaves, roots, and bamboo (Bocherens et al. 2017). Its face is reminiscent of that of modern orangutans and it might belong in the same family, Pongidae (Kelley 2002).
In Africa, the first fossil to be confidently attributed to Pan, and known to be the earliest evidence of a chimpanzee, was described based on teeth found in Middle Pleistocene deposits in the Eastern Rift Valley of Kenya (McBrearty and Jablonski 2005). Paleoenvironmental reconstructions of this locality suggest that this early chimpanzee was living in close proximity to early Homo in a closed-canopy wooded habitat. Similarly, fossil teeth and mandibular remains attributed to two species of Middle-Late Miocene apes—Chororapithecus abyssinicus (from Ethiopia; Suwa et al. 2007) and Nakalipithecus nakayamai (from Kenya; Kunimatsu et al. 2007)—have been suggested as basal members of the gorilla clade.
While the deposits of Eastern Africa have yielded a profound record of our fossil hominin ancestors, the continent’s rainforests remain a “palaeontological desert” (Rosas et al. 2022). Clearly, more work is needed to fill in the large gaps in the fossil record of the nonhuman great apes. The twentieth century witnessed the discovery of many hominin fossils in East Africa, which have been critical for improving our understanding of human evolution. While twenty-first-century conservationists fight to prevent the extinction of the living great apes, perhaps efforts by twenty-first-century paleoanthropologists will yield the evolutionary story of these, our closest relatives.
Summary
While there are large gaps in the fossil records linking primates to early hominins, evolutionary trends make it clear that humans are one branch of the broader primate family tree. In this chapter we go over the major development that characterize primate evolution: enhanced vision, grasping hands and feet, greater reliance on social behavior, and increased brain complexity. It is these traits which distinguish primates from other mammals and furthermore, help define major subsections within the primate class, such as strepsirrhines, haplorhines, monkeys, apes, and ultimately hominins.
Within this chapter, we also examine how anthropologists reconstruct these evolutionary relationships. Fossil evidence has provided key information about when and where different primates lived, while genetic data such as skeletal features allow researchers to understand how extinct species moved, ate, and interacted with their environments. Just as crucial are the influences of Earth’s changing environments: continental drift, glacial cycles, and long-term climate shifts have repeatedly reshaped habitats, driving both extinctions and the emergence of new adaptive forms, including the emergence of our own human lineage.
Review Questions
- Compare three major hypotheses about primate origins, making reference to each one’s key ecological reason for primate uniqueness.
- Explain how changes in temperature, rainfall, and vegetation led to major changes in primate biogeography over the Early Tertiary.
- List some euprimate features that plesiadapiforms have and some that they lack.
- Contrast adapoids and omomyoids in terms of life habits.
- Describe one piece of evidence for each of the adapoid, omomyoid, and tarsier origin hypotheses for anthropoids.
- Discuss the biogeography of the origins of African great apes and orangutans using examples from the Miocene ape fossil record.
Key Terms
Adapoidea: Order: Primates. One of the earliest groups of euprimates (true primates; earliest records from the early Eocene).
Adaptive radiations: Rapid diversifications of single lineages into many species which may present unique morphological features in response to different ecological settings.
Ancestral traits: Features that were inherited from a common ancestor and which remain (largely) unchanged.
Anthropoids:Group containing monkeys and apes, including humans.
Auditory bulla: The rounded bony floor of the middle ear cavity.
Bilophodonty: Dental condition in which the cusps of molar teeth form ridges (or lophs) separated from each other by valleys (seen, e.g., in modern catarrhine monkeys).
Catarrhines: Order: Primates; Suborder: Anthropoidea; Infraorder: Catarrhini. Group, with origins in Africa and Asia, that contains monkeys and apes, including humans.
Clade:Group containing all of the descendants of a single ancestor. A portion of a phylogenetic tree represented as a bifurcation (node) in a lineage and all of the branches leading forward in time from that bifurcation.
Convergent evolution: The independent evolution of a morphological feature in animals not closely related (e.g., wings in birds and bats).
Crown: Smallest monophyletic group (clade) containing a specified set of extant taxa and all descendants of their last common ancestor.
Diastema: Space between adjacent teeth.
Diffuse coevolution: The ecological interaction between whole groups of species (e.g., primates) with whole groups of other species (e.g., fruiting trees).
Ectotympanic: Bony ring or tube that holds the tympanic membrane (eardrum).
Euprimates: Order: Primates. True primates or primates of modern aspect.
Haplorhines: Group containing catarrhines, platyrrhines, and tarsiers.
Hominins: Modern humans and any extinct relatives more closely related to us than to chimpanzees.
Mandibular symphysis: Fibrocartilaginous joint between the left and right mandibular segments, located in the midline of the body.
Omomyoidea: Order: Primates; Superfamily: Omomyoidea. One of the earliest groups of euprimates (true primates; earliest record in the early Eocene).
Petrosal bone: The portion of the temporal bone that houses the inner ear apparatus.
Plagiaulacoid: Dental condition where at least one of the lower cheek-teeth (molars or premolars) is a laterally compressed blade.
Platyrrhines: Order: Primates; Suborder: Anthropoidea; Infraorder: Platyrrhini. Group containing monkeys found in the Americas.
Plesiadapiforms: Order: Plesiadapiformes. Archaic primates or primate-like placental mammals (Early Paleocene–Late Eocene).
Plesiomorphic: Having features that are shared by different groups which arose from a common ancestor.
Stem: Taxa that are basal to a given crown group but are more closely related to the crown group than to the closest living sister taxon of the crown group.
Strepsirrhines: Order: Primates; Suborder: Stresirrhini. Group containing lemurs, lorises, and galagos (does not include tarsiers).
Toothcomb: Dental condition found in modern strepsirrhines in which the lower incisors and canines are laterally compressed and protrude forward at a nearly horizontal inclination. This structure is used in grooming.
For Further Exploration
Beard, Chris. 2004. The Hunt for the Dawn Monkey: Unearthing the Origins of Monkeys, Apes, and Humans. Berkeley: University of California Press.
Begun, David R. 2010. “Miocene Hominids and the Origins of the African Apes and Humans.” Annual Review of Anthropology 39: 67–84.
Fleagle, John G. 2013. Primate Adaptation and Evolution. Third edition. San Diego, CA: Academic Press.
Gebo, Daniel L., ed. 1993. Postcranial Adaptations in Nonhuman Primates. Dekalb: Northern Illinois University Press.
Godfrey, Laurie R., and William L. Jungers. 2002. “Quaternary Fossil Lemurs.” In The Primate Fossil Record, edited by Walter C. Hartwig, 97–121. Cambridge: Cambridge University Press.
Godinot, Marc. 2006. “Lemuriform Origins as Viewed from the Fossil Record.” Folia Primatologica 77 (6): 446–464.
Kay, Richard F. 2018. “100 Years of Primate Paleontology.” American Journal of Physical Anthropology 165 (4): 652–676.
Marivaux, Laurent. 2006. “The Eosimiid and Amphipithecid Primates (Anthropoidea) from the Oligocene of the Bugti Hills (Balochistan, Pakistan): New Insight into Early Higher Primate Evolution in South Asia.” Palaeovertebrata, Montpellier 34 (1–2): 29–109.
Martin, R. D. 1990. Primate Origins and Evolution: A Phylogenetic Reconstruction. Princeton: Princeton University Press.
Rose, Kenneth D., Marc Godinot, and Thomas M. Bown. 1994. “The Early Radiation of Euprimates and the Initial Diversification of Omomyidae.” In Anthropoid Origins: The Fossil Evidence, edited by John G. Fleagle and Richard F. Kay, 1–28. New York: Plenum Press.
Ross, Callum F. 1999. “How to Carry Out Functional Morphology.” Evolutionary Anthropology 7 (6): 217–222.
Seiffert, Erik R. 2012. “Early Primate Evolution in Afro-Arabia.” Evolutionary Anthropology: Issues, News, and Reviews 21(6): 239–253.
Szalay, Frederic S., and Eric Delson. 1979. Evolutionary History of the Primates. New York: Academic Press.
Ungar, Peter S. 2002. “Reconstructing the Diets of Fossil Primates.” In Reconstructing Behavior in the Primate Fossil Record, edited by Joseph Plavcan, Richard F. Kay, William Jungers, and Carel P. van Schaik, 261–296. New York: Kluwer Academic/Plenum Publishers.
References
Agustí, J., A. Sanz de Siria, and M. Garcés M. 2003. “Explaining the End of the Hominoid Experiment in Europe.” Journal of Human Evolution 45 (2): 145–153.
Alba, David M., Sergio Almécija, Daniel DeMiguel, Josep Fortuny, Miriam Pérez de los Ríos, Marta Pina, Josep M. Robles, and Salvador Moyà-Solà. 2015. “Miocene Small-Bodied Ape from Eurasia Sheds Light on Hominoid Evolution.” Science 350 (6260): aab2625.
Andrews, Peter, and Lawrence Martin. 1991. “Hominoid Dietary Evolution.” Philosophical Transactions of the Royal Society of London B: Biological Sciences 334 (1270): 199–209.
Antoine, Pierre-Oliver, Laurent Marivaux, Darren A. Croft, Guillaume Billet, Morgan Ganerød, Carlos Jaramillo, Thomas Martin, et al. 2012. “Middle Eocene Rodents from Peruvian Amazonia Reveal the Pattern and Timing of Caviomorph Origins and Biogeography.” Proceedings of the Royal Society B: Biological Sciences 279 (1732): 1319–1326.
Beard, K. Christopher. 1990. “Gliding Behaviour and Palaeoecology of the Alleged Primate Family Paromomyidae (Mammalia, Dermoptera).” Nature 345 (6273): 340–341.
Beard, K. Christopher. 2002. “Basal Anthropoids.” In The Primate Fossil Record, edited by William C. Hartwig, 133–150. Cambridge: Cambridge University Press.
Beard, K. Christopher, and R. D. E. MacPhee. 1994. “Cranial Anatomy of Shoshonius and the Antiquity of Anthropoidea.” In Anthropoid Origins: The Fossil Evidence, edited by John G. Fleagle and Richard F. Kay, 55–98. New York: Plenum Press.
Beard, K. Christopher, Laurent Marivaux, Soe Thura Tun, Aung Naing Soe, Yaowalak Chaimanee, Wanna Htoon, Bernard Marandat, Htun Htun Aung, and Jean-Jacques Jaeger. 2007. “New Sivaladapid Primates from the Eocene Pondaung Formation of Myanmar and the Anthropoid Status of Amphipithecidae.” Bulletin of Carnegie Museum of Natural History 39: 67–76.
Beard, K. Christopher, Tao Qi, Mary R. Dawson, Banyue Wang, and Chuankuei Li. 1994. “A Diverse New Primate Fauna from Middle Eocene Fissure-Fillings in Southeastern China.” Nature 368 (6472): 604–609.
Beard, K. Christopher, Yongsheng Tong, Mary R. Dawson, Jingwen Wang, and Xueshi Huang. 1996. “Earliest Complete Dentition of an Anthropoid Primate from the Late Middle Eocene of Shanxi Province, China.” Science 272 (5258): 82–85.
Beecher, Robert M. 1983. “Evolution of the Mandibular Symphysis in Notharctinae (Adapidae, Primates).” International Journal of Primatology 4 (1): 99–112.
Begun, David R. 2002. “European Hominoids.” In The Primate Fossil Record, edited by William C. Hartwig, 339–368. Cambridge: Cambridge University Press.
Begun, David R. 2003. “Planet of the Apes.” Scientific American 289 (2): 74–83.
Begun, David R. 2007. “Fossil Record of Miocene Hominoids.” In Handbook of Paleoanthropology, edited by Winfried Henke and Ian Tattersall, 921–977. New York: Springer.
Benefit, Brenda R., and Monte L. McCrossin. 1997. “Earliest Known Old World Monkey Skull.” Nature 388 (6640): 368–371.
Benefit, Brenda R., and Monte L. McCrossin. 2015. “A Window into Ape Evolution.” Science 350 (6260): 515–516.
Bloch, Jonathan I., and David M. Boyer. 2002. “Grasping Primate Origins.” Science 298 (5598): 1606–1610.
Bloch, Jonathan I., and David M. Boyer. 2007. “New Skeletons of Paleocene-Eocene Plesiadapiformes: A Diversity of Arboreal Positional Behaviors in Early Primates.” In Primate Origins: Adaptations and Evolution, edited by Matthew J. Ravosa and Marian Dagosto, 535–581. New York: Springer.
Bloch, Jonathan I., and Mary T. Silcox. 2006. “Cranial Anatomy of the Paleocene Plesiadapiform Carpolestes simpsoni (Mammalia, Primates) Using Ultra High-Resolution X-ray Computed Tomography, and the Relationships of Plesiadapiforms to Euprimates.” Journal of Human Evolution: 50 (1): 1–35.
Blue, Kathleen T., Monte L. McCrossin, and Brenda R. Benefit. 2006. “Terrestriality in a Middle Miocene Context: Victoriapithecus from Maboko, Kenya.” In Human Origins and Environmental Backgrounds, edited by Hidemi Ishida, Russell Tuttle, Martin Pickford, Naomichi Ogihara, and Masato Nakatsukasa, 45–58. New York: Springer.
Bocherens, Hervé, Friedemann Schrenk, Yaowalak Chaimanee, Ottmar Kullmer, Doris Mörike, Diana Pushkina, and Jean-Jacques Jaeger. 2017. “Flexibility of Diet and Habitat in Pleistocene South Asian Mammals: Implications for the Fate of the Giant Fossil Ape Gigantopithecus.” Quaternary International 434 (A): 148–155.
Bond, Mariano, Marcelo F. Tejedor, Kenneth E. Campbell Jr., Laura Chornogubsky, Nelson Novo, and Francisco Goin. 2015. “Eocene Primates of South America and the African Origins of New World Monkeys.” Nature 520 (7548): 539–541.
Bown, T. M., and M. J. Kraus. 1988. “Geology and Paleoenvironment of the Oligocene Jebel Qatrani Formation and Adjacent Rocks, Fayum Depression, Egypt.” Professional Paper, 1452. Washington, DC: U.S. Geological Survey Professional Papers.
Cachel, Susan. 2015. Fossil Primates. Vol. 69. Cambridge: Cambridge University Press.
Cameron, David W. 1997. “A Revised Systematic Scheme for the Eurasian Miocene Fossil Hominidae.” Journal of Human Evolution 33 (4): 449–477.
Cartmill, Matt. 1972. “Arboreal Adaptations and the Origin of the Order Primates.” In The Functional and Evolutionary Biology of Primates, edited by Russell Tuttle, 97–122. Chicago: Aldine-Atherton.
Cartmill, Matt. 1974. “Rethinking Primate Origins.” Science 184 (4135): 436–443.
Cartmill, Matt, and Richard F. Kay. 1978. “Craniodental Morphology, Tarsier Affinities, and Primate Suborders.” In Recent Advances in Primatology: Evolution, edited by D. J. Chivers and K. A. Joysey, 205–214. London: Academic Press.
Casanovas-Vilar, Isaac, David M. Alba, Miguel Garcés, Josep M. Robles, and Salvador Moyà-Solà. 2011. “Updated Chronology for the Miocene Hominoid Radiation in Western Eurasia.” Proceedings of the National Academy of Sciences 108 (14): 5554-5559. https://doi:10.1073/pnas.1018562108.
Chaimanee, Yaowalak, Olivier Chavasseau, K. Christopher Beard, Aung Aung Kyaw, Aung Naing Soe, Chit Sein, Vincent Lazzari, et al. 2012. “Late Middle Eocene Primate from Myanmar and the Initial Anthropoid Colonization of Africa.” Proceedings of the National Academy of Sciences of the United States of America 109 (26): 10293–10297.
Chester, Stephen G. B., Jonathan I. Bloch, Doug M. Boyer, and William A. Clemens. 2015. “Oldest Known Euarchontan Tarsals and Affinities of Paleocene Purgatorius to Primates.” Proceedings of the National Academy of Sciences of the United States of America 112 (5): 1487–1492.
Ciochon, Russell L., and Gregg F. Gunnell. 2002. “Chronology of Primate Discoveries in Myanmar: Influences on the Anthropoid Origins Debate.” Yearbook of Physical Anthropology 45(S35): 2–35.
Ciochon, R. L., D. R. Piperno, and R. G. Thompson. 1990. “Opal Phytoliths Found on the Teeth of the Extinct Ape Gigantopithecus blacki: Implications for Paleodietary Studies.” Proceedings of the National Academy of Sciences of the United States of America 87 (20): 8120–8124.
Clemens, William A. 2004. “Purgatorius (Plesiadapiformes, Primates?, Mammalia), a Paleocene Immigrant into Northeastern Montana: Stratigraphic Occurrences and Incisor Proportions.” Bulletin of Carnegie Museum of Natural History 36: 3–13.
Cooke, Siobhán B., Justin T. Gladman, Lauren B. Halenar, Zachary S. Klukkert, and Alfred L. Rosenberber. 2016. “The Paleobiology of the Recently Extinct Platyrrhines of Brazil and the Caribbean.” In Molecular Population Genetics, Evolutionary Biology and Biological Conservation of Neotropical Primates, edited by Manuel Ruiz-Garcia and Joseph Mark Shostell, 41–89. New York: Nova Publishers.
DeLeon, Valerie B., Timothy D. Smith, and Alfred L. Rosenberger. 2016. “Ontogeny of the Postorbital Region in Tarsiers and Other Primates.” Anatomical Record: Advances in Integrative Anatomy and Evolutionary Biology 299 (12): 1631–1645.
DeMiguel, Daniel, David M. Alba, and Salvador Moyà-Solà. 2014. “Dietary Specialization during the Evolution of Western Eurasian Hominoids and the Extinction of European Great Apes.” PLoS ONE 9 (5): e97442. https://doi.org/10.1371/journal.pone.0097442.
Dunn, Rachel H., Kenneth D. Rose, Rajendra Rana, Kishore Kumar, Ashok Sahni, and Thierry Smith. 2016. “New Euprimate Postcrania from the Early Eocene of Gujarat, India, and the Strepsirrhine–Haplorhine Divergence.” Journal of Human Evolution 99: 25–51.
Fleagle, John G. 2013. Primate Adaptation and Evolution, Third Edition. San Diego, CA: Academic Press.
Fleagle, John G., and Richard F. Kay. 1994. Anthropoid Origins. New York: Plenum Press.
Franzen, Jens Lorenz, Phillip D. Gingerich, Jörg Habersetzer, Jørn Hurum, von Wighart Koenigswald, and B. Holly Smith. 2009. “Complete Primate Skeleton from the Middle Eocene of Messel in Germany: Morphology and Paleobiology.” PLoS ONE 4 (5): e5723. doi:10.1371/journal.pone.0005723.
Gebo, Daniel L., Marian Dagosto, K. Christopher Beard, Tao Qi, and Jingwen Wang. 2000. “The Oldest Known Anthropoid Postcranial Fossils and the Early Evolution of Higher Primates.” Nature 404 (6775): 276–278.
Gebo, Daniel L., and Elwyn L. Simons. 1987. “Morphology and Locomotor Adaptations of the Foot in Early Oligocene Anthropoids.” American Journal of Physical Anthropology 74 (1): 83–101.
Gilbert, Christopher C., Alejandra Ortiz, Kelsey D. Pugh, Christopher J. Campisano, Biren A. Patel, Ningthoujam Premjit Singh, John G. Fleagle, and Rajeev Patnaik. 2020. “New Middle Miocene Ape (Primates: Hylobatidae) from Ramnagar, India, Fills Major Gaps in the Hominoid Fossil Record.” Proceedings of the Royal Society B 287(1934): 20201655.
Gingerich, P. D. 1980. “Eocene Adapidae, Paleobiogeography, and the Origin of South American Platyrrhini.” In Evolutionary Biology of the New World Monkeys and Continental Drift, edited by Russell L. Ciochon and A. Brunetto Chiarelli, 123–138. New York: Plenum Press.
Godfrey, Laurie R., and William L. Jungers. 2002. “Quaternary Fossil Lemurs.” In The Primate Fossil Record, edited by Walter C. Hartwig, 97–121. Cambridge: Cambridge University Press.
Godinot, Marc. 2006. “Lemuriform Origins as Viewed from the Fossil Record.” Folia Primatologica 77 (6): 446–464.
Gregory, William K. 1920. “On the Structure and Relations of Notharctus, an American Eocene Primate.” Memoirs of the American Museum of Natural History (N.S.) 3 (2).
Gunnell, Gregg F., Doug M. Boyer, Anthony R. Friscia, Steven Heritage, Frederik Kyalo Manthi, Ellen R. Miller, Hesham M. Sallam, Nancy B. Simmons, Nancy J. Stevens, and Erik R. Seiffert. 2018. “Fossil Lemurs from Egypt and Kenya Suggest an African Origin for Madagascar’s Aye-aye.” Nature Communications 9 (3193): 1–12.
Habinger, S. G., O. Chavasseau, J. J. Jaeger, Y. Chaimanee, A. N. Soe, C. Sein, and H. Bocherens. 2022. “Evolutionary Ecology of Miocene Hominoid Primates in Southeast Asia.” Scientific Reports 12 (1): 1–12.
Hammond, Ashley, Lorenzo Rook, Alisha D.Anaya, Elisabetta Cioppi, Loïc Costeur, Salvadore Moyà-Solà, and Sergio Almécija. 2020. “Insights into the Lower Torso in Late Miocene Hominoid Oreopithecus bambolii.” Proceedings of the National Academy of Sciences 117 (1): 278–284.
Harrison, Terry. 2010. “Apes among the Tangled Branches of Human Origins.” Science 327 (5965): 532–534.
Harrison, Terry. 2016. “The Fossil Record and Evolutionary History of Hylobatids.” In Evolution of Gibbons and Siamang, edited by Ullrich H. Reichard, Hirohisa Hirai, and Claudia Barelli, 91–110. New York: Springer.
Ibrahim, Yasamin Kh., Lim Tze Tshen, Kira E. Westaway, Earl of Cranbrook, Louise Humphrey, Ross Fatihah Muhammad, Jian-xin Zhao, and Lee Chai Peng. 2013. “First Discovery of Pleistocene Orangutan (Pongo sp.) Fossils in Peninsular Malaysia: Biogeographic and Paleoenvironmental Implications.” Journal of Human Evolution 65 (6): 770–797.
Israfil, Hulya, Sarah M. Zehr, Alan R. Mootnick, Maryellen Ruvolo, and Michael E. Steiper. 2011. “Unresolved Molecular Phylogenies of Gibbons and Siamangs (Family: Hylobatidae) Based on Mitochondrial, Y-linked, and X-linked Loci Indicate a Rapid Miocene Radiation or Sudden Vicariance Event.” Molecular Phylogenetics and Evolution 58 (3): 447–455.
Jablonski, Nina G., and George Chaplin. 2009. “The Fossil Record of Gibbons.” In The Gibbons, edited by Danielle Whittaker and Susan Lappan, 111–130. New York: Springer.
Jones, F. Wood. 1916. Arboreal Man. London: Edward Arnold.
Kay, Richard F. 1977. “Diets of Early Miocene African Hominoids.” Nature 268 (5621): 628–630.
Kay, Richard F. 2015. “Biogeography in Deep Time: What Do Phylogenetics, Geology, and Paleoclimate Tell Us about Early Platyrrhine Evolution?” Molecular Phylogenetics and Evolution 82 (B): 358–374.
Kay, Richard F., and John G. Fleagle. 2010. “Stem Taxa, Homoplasy, Long Lineages, and the Phylogenetic Position of Dolichocebus.” Journal of Human Evolution 59 (2): 218–222.
Kay, Richard F., Jonathan M. G. Perry, Michael Malinzak, Kari L. Allen, E. Christopher Kirk, J. Michael Plavcan, and John G. Fleagle. 2012. “Paleobiology of Santacrucian Primates.” In Early Miocene Paleobiology in Patagonia: High-Latitude Paleocommunities of the Santa Cruz Formation, edited by Sergio F. Vizcaíno, Richard F. Kay, and M. Susana Bargo, 306–330. Cambridge: Cambridge University Press.
Kay, Richard F., Daniel O Schmitt, Christopher J. Vinyard, Jonathan M. G. Perry, Nobuo Shigehara, Masanaru Takai, and Naoko Egi. 2004. “The Paleobiology of Amphipithecidae, South Asian Late Eocene Primates.” Journal of Human Evolution 46 (1): 3–25.
Kay, Richard F., and Elwyn L. Simons. 1980. “The Ecology of Oligocene African Anthropoidea.” International Journal of Primatology 1 (1): 21–37.
Kay, Richard F., Richard W. Thorington, and Peter Houde. 1990. “Eocene Plesiadapiform Shows Affinities with Flying Lemurs Not Primates.” Nature 345 (6273): 342–344.
Kelley, Jay. 2002. “The Hominoid Radiation in Asia.” In The Primate Fossil Record, edited by Walter C. Hartwig, 369–384. Cambridge: Cambridge University Press.
Kirk, E. Christopher, and Elwyn L. Simons. 2001. “Diets of Fossil Primates from the Fayum Depression of Egypt: A Quantitative Analysis of Molar Shearing.” Journal of Human Evolution 40 (3): 203–229.
Kirk, E. Christopher, and Blythe A. Williams. 2011. “New Adapiform Primate of Old World Affinities from the Devil’s Graveyard Formation of Texas.” Journal of Human Evolution 61 (2): 156–168.
Krause, David W. 1991. “Were Paromomyids Gliders? Maybe, Maybe Not.” Journal of Human Evolution 21 (3): 177–188.
Kunimatsu, Yutaka, Masato Nakatsukasa, Yoshihiro Sawada, Tetsuya Sakai, Masayuki Hyodo, Hironobu Hyodo, Tetsumaru Itaya, et al. 2007. “A New Late Miocene Great Ape from Kenya and Its Implications for the Origins of African Great Apes and Humans.” Proceedings of the National Academy of Sciences of the United States of America 104 (49): 19220–19225.
Maclatchy, Laura. 2004. “The Oldest Ape.” Evolutionary Anthropology: Issues, News, and Reviews 13 (3): 90–103.
Marivaux, Laurent, Yaowalak Chaimanee, Stéphane Ducrocq, Bernard Marandat, Jean Sudre, Aung Naing Soe, Soe Thura Tun, Wanna Htoon, and Jean-Jacques Jaeger. 2003. “The Anthropoid Status of a Primate from the Late Middle Eocene Pondaung Formation (Central Myanmar): Tarsal Evidence.” Proceedings of the National Academy of Sciences of the United States of America 100 (23): 13173–13178.
Marivaux, Laurent, Anusha Ramdarshan, El Mabrouk Essid, Wissem Marzougui, Hayet Khayati Ammar, Renaud Lebrun, Bernard Marandat, Gilles Merzeraud, Rodolphe Tabuce, and Monique Vianey-Liaud. 2013. “Djebelemur, a Tiny Pre-ToothCombed Primate from the Eocene of Tunisia: A Glimpse into the Origin of Crown Strepsirrhines.” PLoS ONE 8 (12): e80778. doi.org/10.1371/journal.pone.0080778.
Martin, R. D. 1968. “Towards a New Definition of Primates.” Man (N.S.) 3 (3): 377–401.
Martin, R. D. 1972. “Adaptive Radiation and Behaviour of the Malagasy Primates.” Philosophical Transactions of the Royal Society B: Biological Sciences 264 (862): 295–352.
Martin, R. D. 1990. Primate Origins and Evolution, a Phylogenetic Reconstruction. Princeton: Princeton University Press.
McBrearty, Sally, and Nina G. Jablonski. 2005. “First Fossil Chimpanzee.” Nature 437 (7055): 105–108.
Michel, Lauren A., Daniel J. Peppe, James A. Lutz, Stephen G. Driese, Holly M. Dunsworth, William E. H. Harcourt-Smith, William H. Horner, Thomas Lehmann, Sheila Nightingale, and Kieran P. McNulty. 2014. “Remnants of an Ancient Forest Provide Ecological Context for Early Miocene Fossil Apes.” Nature Communications 5: 1-9.
Miller, E. R., B. R. Benefit, M. L. McCrossin, J. M. Plavcan, M. G. Leakey, A. N. El-Barkooky, M. A. Hamdan, M. K. A. Gawad, S. M. Hassan, and E. L. Simons. 2009. “Systematics of Early and Middle Miocene Old World Monkeys.” Journal of Human Evolution 57 (3): 195–211.
Mocke, H., M. Pickford, B. Senut, and D. Gommery. 2022. “New Information about African Late Middle Miocene to Latest Miocene (13–5.5 Ma) Hominoidea. Communications of the Geological Survey of Namibia 24: 33–66.
Moyà-Solà, Salvadore, David M. Alba, Sergio Almécija, Isaac Casanovas-Vilar, Meike Köhler, Soledad De Esteban-Trivigno, Josep M. Robles, Jordi Galindo, and Josep Fortuny. 2009. “A Unique Middle Miocene European Hominoid and the Origins of the Great Ape and Human Clade.” Proceedings of the National Academy of Sciences of the United States of America 106 (24): 9601–9606.
Moyà-Solà, Salvador, Meike Köhler, David M. Alba, Isaac Casanovas-Vilar, and Jordi Galindo. 2004. “Pierolapithecus catalaunicus, a New Middle Miocene Great Ape from Spain.” Science 306 (5700): 1339–1344.
Ni, Xijun, Daniel L. Gebo, Marian Dagosto, Jin Meng, Paul Tafforeau, John J. Flynn, and K. Christopher Beard. 2013. “The Oldest Known Primate Skeleton and Early Haplorhine Evolution.” Nature 498 (7452): 60–64.
Perry, Jonathan M. G., Richard F. Kay, Sergio F. Vizcaíno, and M. Susana Bargo. 2010. “Tooth Root Size, Chewing Muscle Leverage, and the Biology of Homunculus patagonicus (Primates) from the Late Early Miocene of Patagonia.” Ameghiniana 47 (3): 355–371.
Perry, Jonathan M. G., Richard F. Kay, Sergio F. Vizcaíno, and M. Susana Bargo. 2014. “Oldest Known Cranium of a Juvenile New World Monkey (Early Miocene, Patagonia, Argentina): Implications for the Taxonomy and the Molar Eruption Pattern of Early Platyrrhines.” Journal of Human Evolution 74: 67–81.
Pickford, Martin, Yves Coppens, Brigitte Senut, Jorge Morales, and José Braga. 2009. “Late Miocene Hominoid from Niger.” Comptes Rendus Palevol 8 (4): 413–425.
Pilbeam, David. 1982. “New Hominoid Skull Material from the Miocene of Pakistan.” Nature 295 (5846): 232–234.
Pilbeam, David, Michael D. Rose, John C. Barry, and S. M. Ibrahim Shah. 1990. “New Sivapithecus Humeri from Pakistan and the Relationship of Sivapithecus and Pongo.” Nature 348 (6298): 237–239.
Rasmussen, D. Tab. 1990. “Primate Origins: Lessons from a Neotropical Marsupial.” American Journal of Primatology 22 (4): 263–277.
Ravosa, Matthew J. 1996. “Mandibular Form and Function in North American and European Adapidae and Omomyidae.” Journal of Morphology 229 (2): 171–190.
Rögl, Fred. 1999. “Mediterranean and Paratethys Palaeogeography during the Oligocene and Miocene.” In Hominoid Evolution and Climatic Change in Europe, edited by Jorge Agustí, Lorenzo Rook, and Peter Andrews, 8–22. Cambridge: Cambridge University Press.
Rosas, A., A. García-Tabernero, D. Fidalgo, M. Fero Meñe, C. Ebana Ebana, F. Esono Mba, and P. Saladie. 2022. “The Scarcity of Fossils in the African Rainforest: Archaeo-Paleontological Surveys and Actualistic Taphonomy in Equatorial Guinea.” Historical Biology 34 (8): 1–9.
Rose, Kenneth D., and Thomas M. Bown. 1984. “Gradual Phyletic Evolution at the Generic Level in Early Eocene Omomyoid Primates.” Nature 309 (5965): 250–252.
Rose, Kenneth D., Rachel H. Dunn, Kishor Kumar, Jonathan M. G. Perry, Kristen A. Prufrock, Rajendra S. Rana, and Thierry Smith. 2018. “New Fossils from Tadkeshwar Mine (Gujarat, India) Increase Primate Diversity from the Early Eocene Cambay Shale.” Journal of Human Evolution 122: 93–107.
Rose, Kenneth D., and John M. Rensberger. 1983. “Upper Dentition of Ekgmowechashala (Omomyoid Primate) from the John Day Formation, Oligo-Miocene of Oregon.” Folia Primatologica 41(1-2): 102–111.
Rosenberger, Alfred L. 2010. “Platyrrhines, PAUP, Parallelism, and the Long Lineage Hypothesis: A Reply to Kay et al. (2008).” Journal of Human Evolution 59 (2): 214–217.
Ross, Callum F. 2000. “Into the Light: The Origins of Anthropoidea.” Annual Review of Anthropology 29: 147–194.
Ross, Callum F., and Richard F. Kay, eds. 2004. Anthropoid Origins: New Visions. New York: Kluwer Academic/Plenum Publishers.
Russo, Gabrielle A. 2016. “Comparative Sacral Morphology and the Reconstructed Tail Lengths of Five Extinct Primates: Proconsul heseloni, Epipliopithecus vindobonensis, Archaeolemur edwardsi, Megaladapis grandidieri, and Palaeopropithecus kelyus.” Journal of Human Evolution 90: 135–162.
Schmid, Peter. 1979. “Evidence of Microchoerine Evolution from Dielsdorf (Zürich Region, Switzerland): A Preliminary Report.” Folia Primatologica 31 (4): 301–311.
Seiffert, Erik R. 2012. “Early Primate Evolution in Afro-Arabia.” Evolutionary Anthropology: Issues, News, and Reviews 21 (6): 239–253.
Seiffert, Erik R., Jonathan M. G. Perry, Elwyn L. Simons, and Doug M. Boyer. 2009. “Convergent Evolution of Anthropoid-like Adaptations in Eocene Adapiform Primates.” Nature 461 (7267): 1118–1121.
Seiffert, Erik R., Elwyn L. Simons, and Yousry Attia. 2003. “Fossil Evidence for an Ancient Divergence of Lorises and Galagos.” Nature 422 (6930): 421–424.
Seiffert, Erik R., Elwyn L. Simons, Doug M. Boyer, Jonathan M. G. Perry, Timothy M. Ryan, and Hesham M. Sallam. 2010. “A Fossil Primate of Uncertain Affinities from the Earliest Late Eocene of Egypt.” Proceedings of the National Academy of Sciences of the United States of America 107 (21): 9712–9717.
Seiffert, Erik R., Elwyn L. Simons, and Cornelia V. M. Simons. 2004. “Phylogenetic, Biogeographic, and Adaptive Implications of New Fossil Evidence Bearing on Crown Anthropoid Origins and Early Stem Catarrhine Evolution.” In Anthropoid Origins: New Visions, edited by Callum F. Ross and Richard F. Kay, 157–182. New York: Kluwer/Plenum Publishing.
Simons, Elwyn L. 1961. “The Phyletic Position of Ramapithecus.” Postilla 57: 1–9.
Simons, Elwyn L. 2001. “The Cranium of Parapithecus grangeri, an Egyptian Oligocene Anthropoidean Primate.” Proceedings of the National Academy of Sciences of the United States of America 98 (4): 7892–7897.
Simons, Elwyn L. 2004. “The Cranium and Adaptations of Parapithecus grangeri, a Stem Anthropoid From the Fayum Oligocene of Egypt.” In Anthropoid Origins: New Visions, edited by Callum F. Ross and Richard F. Kay, 183–204. New York: Kluwer/Plenum Publishing.
Simons, Elwyn L. 2008. “Eocene and Oligocene Mammals of the Fayum, Egypt.” In Elwyn Simons: A Search for Origins, edited by John G. Fleagle and Christopher C. Gilbert, 87–105. New York: Springer.
Simons, Elwyn L., and D. Tab Rasmussen. 1994a. “A Remarkable Cranium of Plesiopithecus teras (Primates, Prosimii) from the Eocene of Egypt.” Proceedings of the National Academy of Sciences of the United States of America 91(21): 9946–9950.
Simons, Elwyn L., and D. Tab Rasmussen. 1994b. “A Whole New World of Ancestors: Eocene Anthropoideans from Africa.” Evolutionary Anthropology 3 (4): 128–139.
Simons, Elwyn L., and D. Tab Rasmussen. 1996. “Skull of Catopithecus browni, an Early Tertiary Catarrhine.” American Journal of Physical Anthropology 100 (2): 261–292.
Simons, Elwyn L., and Erik R. Seiffert. 1999. “A Partial Skeleton of Proteopithecus sylviae (Primates Anthropoidea): First Associated Dental and Postcranial Remains of an Eocene Anthropoidean.” Comptes Rendus de l'Académie des Sciences, Paris 329 (12): 921–927.
Simons, Elwyn L., Erik R. Seiffert, Timothy M. Ryan, and Yousry Attia. 2007. “A Remarkable Female Cranium of the Early Oligocene Anthropoid Aegyptopithecus zeuxis (Catarrhini, Propliopithecidae).” Proceedings of the National Academy of Sciences of the United States of America 104 (21): 8731–8736.
Simpson, George Gaylord. 1933. “The ‘Plagiaulacoid’ Type of Mammalian Dentition: A Study of Convergence.” Journal of Mammalogy 14 (2): 97–107.
Simpson, George Gaylord. 1940. “Review of the Mammal-Bearing Tertiary of South America.” Proceedings of the American Philosophical Society 83 (5): 649–709.
Simpson, George Gaylord. 1967. “The Tertiary Lorisiform Primates of Africa.” Bulletin of the Museum of Comparative Zoology at Harvard University 136: 39–62.
Smith, G. Elliot. 1912. “The Evolution of Man.” Smithsonian Institute Annual Report 2012: 553–572.
Smith, Thierry, Kenneth D. Rose, and Philip D. Gingerich. 2006. “Rapid Asia–Europe–North America Geographic Dispersal of Earliest Eocene Primate Teilhardina during the Paleocene–Eocene Thermal Maximum.” Proceedings of the National Academy of Sciences of the United States of America 103 (30): 11223–11227.
Stehlin, Hans G. 1912. “Die säugetiere des schweizerischen Eocaens. Siebenter teil, erst hälfte: Adapis” [“The Mammals of the Swiss Eocene. Part Seven, First Half: Adapis”]. Abhandlungen der Schweizerischen Paläontologischen Gesellschaft 38: 1165–1298.
Strait, Suzanne G. 2001. “Dietary Reconstruction of Small-Bodied Omomyoid Primates.” Journal of Vertebrate Paleontology 21 (2): 322–334.
Sussman, Robert W. 1991. “Primate Origins and the Evolution of Angiosperms.” American Journal of Primatology 23 (4): 209–223.
Suwa, Gen, Reiko T. Kono, Shigehiro Katoh, Berhane Asfaw, and Yonas Beyene. 2007. “A New Species of Great Ape from the Late Miocene Epoch in Ethiopia.” Nature 448 (7156): 921–924.
Teaford, Mark F., Mary C. Maas, and Elwyn L. Simons. 1996. “Dental Microwear and Microstructure in Early Oligocene Primates from the Fayum, Egypt: Implications for Diet.” American Journal of Physical Anthropology 101 (4): 527–543.
Ungar, Peter S., and Richard F. Kay. 1995. “The Dietary Adaptations of European Miocene Catarrhines.” Proceedings of the National Academy of Sciences of the United States of America 92 (12): 5479–5481.
Wang, Cui-Bin, Ling-Xia Zhao, Chang-Zhu Jin, Yuan Wang, Da-Gong Qin, and Wen-Shi Pan. 2014. “New Discovery of Early Pleistocene Orangutan Fossils from Sanhe Cave in Chongzuo, Guangxi, Southern China.” Quaternary International 354: 68–74.
Ward, C. V., A. Walker, and M. F. Teaford. 1991. “Proconsul Did Not Have a Tail.” Journal of Human Evolution 21 (3): 215–220.
Wheeler, Brandon C. 2010. “Community Ecology of the Middle Miocene Primates of La Venta, Colombia: The Relationship between Ecological Diversity, Divergence Time, and Phylogenetic Richness.” Primates 51 (2): 131–138.
Williams, Blythe A., and Richard F. Kay. 1995. “The Taxon Anthropoidea and the Crown Clade Concept.” Evolutionary Anthropology 3 (6): 188–190.
Williams, Blythe A., Richard F. Kay, and E. Christopher Kirk. 2010a. “New Perspectives on Anthropoid Origins.” Proceedings of the National Academy of the United States of America 107 (11): 4797–4804.
Williams, Blythe A., Richard F. Kay, E. Christopher Kirk, and Callum F. Ross. 2010b. “Darwinius masillae Is a European Middle Eocene Stem Strepsirrhine—A Reply to Franzen et al.” Journal of Human Evolution 59(5): 567–573.
Wilson Mantilla, G. P., S. G. B. Chester, W. A. Clemens, J. R. Moore, C. J. Sprain, B. T. Hovatter, W. S. Mitchell, W. W. Mans, R. Mundil, and P. R. Renne. 2021. “Earliest Palaeocene Purgatoriids and the Initial Radiation of Stem Primates.” Royal Society Open Science 8(2):210050. doi:10.1098/rsos.210050.
Acknowledgments
We are immensely grateful to the editors of this book, Drs. Beth Shook, Lara Braff, Katie Nelson, and Kelsie Aguilera, for their time and commitment to making this knowledge freely accessible to all, and for giving us the opportunity to participate in this important project.

